Comparative Efficacy of Endogenous Versus Exogenous Antioxidants in Small Ruminants Sperm Cryopreservation
DOI:
https://doi.org/10.48048/tis.2026.12196Keywords:
Antioxidant, Cryopreservation, Oxidative Stress, Goat Sperm, Zero Hunger, Antioxidant, Cryopreservation, Oxidative stress, Goat sperm, Zero hungerAbstract
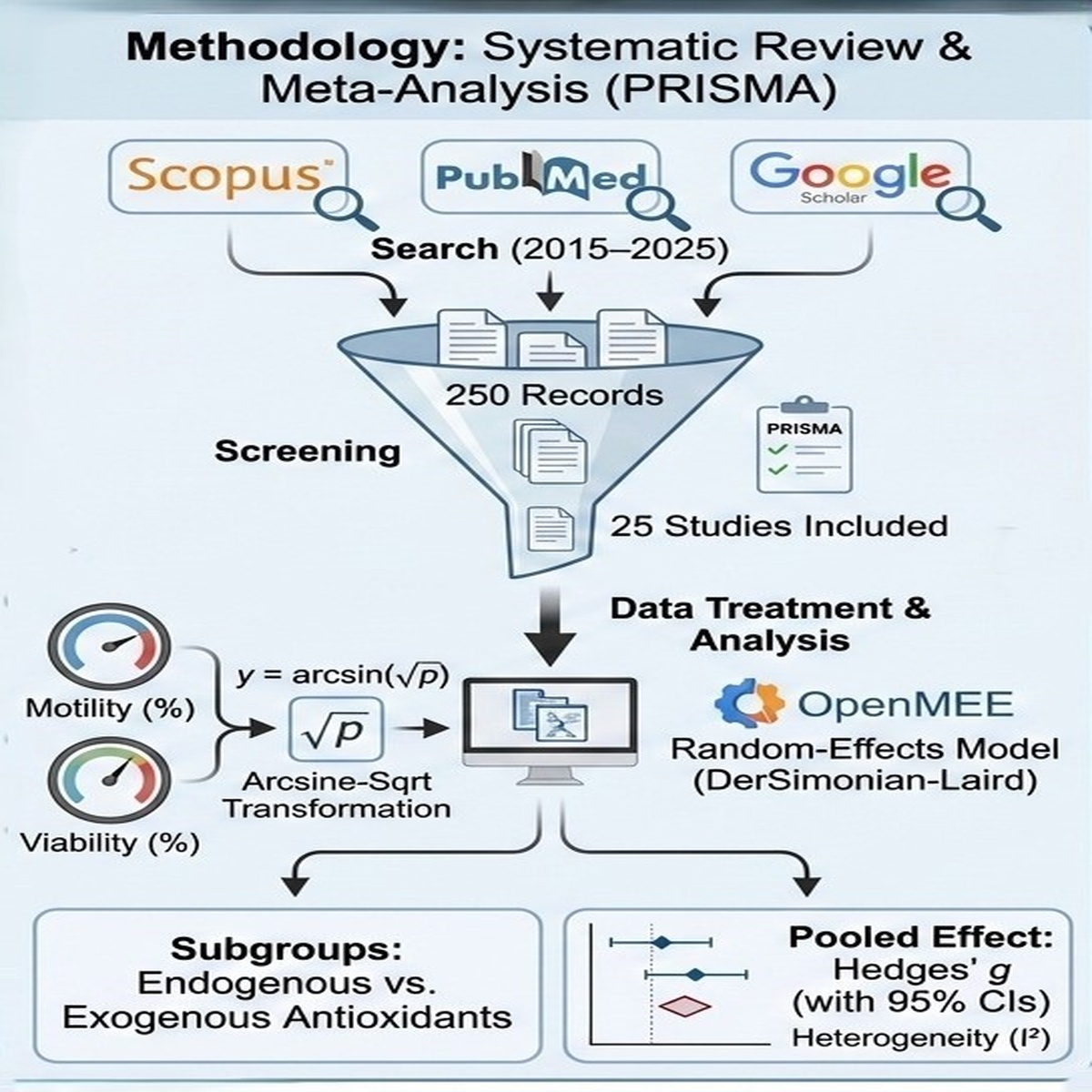
Small ruminant semen is highly susceptible to freeze-thaw oxidative damage due to PUFA-rich membranes, making antioxidant defense central to cryopreservation outcomes. Following PRISMA guidance, we searched Scopus, PubMed, and Google Scholar (2015 - 2025). Of 250 records screened, 25 studies met inclusion criteria. Effects were pooled as Hedges’ g under random-effects models (primary analyses in OpenMEE, DerSimonian-Laird; sensitivity verified with REML). Percentage outcomes (motility, viability) were arcsine-square-root transformed before SMD calculation. Subgroups contrasted endogenous vs exogenous antioxidants; heterogeneity (I²) and 95% CIs are reported. For motility, endogenous antioxidants showed a small decrease versus controls (SMD = –0.182; 95% CI –0.359 to –0.004; p = 0.045; I² = 12%), while exogenous antioxidants showed no clear effect (SMD = 0.025; 95% CI –0.144 to 0.194; p = 0.773; I² = 0%). The overall effect on motility was non-significant (SMD = –0.080; 95% CI –0.199 to 0.039; p = 0.186). For viability, both subgroups improved outcomes versus controls, with a larger pooled effect for exogenous (SMD = 5.150; 95% CI 2.810 to 7.490; I² = 59.94%) than endogenous (SMD = 4.105; 95% CI 0.100 to 8.109; I² = 92.14%); the overall pooled estimate was positive (SMD = 4.621; 95% CI 2.070 to 7.172). Sensitivity analyses (excluding higher-risk studies and using MD on the original percentage scale) did not materially change interpretations. The data suggest that endogenous antioxidants improve post-thaw viability but may not reliably sustain motility, whereas exogenous antioxidants may offer superior advantages to viability under particular circumstances. These results should be approached with caution due to significant variability in reporting and substantial differences in viability. Standardized methods, fixed dosages, and consistent outcome metrics are crucial to clarify the timing and recipients of external supplementation that improves beyond innate defenses.
HIGHLIGHTS
- Conducted a PRISMA-guided systematic review and meta-analysis of 25 studies on small ruminant semen cryopreservation (2015 - 2025).
- Applied OpenMEE random-effects models to evaluate the differential effects of endogenous and exogenous antioxidants.
- Found that endogenous antioxidants significantly reduced motility (SMD = –0.182; p = 0.045) but provided a modest improvement in viability.
- Demonstrated that motility and viability alone are sufficient indicators to reveal contrasting impacts of antioxidant origin, highlighting exogenous supplementation as the more reliable strategy for protecting goat sperm during cryopreservation.
GRAPHICAL ABSTRACT
Downloads
References
Ministry of Agriculture. Annual report on livestock and animal health sector in Indonesia. Ministry of Agriculture, Jakarta, Indonesia, 2023.
Directorate General of Livestock and Animal Health. National livestock revitalization program 2020 - 2025. Ministry of Agriculture, Jakarta, Indonesia, 2022.
W Zhang, L Min, Y Li, Y Lang, SAM Hoque, AO Adetunji and Z Zhu. Beneficial effect of proline supplementation on goat spermatozoa quality during cryopreservation. Animals 2022; 12(19), 2626.
W Shen, Y Fu, H Bai, Z Zhang, Z Cao, Z Liu, C Yang, S Sun, L Wang, C Ren, Y Ling, Z Zhang and H Cao. Antioxidant activity and metabolic regulation of sodium salicylate on goat sperm at low temperature. Animal Bioscience 2024; 37(4), 640-654.
Y Oktanella, I Mustofa, FAR An-Haru, DDM Putri, VF Hendrawan, S Susilowati, NY Degu and T Hernawati. Conserving goat sperm post-thawed gene expression and cellular characteristics using the antioxidant coenzyme Q10 supplementation. Veterinary world 2024; 17(7), 1637-1647.
B Khazravi, M Khodaei-Motlagh, R Masoudi and M Yahyaei. Coenzyme Q10 preserves buck’s sperm quality during cryopreservation process in plant-based extender. Animal Reproduction Science 2024; 265, 107487.
J Zou, L Wei, D Li, Y Zhang, G Wang, L Zhang, P Cao, S Yang and G Li. Effect of glutathione on sperm quality in Guanzhong dairy goat sperm during cryopreservation. Frontiers in Veterinary Science 2021; 8, 771440.
MG Al-Mutary, MQ Al-Ghadi, AA Ammari, AR Al-Himadi, AH Al-Jolimeed, MW Arafah, RA Amran, MS Aleissa and AA-A Swelum. Effect of different concentrations of resveratrol on the quality and in vitro fertilizing ability of ram semen stored at 5 °C for up to 168 h. Theriogenology 2020; 152, 139-146.
IŽ Žaja, V Berta, H Valpotić, M Samardžija, S Milinković-Tur, M Vilić and S Vince. The influence of exogenous melatonin on antioxidative status in seminal plasma and spermatozoa in French Alpine bucks during the nonbreeding season. Domestic Animal Endocrinology 2020; 71, 106400.
M Monteiro, M Silva, RAJA Arruda, LCP Oliveira, AS Mergulhão, FCC Monteiro Júnior, PLJ Batista, AM and MMP Guerra. Effect of melatonin in different extenders on the quality of frozen semen of goats. Emerging Animal Species 2022; 5(1-4), 100015.
M Alvarez-Rodriguez, M Alvarez, L Anel-Lopez, F Martinez-Pastor, S Borragan, L Anel and P de Pazz. Sperm membrane fatty acid composition in the cryopreservation of goat semen: a pilot study. Andrologia 2020; 52(2), e13484.
L Falchi, S Pau, I Pivato, L Bogliolo and MT Zedda. Resveratrol supplementation and cryopreservation of buck semen. Cryobiology 2020; 95, 60-67.
SA Hassan, WA Khalil, MAE Hassan, AI Yousif, OM Sabry, M Wink and M Sobeh. Antioxidant and antiapoptotic effects of a Turraea fischeri leaf extract on cryopreserved goat sperm. Animals 2021; 11(10), 2840.
SN Abedin, A Baruah, KK Baruah, A Bora, DJ Dutta, G Kadirvel, R Katiyar, S Doley, S Das, G Khargharia, B Sarkar, S Sinha, A Phookan, RK Dewry, MK Kalita, H Chakravarty and S Deori. Zinc oxide and selenium nanoparticles can improve semen quality and heat shock protein expression in cryopreserved goat (Capra hircus) spermatozoa. Journal of Trace Elements in Medicine and Biology 2023; 80, 127296.
MA Khalique, H Rehman, SMH Andrabi, KA Majeed, N Ahmad, MH Fayyaz, M Haider, S Naz, SS Qureshi and S Sulaiman. Antioxidant effects of zinc-oxide nanoparticles on post-thaw quality and in vivo fertility of Beetal buck spermatozoa. Small Ruminant Research 2023; 225, 107012.
C Li, Y Zhang, J Wang, H Liu, X Zhou and W Li. Effects of quercetin supplementation on cryopreserved goat sperm quality: Focus on oxidative stress mitigation and mitochondrial protection. PeerJ 2024a; 12, e17580.
F Liu, J Dai, J Gao, M He, J Xu, C Wu, S Zhang, X Zhu and L Sun. BGP-15 improves quality of goat sperm by mitigating oxidative stress during cryopreservation. Cryobiology 2025a; 119, 105232.
C Li, L Allai, J Liang, C Lv, X Zhao, X Ni, G Wu, W Deng, B Badaoui and G Quan. The antioxidant effects of butylated hydroxytoluene on cryopreserved goat sperm from a proteomic perspective. PeerJ 2024b; 12, e17580.
A Memon, HW Wahid, Y Rosnina, YM Goh, M Ebrahimi, FM Nadia and G Audrey. Effect of butylated hydroxytoluene on cryopreservation of Boer goat semen in Tris egg yolk extender. Animal Reproduction Science 2011; 129(1-2), 44-49.
AG Miah, R Bathgate, KI Hamano and U Salma. Effects of pre-freeze Nigella sativa oil supplementation on cryosurvival of ovine spermatozoa. Reproduction in Domestic Animals 2018; 53(6), 1424-1433.
SS Susilowati, I Mustofa, W Wurlina, T Hernawati, Y Oktanella, S Soeharsono and DA Purwanto. Green tea extract in the extender improved the post-thawed semen quality and decreased amino acid mutation of Kacang buck sperm. Veterinary Sciences 2022; 9(8), 403.
E Kerasioti, D Stagos, A Tzimi and D Kouretas. Increase in antioxidant activity by sheep/goat whey protein through nuclear factor-like 2 (Nrf2) is cell type dependent. Food and Chemical Toxicology 2016; 97, 47-56.
M Piri, AH Mahdavi, M Hajian, A Shahverdi, A Asadi, V Asgari, MH NasrEsfahani and H Gourabi. Effects of nano-berberine and berberine loaded on green synthesized selenium nanoparticles on cryopreservation and in vitro fertilization of goat sperm. Scientific Reports 2024; 14, 24171.
A Kafi, M Khalid, T Herath and C Kershaw. Cysteine supplementation pre-freeze and post-thaw improves integrity and reduces oxidative stress in cryopreserved ram spermatozoa. Cryobiology 2024; 114, 104854.
N Asadzadeh, Z Abdollahi, S Esmaeilkhanian and R Masoudi. Fertility and flow cytometry evaluations of ram frozen semen in plant-based extender supplemented with Mito-TEMPO. Animal Reproduction Science 2021; 233, 106836.
MJ Page, D Moher, PM Bossuyt, I Boutron, TC Hoffmann, CD Mulrow, L Shamseer, JM Tetzlaff, EA Akl, SE Brennan, R Chou, J Glanville, JM Grimshaw, A Hróbjartsson, MM Lalu, T Li, EW Loder, E Mayo-Wilson, S McDonald, LA McGuinness, LA Stewart, J Thomas, AC Tricco, VA Welch, P Whiting and JE McKenzie. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021; 372, n160.
Y Wang, X Fu and H Li. Mechanisms of oxidative stress-induced sperm dysfunction. Frontiers in Endocrinology 2025; 16, 1520835.
I Yánez-Ortiz, J Catalán, JE Rodríguez-Gil, J Miró and M Yeste. Advances in sperm cryopreservation in farm animals: Cattle, horse, pig and sheep. Animal Reproduction Science 2022; 246, 106904.
MJ Shepherd and LA Herickhoff. A novel experimental design for boar sperm cryopreservation. Journal of Animal Science 2022; 100(7), skac169.
B Kushawaha, RS Yadav, DK Swain, P Kumari, A Kumar, B Yadav, M Anand, S Yadav, D Singh and SK Garg. Collapsed mitochondrial cristae in goat spermatozoa due to mercury result in lethality and compromised motility along with altered kinematic patterns. Scientific Reports 2021; 11(1), 646.
S Zang, S Zou, X Chen, B Pan, A Ning, J Qin, Y Wei, K Du, J Ye, Q Liang, Y Fang, Q Cirenlamu, T Song and G Zhou. Abnormalities in mitochondrial energy metabolism induced by cryopreservation negatively affect goat sperm motility. Frontiers in Veterinary Science 2025; 11, 1514362.
Z Al-Kass and JM Morrell. Freezing stallion semen - What do we need to focus on for the future? Veterinary Sciences 2024; 11(2), 65.
A Kowalczyk. The role of the natural antioxidant mechanism in sperm cells. Reproductive Sciences 2022; 29, 1387-1394.
F Ren, T Feng, G Dai, Y Wang, H Zhu and J Hu. Lycopene and alpha-lipoic acid improve semen antioxidant enzymes activity and cashmere goat sperm function after cryopreservation. Cryobiology 2018; 84, 27-32.
Y Oktanella, I Mustofa, S Susilowati, VV Hendrawan and T Hernawati. Effect of cryopreservation duration time on post-thawing sperms’ characteristics of goat semen. Jurnal Veteriner 2024b; 25(2), 175-185.
M Shafiei, M Forouzanfar, SM Hosseini and MH Esfahani. The effect of superoxide dismutase mimetic and catalase on the quality of post-thawed goat semen. Theriogenology 2015; 83(8), 1321-1327.
F Suwor, S Kubota, S Nawong, A Thuangsanthia, M Toyra, P Paengkoum and S Ponchunchoovong. Effects of cryoprotectant combinations on post-thawed sperm quality, biomolecular changes, DNA methylation, and pregnancy rates in Boer goat semen. Veterinary Sciences 2025; 12(2), 178.
R Ranjan, P Singh, C Gangwar, SP Singh, DK Swain and SD Kharche. Fortification of catalase improves post-thaw fertility of goat semen. Iranian Journal of Applied Animal Science 2021; 11(3), 587-593.
A Sarangi, P Singh, M Virmani, AS Yadav, S Sahu, HM Ajithakumar, A Kumari and AP Rath. Effect of antioxidants supplementation on the quality of Beetal buck semen stored at 4°C. Veterinary World 2017; 10(10), 1184-1188.
SMT Tironi, L Sitó-Silva, BL de Camillo, R Denadai, ALA Silva, CP Freitas-Dell'Aqua, JAD Junior, RA de Oliveira, MIL Souza and E Oba. Use of coenzyme Q-10 to improve the pregnancy rate in sheep. Animal Reproduction Science 2024, 266, 107498.
CV Souza, FZ Brandão, JDR Santos, VAP Alfradique, VMB Santos, MCC Morais, PSC Rangel, AA Silva and JMG Souza-Fabjan. Effect of different concentrations of L-carnitine in extender for semen cryopreservation in sheep. Cryobiology 2019; 89, 104-108.
A Najafi, MH Najafi, Z Zanganeh, M Sharafi, F Martínez-Pastor and H Adeldust. Effect of different concentrations of L-carnitine in extender for semen cryopreservation in sheep. Cryobiology 2019; 89, 6-10.
C Carriço, JP Barbas, J Pimenta and J Simões. Effect of in vitro addition of melatonin and glutathione on seminal parameters of rams in diluted semen and after thawing. Veterinary Sciences 2023; 10(7), 446.
S Akhter, M Zubair, M Mahmood, SMH Andrabi, N Hameed, E Ahmad and MK Saleemi. Effects of vitamins C and E in tris citric acid glucose extender on chilled semen quality of Kail ram during different storage times. Scientific Reports 2023; 13(1), 18123.
MS Nazif, ZU Rehman, H Khan, FA Khan, T Hussain, A Ahmad, Farmanullah, A Husnain, S Muhammad, G Murtaza and L Gang. Glycine improved cryopreserved spermatozoa quality in Achai bull. BioMed Research International 2022; 2022(1), 8282387.
I Batool, MH Fayyaz, A Hameed, SMH Andrabi, R Kausar, M Shahzad, Y Mubashir, AD Omur, G Murtaza, A Ditta and T Hussain. Quercetin in semen extender improves frozen-thawed spermatozoa quality and in-vivo fertility in crossbred Kamori goats. Frontiers in Veterinary Science 2024; 11, 1385642.
Z Zhu, H Zhao, H Cui, AO Adetunji and L Min. Resveratrol improves the frozen-thawed ram sperm quality. Animals 2023; 13(24), 3887.
I Mustofa, S Susilowati, TW Suprayogi, Y Oktanella, DA Purwanto and AO Akintunde. Combination of nanoparticle green tea extract in tris-egg yolk extender and 39 °C thawing temperatures improve the sperm quality of post-thawed Kacang goat semen. Animal Reproduction 2023; 19(4), e20220025.
T Suprayogi, W Wurlina, T Hernawati, S Susilowati, Y Oktanella and DA Purwanto. Improved quality of Kambing Kacang sexing frozen semen with the addition of green tea extract. Journal of Advanced Veterinary and Animal Research 2022; 9(3), 412.
S Nateq, G Moghaddam, S Alijani and M Behnam. The effects of different levels of nano selenium on the quality of frozen-thawed sperm in ram. Journal of Applied Animal Research 2020; 48(1), 434-439.
A Saratsi, F Samartzi, I Panagiotidis, A Basioura, D Tsiokos, C Ligda and CA Rekkas. Post-thaw parameters of buck semen quality after soy lecithin extender supplementation with fumaric acid. Veterinary Sciences 2023; 10(9), 569.
Z Abdollahi, S Zeinoaldini, M Zhandi, A Towhidi and H Baghshahi. Supplementation of plant-based cryopreservation medium with folic acid conserves the quality of bulk post-thawed spermatozoa. Archives of Razi Institute 2021; 76(3), 553-559.
DI Berean, LM Bogdan and R Cimpean. Advancements in understanding and enhancing antioxidant-mediated sperm cryopreservation in small ruminants: Challenges and perspectives. Antioxidants 2024; 13(6), 624.
H Younus. Therapeutic potentials of superoxide dismutase. International Journal of Health Sciences 2018; 12(3), 88-93.
J Zou, Y Chen, J Yang, H Shi, Z Wang and Y Xu. Melatonin protects goat spermatozoa against oxidative damage during cryopreservation by improving antioxidant capacity and mitochondrial function. Frontiers in Veterinary Science 2021; 8, 771440.
I Yousefian, M Emamverdi, A Karamzadeh-Dehaghani, R Sabzian-Melei, M Zhandi and A Zare-Shahneh. Attenuation of cryopreservation-induced oxidative stress by antioxidant: Impact of Coenzyme Q10 on the quality of post-thawed buck spermatozoa. Cryobiology 2018; 81, 88-93.
L Gallego-Calvo, MC Gatica, J Santiago-Moreno, JL Guzmán and LA Zarazaga. Exogenous melatonin does not improve the freezability of Blanca Andaluza goat semen over exposure to 2 months of short days. Animal Reproduction Science 2015; 157, 24-32.
A Sharon Sangeeta, S Arangasamy, S Kulkarni and S Selvaraju. Role of amino acids as additives on sperm motility, plasma membrane integrity and lipid peroxidation levels at pre-freeze and post-thawed ram semen. Animal Reproduction Science 2015; 161, 82-88.
J Zhao, P Meng, M Jin, X Ma, H Ma, H Yang and J Liu. Combined addition of L-carnitine and L-proline improves cryopreservation of dairy goat semen. Animal Reproduction Science 2023; 257, 107325.
C Lv, A Larbi, G Wu, Q Hong and G Quan. Improving the quality of cryopreserved goat semen with a commercial bull extender supplemented with resveratrol. Animal Reproduction Science 2019; 208, 106127.
T Hussain, MH Fayyaz, A Hameed, SMH Andrabi, R Kausar, Y Mubashir and AD Omur. Effect of resveratrol on post-thaw motility, kinematics, structural parameters and antioxidant/oxidant status of Kamori buck spermatozoa. Cryobiology 2025; 118, 105202.
F Liu, L Sun, J Xu, M He, C Wu, H Shen, H Zhu, F Luo, J Gao and J Dai. Isoglycyrrhizin supplementation of frozen goat semen-extender improves post-thaw sperm quality and in vitro fertilization rates. Theriogenology 2025; 234, 133-142.
I Mustofa, S Susilowati, W Wurlina, T Hernawati and Y Oktanella. Green tea extract increases the quality and reduced DNA mutation of post-thawed Kacang buck sperm. Heliyon 2021; 7(3), e06372.
JO Daramola, EO Adekunle, OE Oke, OM Onagbesan, OS Iyasere, TJ Williams and JA Oyewusi. Effects of pyridoxine supplementation or in combination with other antioxidants on motility, in vitro capacitation and acrosome reaction of goat buck spermatozoa during cryopreservation. Small Ruminant Research 2015; 131, 113-117.
JO Daramola, EO Adekunle, OS Iyasere, OE Oke, TA Sorongbe, OA Iyanda, AR Kehinde, SP Aluko, IO Olaoye, OE Gbadebo, LI Falolu, EO Olukayode, RA Ajayi, OJ Enikannaye and ED Osunjaiye. Effects of coconut milk alone or supplementation with pyridoxine in tris-extenders on viability of buck spermatozoa during vitrification. Small Ruminant Research 2016; 136, 208-213.
S Alcay, A Aktar, D Koca, MA Kilic, M Akkasoglu and H Sagirkaya. Positive effect of autologous platelet rich plasma on Saanen buck semen cryopreservation in non-breeding season. Cryobiology 2021; 103, 45-48.
MS Salama, AM Shehabeldin, MA Ashour, MQ Al-Ghadi, BH Marghani, I El-kon and M Shukry. Effect of the addition of platelet-rich plasma to Boer buck semen on sperm quality and antioxidant activity before and after cryopreservation and in vivo fertility. Small Ruminant Research 2024; 230, 107167.
VS Reddy, B Yadav, CL Yadav, M Anand, DK Swain, D Kumar and S Yadav. Effect of sericin supplementation on heat shock protein 70 (HSP70) expression, redox status and post thaw semen quality in goat. Cryobiology 2018; 84, 33-39.
A Kumar, JK Prasad, N Kumar, M Anand, S Verma, R Dhariya, A Kumar and A Gattani. Quercetin in semen extender curtails reactive oxygen and nitrogen species and improves functional attributes of cryopreserved buck semen. Cryobiology 2024; 116, 104931.
WL Singh, S Sinha, K Ahmed, L Buragohain, M Das Gupta, D Bhuyan, S Tamuly, SN Abedin, S Dhara and M Nath. IGF-1 outperforms vitamin E and crocin in improving semen quality, antioxidant profile and fertility gene expression in cryopreserved goat (Capra hircus) semen. Animal Reproduction Science 2025; 274, 107791.
A Rezaei, HR Bahmani, S Mafakheri, A Farshad, P Nazari and R Masoudi. Protective effects of different doses of MitoQ separately and combined with trehalose on oxidative stress and sperm function of cryopreserved Markhoz goat semen. Cryobiology 2023; 110, 36-43.
M Hatami, R Masoudi, A Hatefi, P Alipour-Jenaghard and V Esmaeili. The effects of MitoQ as a mitochondrial-targeted antioxidant in a plant-based extender on buck sperm quality parameters during cryopreservation. Animal Reproduction Science 2024; 266, 107517.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






