The Influence of the Simultaneous Addition of Fe2+ and Fe3+ on Changes in the Structural, Optical, and Electronic Properties of TiO2 Powder
DOI:
https://doi.org/10.48048/tis.2026.12406Keywords:
TiO₂ powder, Fe2 : Fe3 ratio, TiO₂: Fe₂O₃ ratio, Simultaneous, Structural, Optical, Electronic, PropertiesAbstract
Modification of TiO₂ powder to form TiO₂-Fe/Fe₂O₃ composites with simultaneous incorporation of Fe²⁺ and Fe³⁺ ions via a simple approach has been conducted. The characterization of photocatalyst composites has been carried out using FTIR, Raman, XRD, SAA, TEM, DRS, XPS, and VSM. The simultaneous incorporation of Fe²⁺ and Fe³⁺ ions has modified the structural, optical, and magnetic properties of TiO2 powder. Systematic variation of Fe²⁺: Fe³⁺ compositions (mole/mole) revealed that Fe³⁺ had a stronger impact than Fe²⁺. At low Fe³⁺ and Fe₂O₃ composition, interfacial interaction and heterojunction formation dominated, whereas higher levels promoted substitutional doping. At low Fe³⁺ composition (Fe2+: Fe3+ = 1:0.3 - 1:1), crystallite size increased (29.95 - 34.65 nm) but crystallinity decreased (62.34% - 57.99%), surface area increased (25.90 - 29.71 m2/g) but pore size decreased (0.097 - 0.085 cc/g), the bandgap narrowed (3.14 - 2.98 eV), and maghemite formation and magnetic moment decreased (36.83 - 17.66 emu/g). Whereas at Fe³⁺ higher ratio (Fe2+: Fe3+ = 1:1 - 1:3), the bandgap significantly narrowed (2.98 - 2.80 eV), crystallite size (34.65 - 27.30 nm) and crystallinity (57.99% - 49.27%) decreased, surface area (29.09 - 43.49 m2/g) and pore size (0.085 - 0.139 cc/g) increased, and hematite formation increased and magnetic moment decreased (17.66 - 14.86 emu/g). Furthermore, increasing the proportion of Fe₂O₃ in TiO₂ powder (TiO₂: Fe₂O₃) has enlarged the surface area and pore size, narrowed the bandgap, decreased the crystallite sizes and crystallinity, and improved magnetic properties. Photocatalytic evaluation under visible light showed that increasing Fe³⁺ and Fe₂O₃ enhanced both Methyl Violet (MV) degradation up to 84.54% and Cr(VI) reduction up to 36.55%, with stronger MV degradation indicating that Fe³⁺ mainly serves as an electron trap.
HIGHLIGHTS
- Simultaneous addition of Fe2+ and Fe3+ ions in basic conditions to TiO2 powder has resulted in interesting changed properties, in which the Fe³⁺ effect is stronger than Fe²⁺.
- At low Fe³⁺ composition (Fe2+:Fe3+ = 1:0.3 - 1:1), the interfacial interaction and heterojunction formation dominated, bandgap narrowed, crystallite size increased but crystallinity decreased, surface area increased but pore size decreased, and maghemite formation and magnetic moment decreased.
- Whereas at Fe³⁺ higher ratio (Fe2+:Fe3+ = 1:1 - 1:3), the substitutional doping increased, bandgap significantly narrowed, crystallite size and crystallinity decreased, surface area and pore size increased, hematite formation increased and magnetic moment decreased.
- Increasing the proportion of Fe₂O₃ in TiO₂ powder (TiO₂:Fe₂O₃) has enlarged the surface area, narrowed the bandgap, decreased the crystallite sizes and crystallinity, and improved magnetic properties.
- Increasing Fe³⁺ and Fe₂O₃ enhanced both Methyl Violet (MV) degradation and Cr(VI) reduction under visible light.
GRAPHICAL ABSTRACT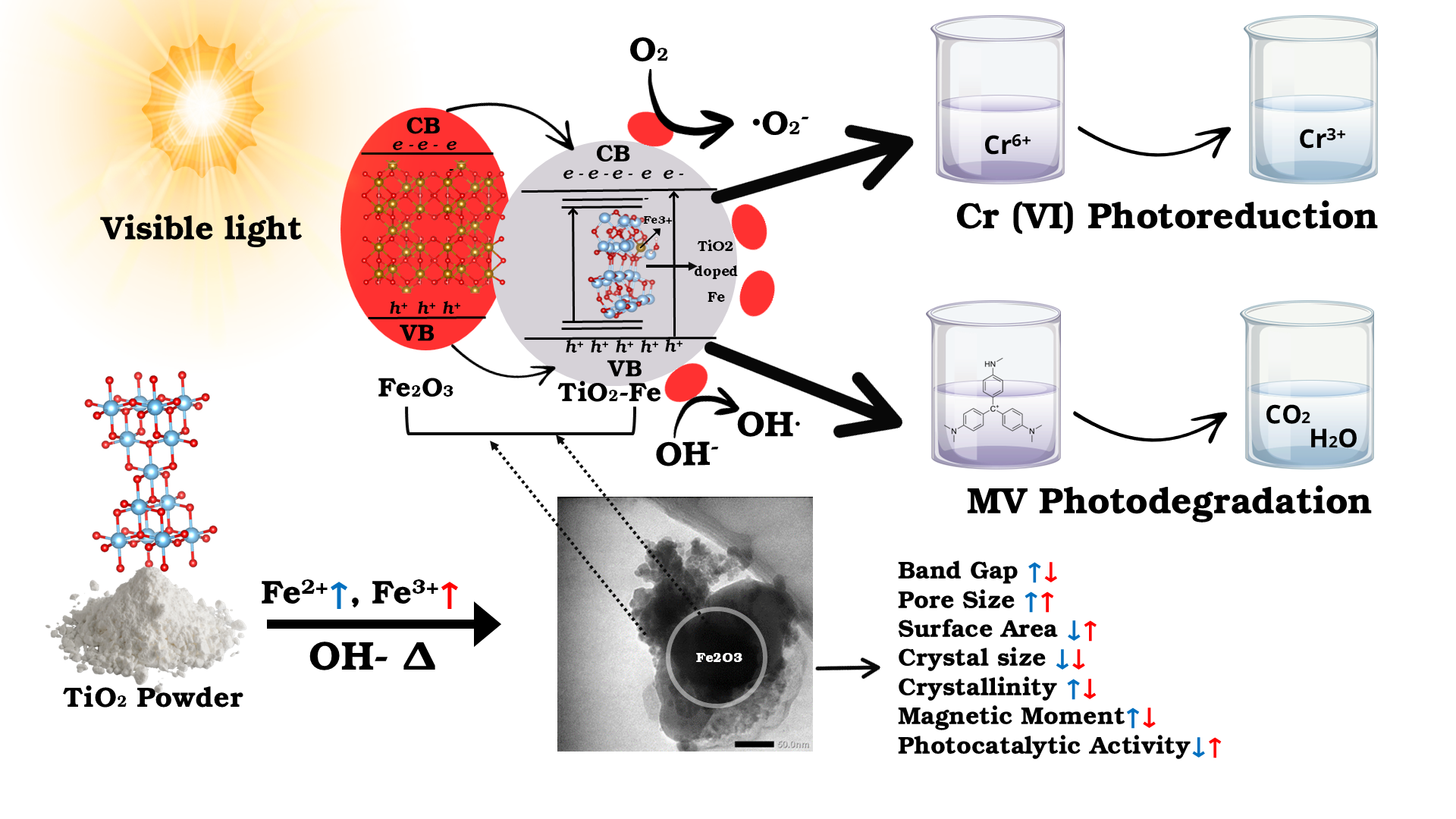
Downloads
References
A Kubiak, M Kubacka, E Gabała, A Dobrowolska, K Synoradzki, K Siwińska-Ciesielczyk, K Czaczyk and T Jesionowski. Hydrothermally assisted fabrication of TiO2-Fe3O4 composite materials and their antibacterial activity. Materials 2020; 13(21), 4715.
VV Shymanovska, TA Khalyavka, EV Manuilov, TA Gavrilko, A Aho, VV Naumov and ND Shcherban. Effect of surface doping of TiO2 powders with Fe ions on the structural, optical and photocatalytic properties of anatase and rutile. Journal of Physics and Chemistry of Solids 2022; 160, 110308.
N Madima, KK Kefeni, SB Mishra, AK Mishra and AT Kuvarega. Fabrication of magnetic recoverable Fe3O4/TiO2 heterostructure for photocatalytic degradation of Rhodamine B dye. Inorganic Chemistry Communications 2022; 145, 109966.
E Savitri, SW Yunita, C Winoto and RK Widi. Innovative waste benzene remediation by enhanced photocatalytic with TiO2/Fe3O4-Based material supported by bentonite in batch and continuous system. Trends in Sciences 2024; 21(7), 7872.
C Afonso, O Lima, IR Segundo, S Landi, E Margalho, N Homem, M Pereira, MFM Costa, E Freitas and J Carneiro. Effect of Iron-doping on the structure and photocatalytic activity of TiO2 nanoparticles. Catalysts 2023; 13(1), 58.
P Sun, S Han, J Liu, J Zhang, S Yang, F Wang, W Liu, S Yin, Z Ning and W Cao. Introducing oxygen vacancies in TiO2 lattice through trivalent iron to enhance the photocatalytic removal of indoor NO. International Journal of Minerals, Metallurgy and Materials 2023; 30, 2025-2035.
B Taudul, F Tielens and M Calatayud. On the origin of raman activity in anatase TiO2 (Nano) materials: An Ab initio investigation of surface and size effects. Nanomaterials 2023; 13(12), 1856.
SJ Babu, VN Rao, DHK Murthy, MMM Shastri, M Shetty, KSA Raju, PD Shivaramu, CSA Kumar, MV Shankar and D Rangappa. Significantly enhanced cocatalyst-free H2 evolution from defect-engineered Brown TiO2. Ceramics International 2021; 47(10), 14821-14828.
AHA Gonçalves, PHC Siciliano, OC Alves, DV Cesar, CA Henriques and AB Gaspar. Synthesis of a magnetic Fe3O4/RGO composite for the rapid photo-fenton discoloration of indigo carmine dye. Topics in Catalysis 2020; 63, 1017-1029.
CV Tran, PTH Nguyen, DD Nguyen, HTT Pham, DT Do and DD La. Facile fabrication of Fe2O3/TiO2 composite from titanium slag as adsorbent for As(V) removal from aqueous media. Sustainability 2023; 15(9), 7253.
TT Loan, VH Huong, NT Huyen, LV Quyet, NA Bang and NN Long. Anatase to rutile phase transformation of iron-doped titanium dioxide nanoparticles: The role of iron content. Optical Materials 2021; 111, 110651.
MD Patekari, KK Pawar, GB Salunkhe, PM Kodam, MN Padvi, PP Waifalkar, KK Sharma and PS Patil. Synthesis of maghemite nanoparticles for highly sensitive and selective NO2 sensing. Materials Science and Engineering: B 2021; 272, 115339.
N Abbas, GN Shao, MS Haider, SM Imran, SS Park and HT Kim. Sol-gel synthesis of TiO2-Fe2O3 systems: Effects of Fe2O3 content and their photocatalytic properties. Journal of Industrial and Engineering Chemistry 2016; 39, 112-120.
ES Wibowo and B-D Park. Determination of crystallinity of thermosetting urea-formaldehyde resins using deconvolution method. Macromolecular Research 2020; 28, 615-624.
MS Khan, JA Shah, N Riaz, TA Butt, AJ Khan, W Khalifa, HH Gasmi, ER Latifee, M Arshad, AAA Al-Naghi, A Ul-Hamid, M Arshad and M Bilal. Synthesis and characterization of Fe-TiO2 nanomaterial: Performance evaluation for RB5 decolorization and in vitro antibacterial studies. Nanomaterials 2021; 11(2), 436.
AM Oviedo, H Truong Thi, QC Van and HH Nguyen. Physicochemical properties of Fe-doped TiO2 and the application in Dye-sensitized solar cells. Optical Materials 2023; 137, 113587.
R Hatefi, H Younesi, A Mashinchian-Moradi and S Nojavan. A facile decoration of anatase Fe3O4/TiO2 nanocomposite with graphene quantum dots: Synthesis, characterization, and photocatalytic activity. Advanced Powder Technology 2021; 32(7), 2410-2422.
S Pan, M Zha, C Gao, J Qu and X Ding. Pore structure and fractal characteristics of organic-rich lacustrine shales of the kongdian formation, cangdong sag, bohai bay basin. Frontiers in Earth Science 2021; 9, 760583.
FJ Sotomayor, KA Cychosz and M Thommes. Characterization of micro/mesoporous materials by physisorption: Concepts and case studies. Accounts of Materials & Surface Research 2018; 3(2), 34-50.
S Badawy and P Pandey. Chapter 28 - design, development, and scale-up of the high-shear wet granulation process. Developing Solid Oral Dosage Forms (Second Edition) 2017; 2017, 749-776.
YQ Cao, TQ Zi, XR Zhao, C Liu, Q Ren, JB Fang, WM Li and AD Li. Enhanced visible light photocatalytic activity of Fe2O3 modified TiO2 prepared by atomic layer deposition. Scientific Reports 2020; 10, 13437.
A Khalid, P Ahmad, AI Alharth, S Muhammad, MU Khandaker, MRI Faruque, IU Din and MA Alotaibi. A practical method for incorporation of Fe (III) in Titania matrix for photocatalytic applications. Materials Research Express 2021; 8(4), 045006.
M Gao, L Zhu, CK Peh and GW Ho. Solar absorber material and system designs for photothermal water vaporization towards clean water and energy production. Energy and Environmental Science 2019; 12(3), 841-864.
E Inico, C Saetta and GD Liberto. Impact of quantum size effects to the band gap of catalytic materials: A computational perspective. Journal of Physics Condensed Matter 2024; 36(36), 361501.
P Kaur and G Verma. Converting fruit waste into carbon dots for bioimaging applications. Materials Today Sustainability 2022; 18, 100137.
C Wen, X Ni, M Han, Y Yu, C Liu, Y Zhang, B Zheng and S Feng. The function of photocatalytic performance and carrier separation efficiency tuned by doping content in homogeneous photocatalysts. Advanced Science 2025; 12(25), 2501026.
N Kanjana, W Maiaugree, P Poolcharuansin and P Laokul. Synthesis and characterization of Fe-doped TiO2 hollow spheres for dye-sensitized solar cell applications. Materials Science and Engineering: B 2021; 271, 115311.
M Coduri, P Masala, L Bianco, F Spizzo, D Ceresoli, C Castellano, S Cappelli, C Oliva, S Checchia, M Allieta, DV Szabo, S Schlabach, M Hagelstein, C Ferrero and M. Scavini. Local structure and magnetism of Fe2O3 maghemite nanocrystals: The role of crystal dimension. Nanomaterials 2020; 10(5), 867.
AA Isari, A Payan, M Fattahi, S Jorfi and B Kakavandi. Photocatalytic degradation of rhodamine B and real textile wastewater using Fe-doped TiO2 anchored on reduced graphene oxide (Fe-TiO2/rGO): Characterization and feasibility, mechanism and pathway studies. Applied Surface Science 2018; 462, 549-564.
MSM Zaini and SSA Syed-Hassan. Effects of different physical activation agents on adsorbent pore development and methane uptake. Recent Innovations in Chemical Engineering 2022; 15(2), 127-137.
A Srivastava, JAK Satrughna, MK Tiwari, A Kanwade, SC Yadav, K Bala and PM Shirage. Effect of Ti1-xFexO2 photoanodes on the performance of dye-sensitized solar cells utilizing natural betalain pigments extracted from Beta vulgaris (BV). Energy Advances 2023; 2(1), 148-160.
E Faustino, TF da Silva, RF Cunha, DRV Guelfi, PS Cavalheri, SC de Oliveira, ARL Caires, GA Casagrande, RP Cavalcante and AM Junior. Synthesis and characterization of N and Fe-Doped TiO2 nanoparticles for 2,4-Dimethylaniline mineralization. Nanomaterials 2022; 12(15), 2538.
VE Novala and JG Carriazo. Fe3O4-TiO2 and Fe3O4-SiO2 core-shell powders synthesized from industrially processed magnetite (Fe3O4) microparticles. Materials Research 2019; 22(3), 20180660.
PRS Baabu, HK Kumar, MB Gumpu, J Babu K, AJ Kulandaisamy and JBB Rayappan. Iron oxide nanoparticles: A review on the province of its compounds, properties and biological applications. Materials 2023; 16(1), 59.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






