Dual Inhibition and Safety Profiling of Padina Australis Extract as A Marine-Derived α-Glucosidase Modulator: An Integrated In Vitro and In Silico Study
DOI:
https://doi.org/10.48048/tis.2026.12559Keywords:
Padina australis, α-glucosidase, In vitro, In silico, Molecular docking, Fatty acids, AntidiabeticAbstract
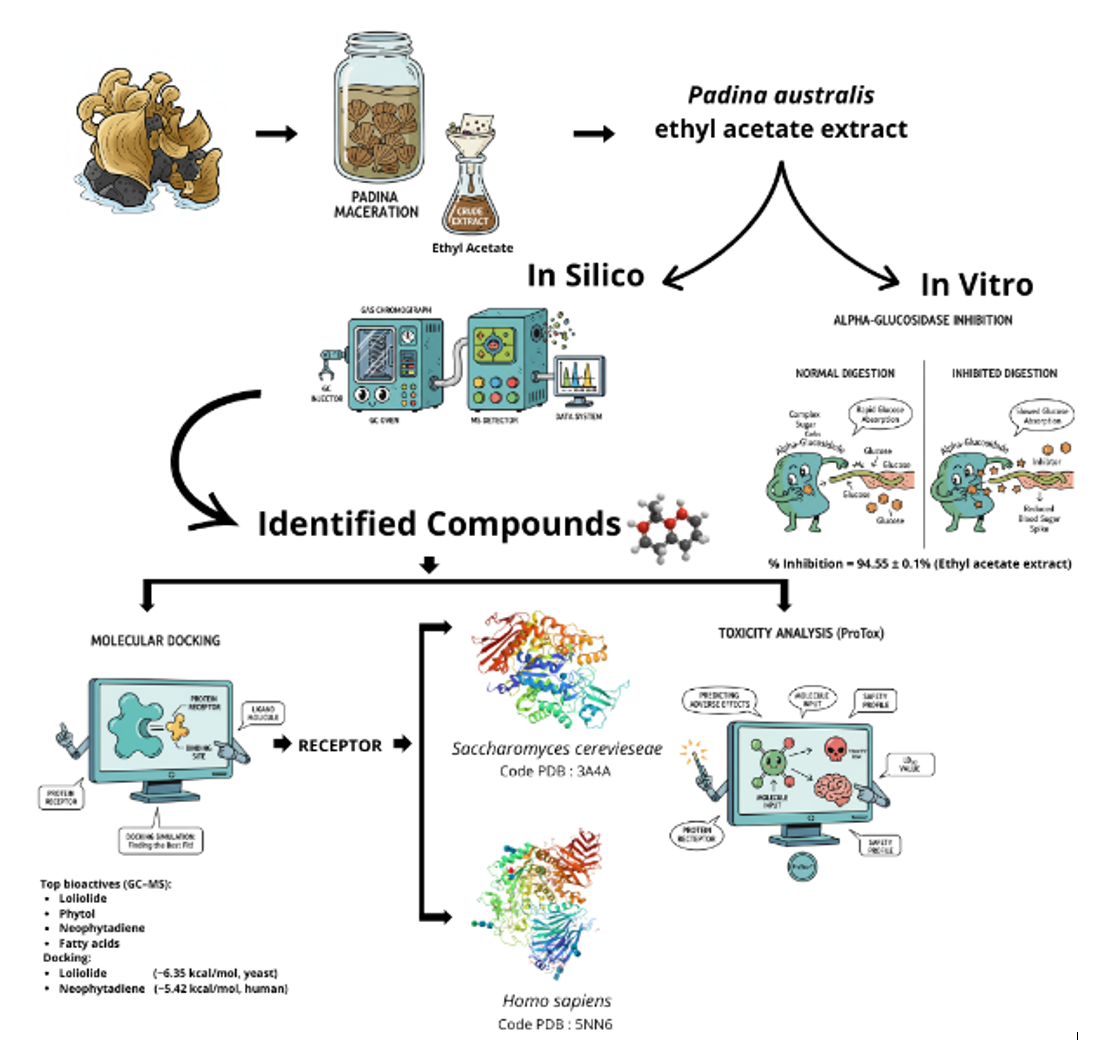
Padina australis is a brown seaweed that shows promise as a natural source of α-glucosidase inhibitors for diabetes management. This study evaluated the α-glucosidase inhibitory activity of P. australis extracts and examined how extraction solvents influence this activity. It also identified potential active metabolites using gas chromatography–mass spectrometry (GC–MS), molecular docking, and toxicity prediction, thereby providing new evidence on the α-glucosidase inhibitory potential of P. australis. Ultrasonic-assisted extraction with n-hexane, ethyl acetate, and ethanol revealed that the ethyl acetate extract exhibited the highest inhibitory activity at 500 µg∙mL−1 (94.55 ± 0.16%), comparable to the ethanol extract (93.75 ± 2.56%) and higher than the n-hexane extract (45.37 ± 4.59%). GC–MS analysis identified 23 compounds, including loliolide, 2(4H)-benzofuranone, 5,6,7,7a-tetrahydro-4,4,7a-trimethyl, phytol, neophytadiene, and several fatty acids. Docking against yeast α-glucosidase (3A4A), human maltase–glucoamylase (3L4T), and lysosomal α-glucosidase (5NN6) indicated that loliolide and benzofuranone derivatives showed affinities approaching acarbose on the yeast enzyme, while neophytadiene and polyunsaturated fatty acids exhibited the most favorable interactions with the human intestinal enzyme. Binding to lysosomal α-glucosidase (5NN6) was consistently weaker, suggesting a degree of selectivity away from lysosomal targets. ProTox-3.0 predicted low acute toxicity for most metabolites (Classes V-VI) and moderate toxicity for two compounds (Class IV), whereas loliolide was classified as higher-risk (Class II). These findings support Padina australis as a potential source of α-glucosidase-modulating metabolites at a screening level. However, comprehensive studies including dose-response assays, enzyme kinetic characterization, fractionation, and toxicity testing are required to validate and extend these preliminary observations.
HIGHLIGHTS
- Padina australis ethyl acetate extract showed in vitro α-glucosidase inhibition of > 90% at 500 µg∙mL−1.
- Molecular docking indicated strong binding of key metabolites to yeast (3A4A) and human intestinal (3L4T) α-glucosidase, with consistently weaker binding to the lysosomal isoform (5NN6), suggesting selectivity.
- GC–MS identified 23 compounds; key metabolites formed favorable interactions with catalytic site residues in docking simulations.
- In silico toxicity prediction indicated that most compounds were in low-toxicity classes (loliolide was a notable exception, flagged as higher toxicity).
- These findings support australis as a promising marine source of α-glucosidase-modulating metabolites, though further dose–response and safety studies are warranted.
GRAPHICAL ABSTRACT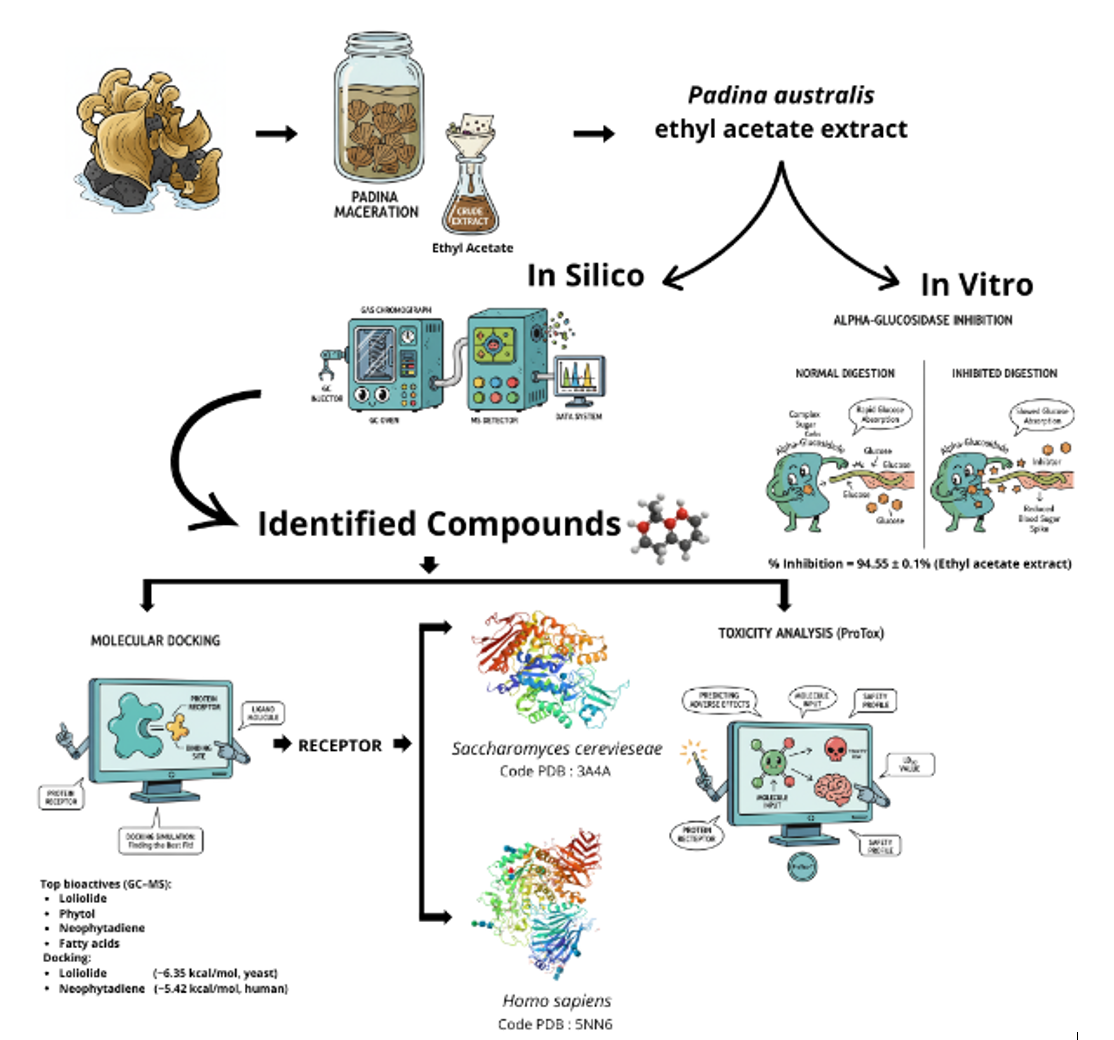
Downloads
References
L Zhang, W Liao, Y Huang, Y Wen, Y Chu and C Zhao. Global seaweed farming and processing in the past 20 years. Processing and Nutrition 2022; 4, 23.
MA Rimmer, S Larson, I Lapong, AH Purnomo, PR Pong-masak, L Swanepoel and NA Paul. Seaweed aquaculture in Indonesia contributes to social and economic aspects of livelihoods and community wellbeing. Sustainability 2021; 13(19), 10946.
M Basyuni, M Puspita, R Rahmania, H Albasri, SS Al, F Menne, Y Ihrami, SG Salmo, A Susilowati, SH Larekeng and E Ardli. Heliyon current biodiversity status , distribution , and prospects of seaweed in Indonesia: A systematic review. Heliyon 2024; 10(10), e31073.
S Lomartire and AMM Gonçalves. An overview of potential seaweed-derived bioactive compounds for pharmaceutical applications. Marine Drugs 2022; 20(2), 141.
Y Bouafir, M Mounir, A Nebbak, L Belfarhi, B Aouzal, F Boufahja, H Bendif and M Bruno. Fitoterapia algal bioactive compounds : A review on their characteristics and medicinal properties. Fitoterapia 2025; 183, 106591.
THP Brotosudarmo, D Pringgenies and AA Wibawa. Current progress in exploring structural changes in brown algae fucoxanthin and its potential bioactivity for human health. Trends in Sciences 2024; 21(9), 7984.
N Moheimanian, H Mirkhani, A Purkhosrow, J Sohrabipour and A Reza. In Vitro and In Vivo antidiabetic , α -glucosidase inhibition and antibacterial activities of three brown algae , polycladia myrica , padina antillarum , and sargassum boveanum , and a red alga , palisada perforata from the persian gulf. The IJ Pharmaceutical Research 2023; 22(1), e133731.
A Maheswari and D Sdep. In vitro correlation studies of antidiabetic, antioxidant activity and HPLC-ESI-MS/MS analysis of marine seaweeds from Gulf of Mannar. Regional Studies in Marine Science 2022; 56, 102682.
YL Cheryl-Low, KL Theam and HV Lee. Alginate-derived solid acid catalyst for esterification of low-cost palm fatty acid distillate. Energy Conversion and Management 2015; 106, 932-940.
J Naveen, R Baskaran and V Baskaran. Profiling of bioactives and in vitro evaluation of antioxidant and antidiabetic property of polyphenols of marine algae Padina tetrastromatica. Algal Research 2021; 55, 102250.
G Gomori. Preparation of buffers for use in enzyme studies. Methods in Enzymology 1955; 1, 138-146.
H Widwiastuti, RY Asworo, YS Tjahjaningsih, NC Wulandari and A Dewi. Pengaruh ukuran simplisia dan lama kontak pada ekstraksi senyawa aktif simplisia kayu jawa (Lannea Coromandelica) menggunakan metode maserasi. Jurnal Kimia Mulawarman 2022; 19(2), 86-90.
L Efriani, I Hadi, A Irawan, M Ulfah and TA Putra. uji aktivitas inhibisi enzim a-glukosidase terhadap ekstrak aseton, etanol, dan methanol daun mangrove (rhizopora mucronata) sebagai antidiabetes. Medical Sains Jurnal Ilmiah Kefarmasian 2023; 8(4), 1371-1377.
NNW Win, T Hanyuda, S Arai, M Uchimura, A Prathep, SGA Draisma, SM Phang, IA Abbott, AJK Millar and H Kawai. A taxonomic study of the genus padina ( dictyotales , phaeophyceae ) including the descriptions of four new species from japan , hawaii , and the andaman sea. Journal of Phycology 2011; 47, 1193-1209.
CWF Fu, CW Ho, WTL Yong, F Abas, TB Tan and CP Tan. Extraction of phenolic antioxidants from four selected seaweeds obtained from Sabah. International Food Research Journal 2016; 23(6), 2363-2369.
C Reichardt and T Welton. Solvents and solvent effects in organic chemistry. 4th ed. Wiley, New Jersey, 2010.
H Nawaz, MA Shad, N Rehman, H Andaleeb and N Ullah. Effect of solvent polarity on extraction yield and antioxidant properties of phytochemicals from bean (Phaseolus vulgaris) seeds. Brazilian Journal of Pharmaceutical Sciences 2015; 56, e17129.
A Permatasari, I Batubara, M Nursid and K Kelautan. Pengaruh konsentrasi etanol dan waktu maserasi terhadap rendemen , kadar total fenol dan aktivitas antioksidan ekstrak rumput laut padina australis. Majalah Ilmiah Biologi Biosfera: A Scientific Journal 2020; 37(2), 78-84.
M Ponnanikajamideed, M Malini, C Malarkodi and S Rajeshkumar. Bioactivity and phytochemical constituents of marine brown seaweed (padina tetrastromatica) extract from various organic solvents. International Journal of Pharmacy & Therapeutics 2014; 5(2), 108-112.
M Gazali, O Jolanda, A Husni, F Adibah and A Majid. In vitro α-Amylase and α-Glucosidase Inhibitory Activity of Green Seaweed Halimeda tuna Extract from the Coast of Lhok Bubon, Aceh. Plants 2023; 12(2), 393.
SN Hikmah, R Setianto, BA Dewi and R Utami. Antibacterial activities of N-hexan ethyl acetate fraction and water from ethanol extract of yodium leaves (Jatrophora multifida L.) Against pseudomonas aeruginosa ATCC 27853. STRADA Jurnal Ilmiah Kesehatan 2021; 10(1), 1242-1249.
S Chen, B Lin, J Gu, T Yong, X Gao, Y Xie, C Xiao, JY Zhan and Q Wu. Binding interaction of betulinic acid to α -glucosidase and its alleviation on postprandial hyperglycemia. Molecules 2022; 27(8), 1-12.
Y Isono, H Watanabe, M Kumada and T Takara. Black tea decreases postprandial blood glucose levels in healthy humans and contains high-molecular-weight polyphenols that i nhibit α -g lucosidase and α -amylase in vitro: A randomized, double blind, placebo-controlled, crossover trial. Functional Foods in Health and Disease 2021; 11(5), 222-237.
ND Tuan, NC Quoc, DN Ly, BT Tuyen, LD Quang, HH Phien, TQ De and TT Men. bioactive extracts from padina boryana thivy from Phu Quoc Island, Vietnam: In vitro antioxidant, anticancer, alpha-glucosidase inhibitory, anti-inflammatory, antimicrobial, and hepatoprotective activities. Tropical Journal of Natural Product Research 2024; 8(12), 9555-9559.
NSA Elalal, GM El Seedy and YA Elhassaneen. Chemical composition, nutritional value, bioactive compounds content and biological activities of the brown alga (Sargassum Subrepandum) collected from the mediterranean Sea, Egypt. Alexandria Science Exchange Journal 2021; 42(4), 893-906.
S Palaniyappan, A Sridhar, ZA Kari, T Guillermo and T Ramasamy. Evaluation of phytochemical screening, pigment content, in vitro antioxidant, antibacterial potential and GC-MS metabolite profiling of green seaweed caulerpa racemosa. Marine Drugs 2023; 21(5), 278.
MKA Sobuj, A Islam, S Islam, M Islam, Y Mahmud and SUM Rafiquzzaman. Effect of solvents on bioactive compounds and antioxidant activity of Padina tetrastromatica and Gracilaria tenuistipitata seaweeds collected from Bangladesh. Scientific Reports 2021; 11(1), 19082.
N Astalakshmi, T Gokul, KBG Sankar, M Nandhini, HH Sudhan, S Gowtham, ST Latha and MS Kumar. Over view on molecular docking: A powerful approach for structure based drug discovery. International Journal of Pharmaceutical Sciences Review and Research 2022; 77(2), 180-198.
A Abudurexiti, R Zhang, Y Zhong, H Tan, J Yan and S Bake. Identification of α -glucosidase inhibitors from Mulberry using UF-UPLC-QTOF-MS / MS and molecular docking. Journal of Functional Foods 2023; 101, 105362
A Khaldan, S Bouamrane, R El-mernissi, H Maghat, M Aziz, A Sbai, M Bouachrine and T Lakhlifi. 3D-QSAR modeling , molecular docking and ADMET properties of benzothiazole derivatives as a-glucosidase inhibitors. Materials Today: Proceedings 2021; 45, 7643-7652.
HK Rundla, S Soni, S Teli, A Manhas, PC Jha, S Agarwal and LK Agarwal. Theophylline hydrogen sulfate as a highly efficient catalyst for the synthesis of quinoxaline derivatives: Exploring potential antidiabetic agents through molecular docking. Catalysis Letters 2025; 155(243), 243.
P Banerjee, AO Eckert, AK Schrey and R Preissner. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Research 2018; 46, W257-W263.
M Lacueva-Arnedo, A Gómez-Barrio, A Ibáñez-Escribano, TJ López-Pérez, B Casarrubias-Tabarez, F Calzada, PY López-Camacho and N Rivera-Fernández. Antibacterial, trichomonacidal, and cytotoxic activities of pleopeltis crassinervata extracts. Pharmaceutics 2024; 16(5), 624.
I Oktavianawati, U Wulandari, INA Winata, AA Ridho, YI Kedang, DT Fauziah and F Yusro. The volatile compound profiles of Fire-cured and fermented Na-oogst tobacco leaves (Nicotiana tabacum L.) and its in-silico study. Indonesian Chimica Letters 2025; 4(1), 7-15.
OM Oluwakeyede and BA Odeyemi. GC-MS profiling of bioactive compounds in ethanol extract of Annona Squamosa L. Leaves. Journal of Chemical Society of Nigeria 2025; 50(4), 747-755.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






