Characterization and Biotechnological Potential of Halophilic Fungi Producing Salt-Tolerant Amylase and Protease from Marine Environments
DOI:
https://doi.org/10.48048/tis.2026.12614Keywords:
Halophilic fungi, Amylase, Protease, Salt tolerance, Detergent enzyme, BiocatalystAbstract
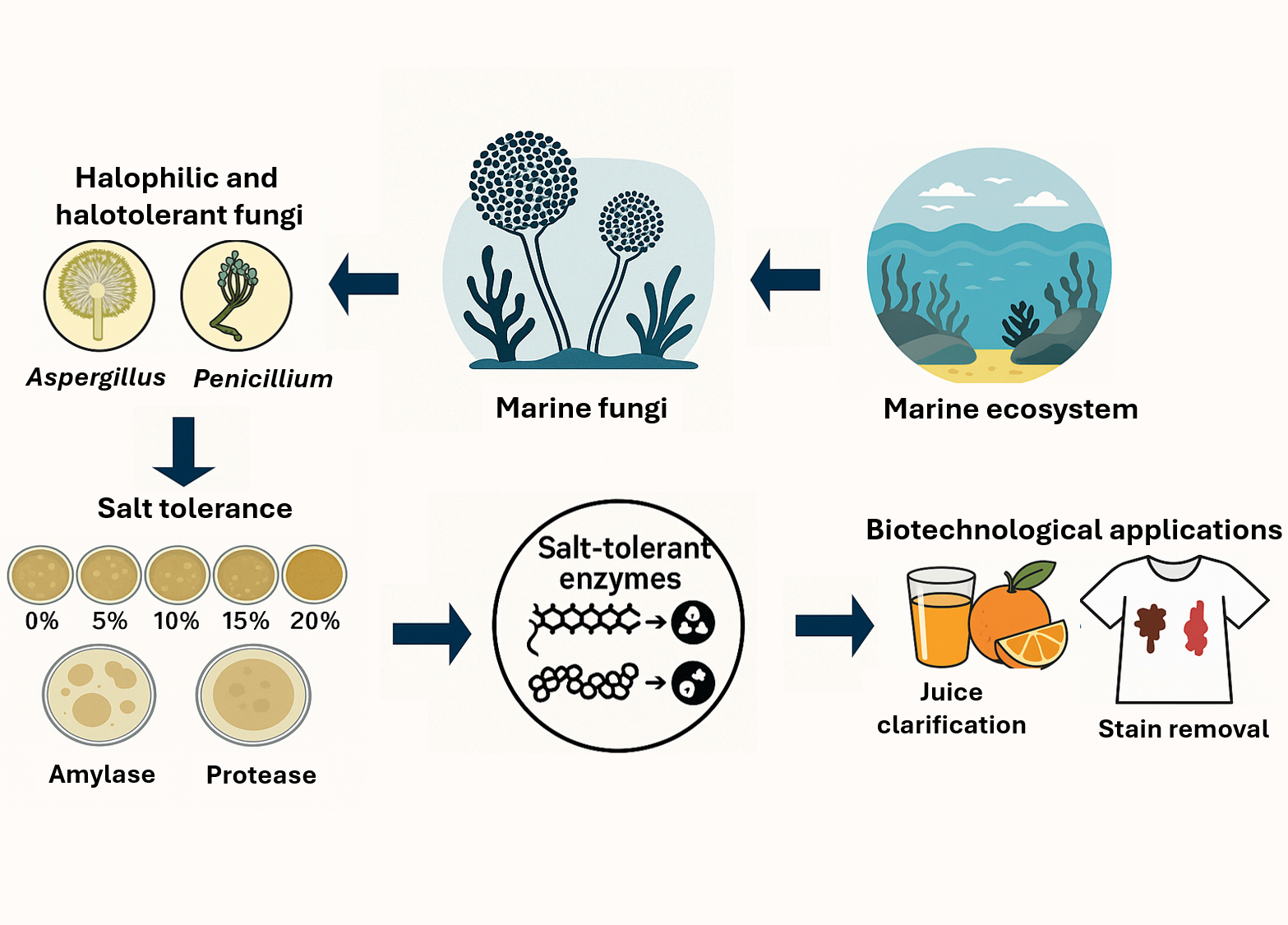
This study aimed to characterize halophilic and halotolerant fungi from marine environments and evaluate their potential as sources of salt-tolerant hydrolases for biotechnological applications in the food and detergent industries. Seven fungal isolates were identified through morphological and molecular characterization of the ITS region and phylogenetic analysis using the Maximum Likelihood method. Salt tolerance was evaluated on PDA supplemented with 0% - 20% NaCl, while amylase and protease activities were assessed by plate assays and quantified spectrophotometrically during 7 - 21 days of cultivation. Crude enzyme extracts were further examined for orange juice clarification and stain-removal efficiency on cotton fabrics, both individually and in combination with sodium dodecyl sulfate (SDS). All isolates belonged to the genera Aspergillus and Penicillium and exhibited facultative halophilism, with optimal growth at 5% - 10% NaCl. A. subalbidus NM-16 and NM-17.1 showed the highest amylase activities (3.16 ± 0.43 and 2.63 ± 0.24 EI), whereas protease production occurred exclusively in P. citrinum isolates. Among them, P. citrinum I22M1 exhibited maximal enzyme activities at day 21 (amylase = 0.93 ± 0.01 U/mL; protease = 1.58 ± 0.01 U/mL). Crude enzyme preparations achieved 32.53 ± 2.01% juice clarification and up to 95.67 ± 8.85% and 88.99 ± 1.21% removal of chocolate and blood stains, respectively. The synergistic combination of enzyme and SDS completely restored fabric whiteness. These findings demonstrate that marine-derived Aspergillus and Penicillium produce stable, halotolerant amylase and protease enzymes capable of functioning under saline conditions, highlighting their potential as eco-friendly biocatalysts for sustainable industrial processes.
HIGHLIGHTS
- Seven halophilic and halotolerant fungi were isolated from marine environments.
- Aspergillus and Penicillium strains exhibited strong salt tolerance up to 20% NaCl.
- Crude enzymes showed significant amylase and protease activities under saline conditions.
- Fungal enzymes effectively clarified orange juice and removed chocolate and blood stains.
- Results highlight marine-derived fungi as promising sources of salt-tolerant hydrolases for eco-friendly industrial use.
GRAPHICAL ABSTRACT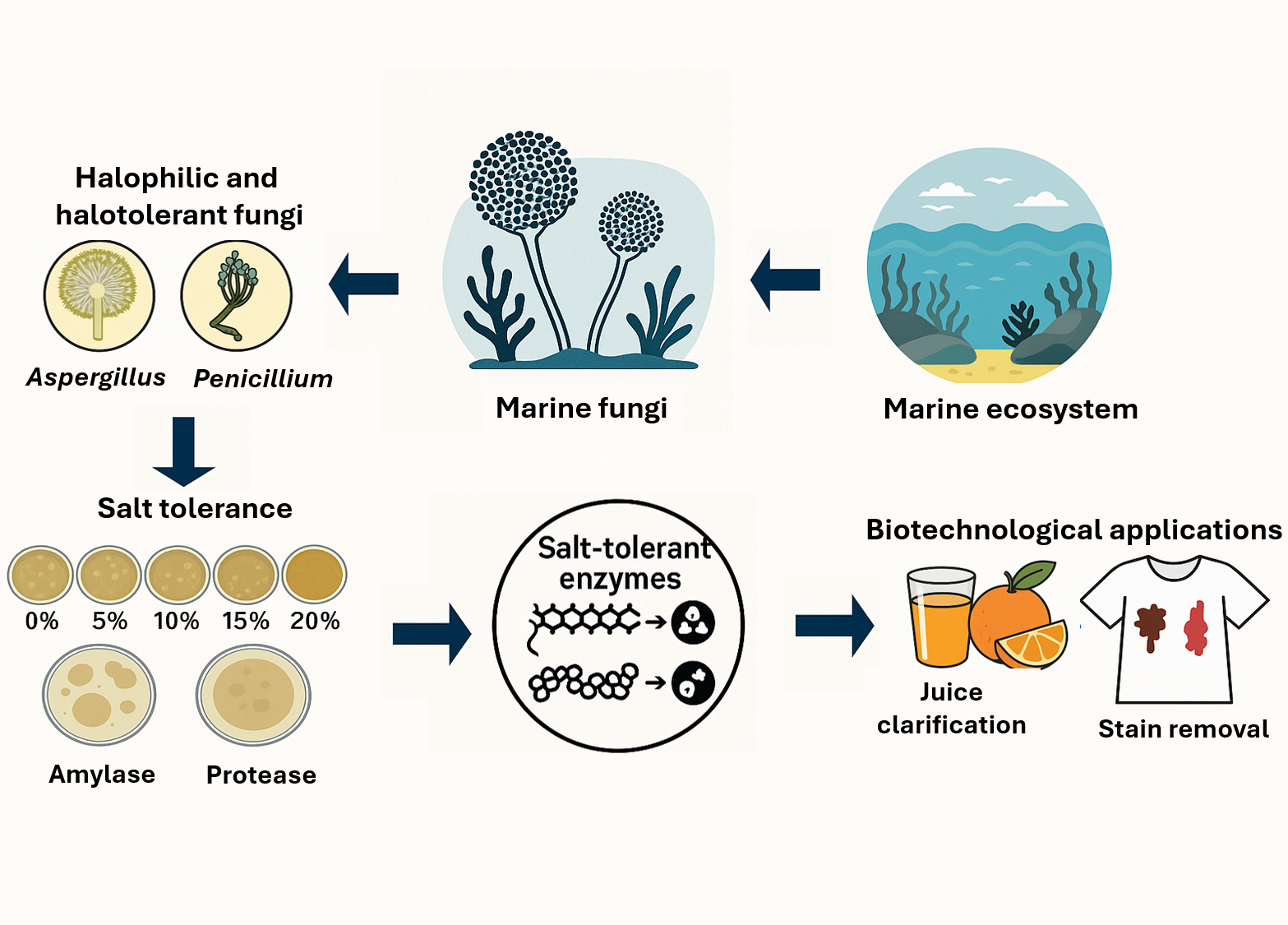
Downloads
References
Y He, B Sen and G Wang. Diversity and dynamics of microbial communities in marine ecosystems. Water 2024; 16(1), 31.
SM Tiquia-Arashiro and M Grube. Fungi in extreme environments ecological role and biotechnological significance. Springer Nature, Cham, Switzerland, 2019.
MFM Gonçalves, AC Esteves and A Alves. Marine fungi: Opportunities and challenges. Encyclopedia 2022; 2, 559-577.
H Yaakoub, NS Sanchez, L Ongay-Larios, V Courdavault, A Calenda, JP Bouchara, R Coria and N Papon. The high osmolarity glycerol (HOG) pathway in fungi. Critical Reviews in Microbiology 2022; 48(6), 657-695.
GEB Jones and KL Pang. Introduction marine fungi. In: GEB Jones and KL Pang (Eds.). Marine fungi. De Gruyter, Berlin, Germany, 2012.
GEB Jones, KL Pang, MA Abdel-Wahab, B Scholz, KD Hyde, T Boekhout, R Ebel, ME Rateb, L Henderson, J Sakayaroj, S Suetrong, MC Dayarathne, V Kumar, S Raghukumar, KR Sridhar, AHA Bahkali, FH Gleason and C Norphanphoun. An online resource for marine fungi. Fungal Diversity 2019; 96, 347-433.
Q Li and G Wang. Diversity of fungal isolates from 3 Hawaiian marine sponges. Microbiological Research 2009; 164, 233-241.
N Gunde-Cimerman, P Zalar, S de Hoog and A Plemenitaš. Hypersaline waters in salterns - natural ecological niches for halophilic black yeasts. FEMS Microbiology Ecology 2000; 32(3), 235-240.
A Plemenitaš, T Vaupotič, M Lenassi, T Kogej and N Gunde-Cimerman. Adaptation of the extremely halotolerant black yeast Hortaea werneckii to salinity: A molecular perspective. Frontiers in Microbiology 2014; 5, 199.
J Zajc, T Kogej, EA Galinski, J Ramos and N Gunde-Cimerman. Osmoadaptation strategy of the most halophilic fungus, Wallemia ichthyophaga, growing optimally at salinities above 15% NaCl. Applied and Environmental Microbiology 2014; 80(1), 247-256.
S Hohmann. Osmotic stress signaling and osmoadaptation in yeasts. Microbiology and Molecular Biology Reviews 2002; 66(2), 300-372.
C Gostinčar, J Zajc, A Plemenitaš and N Gunde-Cimerman. Fungal adaptation to extremely high salt concentrations. Fungal Biology Reviews 2011; 25(4), 133-146.
L Butinar, S Santos, I Spencer-Martins, A Oren and N Gunde-Cimerman. Yeast diversity in hypersaline habitats. FEMS Microbiology Letters 2005; 244(2), 229-234.
S Hasan, MI Ansari, A Ahmad and M Mishra. Major bioactive metabolites from marine fungi: A review. Bioinformation 2015; 11(4), 176-181.
N Kango, UK Jana and R Choukade. Fungal enzymes: Sources and biotechnological applications. In: T Satyanarayana, SK Deshmukh and MV Deshpande (Eds.). Advancing frontiers in mycology & mycotechnology: Basic and applied aspects of fungi. Springer, Singapore, 2019, p. 515-538.
H El-Gendi, AK Saleh, R Badierah, EM Redwan, YA El-Maradny and EM El-Fakharany. A comprehensive insight into fungal enzymes: Structure, classification, and their role in mankind’s challenges. Journal of Fungi 2021; 8(1), 23.
RA Samson, ES Hoekstra and JC Frisvad. Introduction to food and airborne fungi. 6th ed. Centraalbureau voor Schimmelcultures, Utrecht, Netherlands, 2002.
FM Dugan. The Identification of fungi-An illustrated introduction with keys, glossary, and guide to literature. The American Phytopathological Society Press, St. Paul, United States, 2015.
D Ellis, S Davis, H Alexiou, R Handke and R Bartley. Description of medical fungi. 3rd ed. South Australia Nexus Print Solutions, Underdale South Australia, Australia, 2016.
LK Wingfield, J Atcharawiriyakul and N Jitprasitporn. Diversity and characterization of culturable fungi associated with the marine sea cucumber Holothuria scabra. PLoS One 2024; 19(1), e0296499.
K Tamura, G Stecher and S Kumar. MEGA11: Molecular evolutionary genetics analysis version 11. Molecular Biology and Evolution 2021; 38, 3022-3027.
L Hankin and SL Anagnostakis. Solid media containing carboxymethylcellulose to detect C x cellulase activity of microorganisms. Journal of General Microbiology 1975; 98(1), 109-115.
RM Teather and PJ Wood. Use of Congo red-polysaccharide interactions in enumeration and characterization of cellulolytic bacteria from the bovine rumen. Applied and Environmental Microbiology 1982; 43(4), 777-780.
GL Miller. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry 1959; 31(3), 426-428.
DF Coelho, TP Saturnino, FF Fernandes, PG Mazzola, E Silveira and EB Tambourgi. Azocasein substrate for determination of proteolytic activity: Reexamining a traditional method using bromelain samples. BioMed Research International 2016; 2016(1), 8409183.
C Han, L Kautto and H Nevalainen. Secretion of proteases by an opportunistic fungal pathogen Scedosporium aurantiacum. PLOS One 2017; 12(1), e0169403.
MM Bradford. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 1976; 72(1-2), 248-254.
JA Elegbede and A Lateef. Valorization of corn-cob by fungal isolates for production of xylanase in submerged and solid-state fermentation media and potential biotechnological applications. Waste Biomass Valorization 2018; 9(8), 1273-1287.
UC Banerjee, RK Sani, WAzmi and R Soni. Thermostable alkaline protease from Bacillus brevis and its characterization as a laundry detergent additive. Process Biochemistry 1999; 35(1-2), 213-219.
W Śliżewska, K Struszczyk-Świta, A Otlewska, F Pinzari, L Canfora, K Dybka-Stȩpień, R Napoli, M Migliore, A Manfredini and O Marchut-Mikołajczyk. Halophilic and halotolerant fungi across diverse climates: A comparative study of Polish and Italian soil ecosystems. Frontiers in Microbiology 2025; 16, 1637496.
L Yovchevska, Y Gocheva, G Stoyancheva, J Miteva-Staleva, V Dishliyska, R Abrashev, T Stamenova, M Angelova and E Krumova. Halophilic fungi-Features and potential applications. Microorganisms 2025; 13, 175.
LK Wingfield, N Jitprasitporn and N Che-alee. Isolation and characterization of halophilic and halotolerant fungi from man-made solar salterns in Pattani Province, Thailand. PLoS One 2023; 18(2), e0281623.
I Pócsi, J Dijksterhuis, J Houbraken and RP de Vries. Biotechnological potential of salt tolerant and xerophilic species of Aspergillus. Applied Microbiology and Biotechnology 2024; 108(1), 521.
IB Hmad and A Gargouri. Halophilic filamentous fungi and their enzymes: Potential biotechnological applications. Journal of Biotechnology 2024; 381, 11-18.
G Ozyilmaz and E Gunay. Clarification of apple, grape and pear juices by co-immobilized amylase, pectinase and cellulase. Food Chemistry 2023; 398, 133900.
T Roheen, R Ramzan, W Khalid M Nadeem, FA Atif, M Munir and TM Qureshi. Synthesis and characterization of CMC/PAM-amy hydrogel and its efficacy in apple juice clarification. Processes 2024; 12, 2264.
K Farooq, Z Anwar, S Hasan, F Afzal, M Zafar, U Ali, O Alghamdi, A AL-Farga and SM Al-maaqar. Optimization and detergent compatibility of protease produced from Aspergillus oryzae by utilizing agro wastes. ACS Omega 2024; 9(15), 17446-17457.
X Wang, X Qin, L Tong, J Zheng, T Dong, X Wang, Y Wang, H Huang, B Yao, H Zhang and H Luo. Improving the catalytic activity of a detergent-compatible serine protease by rational design. Microbial Biotechnology 2023; 16(5), 947-960.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






