α-Glucosidase Inhibitory Constituents from the Fruits of Thai Helicteres isora L.
DOI:
https://doi.org/10.48048/tis.2026.12491Keywords:
Helicteres isora L., Phytochemicals, α-Glucosidase inhibition, Xanthine oxidase inhibition, Molecular docking, ADMETAbstract
Helicteres isora L. is a traditionally recognized medicinal plant in Asia, used for the treatment of gastrointestinal, metabolic, respiratory, and infectious diseases, with different parts exhibiting diverse pharmacological activities. In the present investigation, the fruits of H. isora L. were successfully investigated, leading to the isolation of β-sitosterol (1), betulinic acid (2), ursolic acid (3), 3b-hydroxyurs-11-ene-28(13)-lactone (4), ergosterol-5,8-peroxide (5), syringaresinol (6), β-sitosterol-3-O-β-D-glucoside (7), curcumin (8), demethoxycurcumin (9), and bisdemethoxycurcumin (10). The inhibitory effects of all isolated compounds on α-glucosidase and xanthine oxidase (XO) were assessed. Among them, compound 9 exhibited the most potent α-glucosidase inhibition (IC50 75.5 ± 0.06 µM), whereas compound 8 showed weak XO inhibition (IC50 95.0 ± 0.16 µM). In the molecular docking results, compounds 8 - 10 demonstrated strong α-glucosidase inhibition with conserved interactions at key residues (Asp203, Trp299, Trp406, Met444, Phe575, and Arg526), and their predicted physicochemical and pharmacokinetic properties support their potential as orally active, locally acting antidiabetic agents.
HIGHLIGHTS
- Ten compounds (1 - 10) were isolated from the fruits of Thai Helicteres isora, including 5 triterpenoids (1 - 5), 1 lignan (6), 1 triterpenoid glycoside (7), and 3 curcuminoids (8 - 10).
- This study reports the isolation of compounds 2 - 10 from isora L. for the first time.
- Compounds 8 - 10 exhibited potent α-glucosidase inhibitory activity, with compound 9 being the most active (IC50 5 ± 0.06 µM), surpassing acarbose (IC50 198.9 ± 0.02 µM).
- Molecular docking revealed that compounds 8 - 10 shared conserved binding interactions with key α-glucosidase residues (Asp203, Trp299, Trp406, Met444, Phe575, and Arg526).
- In silico ADMET predictions suggested favorable solubility and intestinal permeability profiles, supporting their potential as orally active, locally acting antidiabetic agents.
GRAPHICAL ABSTRACT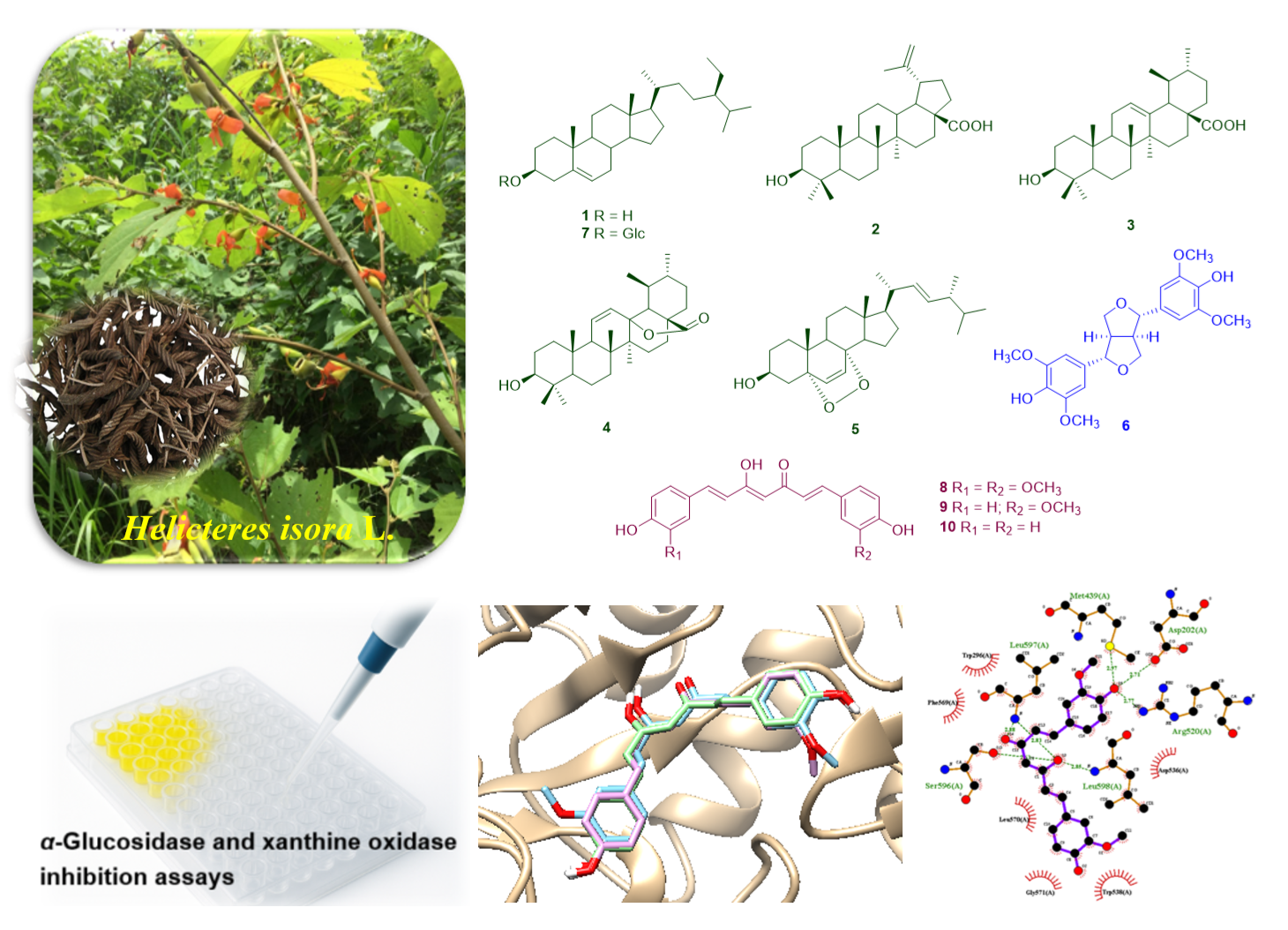
Downloads
References
TA Siswoyo, A Supriyadi, A Isnainun, E Novianti and R Harmoko. Impact of maturity stage on free radical scavenging and antidiabetic activities of Melinjo (Gnetum gnemon L.) seed proteins. Tropical Journal of Natural Product Research 2024; 8(8), 8001-8006.
SMA Shah, F Akram, H Naeem, R Fatima, M Murtza, MS Aslam, AU Rehman and M Aamir. Investigation and evaluation of organic chemistry of alpha-glucosidase inhibitors for managing blood sugar levels. Indus Journal of Bioscience Research 2025; 3(6), 345-353.
SK Liu, H Hao, Y Bian, YX Ge, S Lu, HX Xie, KM Wang, H Tao, C Yuan, J Zhang, J Zhang, CS Jiang and K Zhu. Discovery of new α-glucosidase inhibitors: Structure-based virtual screening and biological evaluation. Frontiers in Chemistry 2021; 9, 639279.
N Kumar and AK Singh. Plant profile, phytochemistry and pharmacology of Avartani (Helicteres isora Linn.): A review. Asian Pacific Journal of Tropical Biomedicine 2014; 4(S1), S22-S26.
KS Saravanan, R Gowri, KK Ramesh, S Shetty and S Maity. Potential use of Helicteres isora L. in diabetes mellitus: A systematic review of scientific literature. Pharmacognosy Communications 2023; 13(4), 163-168.
K Beauty, S Talib and E Mohd. Phytochemistry and pharmacology of Helicteres isora Linn. (Marodphali): Ayurvedic insights and medicinal overview. International Journal of Ayurveda and Pharma Research 2024; 12(12), 118-128.
S Pandey, D Patel, P Mishra and R Tiwari. Morphological, phytochemical and pharmacological study of Helicteres isora (Marorphali). International Journal of Research in Pharmacy and Pharmaceutical Sciences 2021; 6(3), 13-17.
SP Mahire and SN Patel. Extraction of phytochemicals and study of its antimicrobial and antioxidant activity of Helicteres isora L. Clinical Phytoscience 2020; 6(1), 40.
NH Nguyen, BP Vuong, CH Nguyen, AD Huynh, DM Nguyen, TH Duong and J Sichaem. Antimicrobial and α-glucosidase inhibitory compounds from the branches of Uvaria siamensis. Records of Natural Products 2024; 18(3), 331-338.
TT Nguyen, TQ Tran, L Do, NM Huynh, K Inthanon and J Sichaem. α-Glucosidase and xanthine oxidase inhibitory activities from the fruits of Thai Averrhoa bilimbi L. Tropical Journal of Natural Product Research 2025; 9(3), 1105-1111.
HM Berman, J Westbrook, Z Feng, G Gilliland, TN Bhat, H Weissig, IN Shindyalov and PE Bourne. The protein data bank. Nucleic Acids Research 2000; 28(1), 235-242.
S Kim, J Chen, T Cheng, A Gindulyte, J He, Q Li, BA Shoemaker, PA Thiessen, B Yu, L Zaslavsky, J Zhang and EE Bolton. PubChem in 2021: New data content and improved web interfaces. Nucleic Acids Research 2021; 49, D1388-D1395.
GM Morris, R Huey, W Lindstrom, MF Sanner, RK Belew, DS Goodsell and AJ Olson. AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. Journal of Computational Chemistry 2009; 30(16), 2785-2791.
EF Pettersen, TD Goddard, CC Huang, GS Couch, DM Greenblatt, EC Meng and TE Ferrin. UCSF Chimera - a visualization system for exploratory research and analysis. Journal of Computational Chemistry 2004; 25(13), 1605-1612.
RA Laskowski and MB Swindells. LigPlot+: Multiple ligand-protein interaction diagrams for drug discovery. Journal of Chemical Information and Modeling 2011; 51(10), 2778-2786.
L Fu, S Shi, J Yi, N Wang, Y He, Z Wu, L Zhang, X Liu, M Li and H Sun. ADMETlab 3.0: An updated comprehensive online ADMET prediction platform enhanced with broader coverage, improved performance, API functionality and decision support. Nucleic Acids Research 2024; 52, W422-W431.
RA El-Shiekh, DA Al-Mahdy, MS Hifnawy, T Tzanova, E Evain-Bana, S Philippot and EA Abdelsattar. Chemical and biological investigation of Ochrosia elliptica Labill. cultivated in Egypt. Records of Natural Products 2017; 11(6), 552-557.
Noviany and H Osman. Structure elucidation of betulinic acid from Sesbania grandiflora root. Journal of Physics: Conference Series 2021; 1751, 012090.
BC Huu and NKP Phung. Contribution to the study on chemical constituents of Hedyotis crassifolia L. (Rubiaceae). Vietnam Journal of Chemistry 2007; 45(3), 363-363.
NTM Dung, PNKT Tuyen, J Mortier and NKPP Phung. Some triterpenoids and steroids from the lichen Lobaria orientalis, Lobariaceae. Journal of Science and Technology 2016; 54, 313-319.
W Monthong, S Pitchuanchom, N Nuntasaen and W Pompimon. (+)-Syringaresinol lignan from new species Magnolia thailandica. American Journal of Applied Sciences 2011; 8(12), 1268-1271.
NTT Nhung and VTB Huệ. Phân lập một số hợp chất tự nhiên từ thân hành Trinh nữ Hoàng cung (Crinum latifolium L., Amaryllidaceae) (in Vietnamese). Tạp chí Khoa học Lạc Hồng 2020; 9, 10-13.
L Péret-Almeida, APF Cherubino, RJ Alves, L Dufossé and MBA Glória. Separation and determination of the physico-chemical characteristics of curcumin, demethoxycurcumin and bisdemethoxycurcumin. Food Research International 2005; 38(8-9), 1039-1044.
FAV de Laar. Alpha-glucosidase inhibitors in the early treatment of type 2 diabetes. Vascular Health and Risk Management 2008; 4(6), 1189-1195.
JB Dressman and J Krämer. Pharmaceutical dissolution testing. Taylor & Francis, New York, 2005.
CA Lipinski, F Lombardo, BW Dominy and PJ Feeney. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development setting. Advanced Drug Delivery Reviews 2001; 46, 3-26.
H Lu and Q Chen. A review on preparation of betulinic acid and its biological activities. Molecules 2021; 26, 5583.
R Arulnangai, HA Thabassoom, HV Banu, K Thirugnanasambandham and R Ganesamoorthy. Recent developments on ursolic acid and its potential biological applications. Toxicology Reports 2025; 14, 101900.
Y Mulyani, A Arofatus Naini, K Farabi, FF Abdullah, W Safriansyah, SE Sinaga, A Azhari, S Purbaya, A Peni Wulandari, S Fajriah, Y Shiono and U Supratman. The steroids produced by Cladosporium anthropophilum, an endophytic fungus isolated from Avicennia marina (Forssk.) Vierh and their antibacterial activity. Trends in Sciences 2024; 21(12), 8498.
I Mssillou, M Bakour, FEZ Amrati, B Oraibi, ME Elmobark and A Khalid. Syringaresinol: Exploring its natural sources, bioactivity, and health benefits. Chemistry & Biodiversity 2025; 22, e00352.
NL Harfita, A Santoni and S Suryati. Beta-sitosterol glycoside from Paraboea leuserensis and cytotoxicity test against MCF-7 human breast cancer cells. Riset Informasi Kesehatan 2023; 12(2), 272-276.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






