Effects of Different Morus alba L. Parts on Glycemic and Lipid Profiles: A Systematic Review and Meta-Analysis
DOI:
https://doi.org/10.48048/tis.2026.12148Keywords:
Morus, Mulberry, Metabolic syndrome, Blood glucose, Insulin, Lipids, Triglycerides, Cholesterol, Diabetes mellitus, DyslipidemiasAbstract
Metabolic syndrome is a growing global health challenge. Given the limitations of conventional therapies, mulberry (Morus alba L.) has gained attention as a functional food with potential metabolic benefits. This systematic review and meta-analysis evaluated recent evidence on its efficacy and safety across various plant parts. The review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and was registered with PROSPERO (CRD42024600762). Randomized controlled trials (RCTs) were identified from PubMed, Embase, Scopus, and gray literature sources, up to May 2025. Risk of bias was assessed using the Risk of Bias 2 tool. Meta-analysis applied a random-effects model, with subgroup analyses to explore heterogeneity. Eighteen RCTs were included in this review. The meta-analysis showed that single mulberry intervention significantly lowered postprandial glucose (PPG) levels compared to the control group at 30 min (MD = −10.37 mg/dL; 95% CI: −19.30 to −1.45; p = 0.02) and 60 min (MD = −6.01 mg/dL; 95% CI: −11.97 to −0.05; p = 0.05). In addition, significant reductions were observed in the PPG area under the curve (AUC), PPG-positive incremental AUC, and the postprandial insulin (PPI) total AUC over 120 min. The leaf subgroup showed notable effects on both PPG and PPI levels. For long-term effects, 4 - 16 weeks of mulberry intervention significantly lowered glycated hemoglobin A1c (HbA1c) (MD = −0.30%; 95% CI: −0.56 to −0.05; p = 0.02), with no significant changes in fasting blood glucose (FBG), fasting plasma insulin (FPI), or lipid profiles. Mulberry treatment was well tolerated and considered safe for use. Mulberry, particularly the leaf, demonstrated significant efficacy in improving postprandial glycemic responses and HbA1c levels, with a favorable safety profile. Further research is called for to assess any long-term effects and the potential of other plant parts.
HIGHLIGHTS
- Single administration of mulberry products significantly reduced postprandial glycemic responses, including glucose and insulin parameters.
- Long-term use (4 - 16 weeks) improved HbA1c levels but showed no effect on fasting blood glucose, insulin, and lipid profiles.
- Mulberry leaf subgroup demonstrated reductions in both postprandial glucose and insulin, as well as a significant decrease in HbA1c, supporting its potential as a functional food for glycemic control.
- Mulberry interventions were well tolerated overall, with gastrointestinal symptoms as the most frequent adverse events. No serious adverse events were reported.
GRAPHICAL ABSTRACT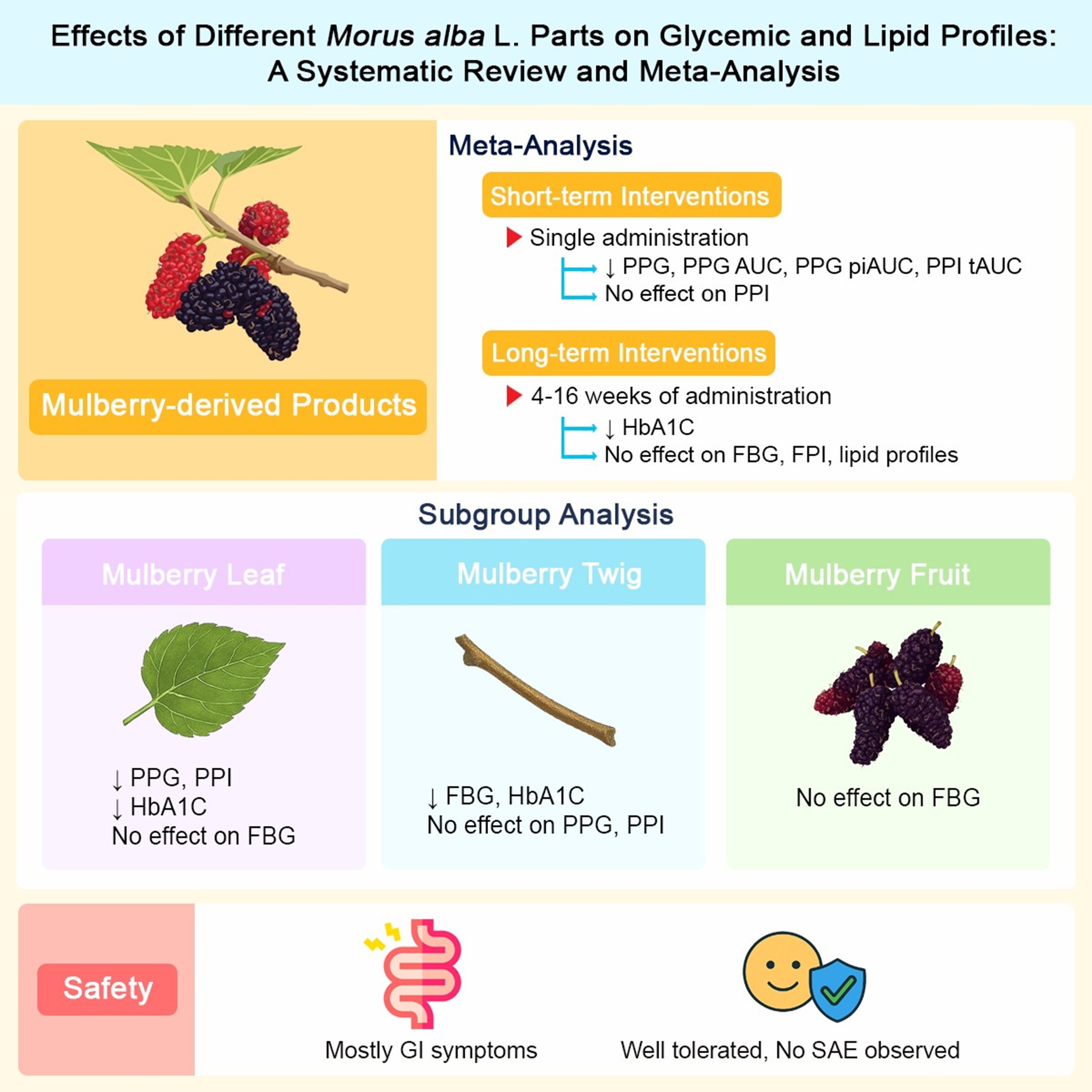
Downloads
References
A Engin. The definition and prevalence of obesity and metabolic syndrome. In: A Engin and AB Engin (Eds.). Obesity and lipotoxicity. Springer, Cham, Switzerland, 2017.
G Yousefzadeh, A Sayyadi, H Najafipour, V Sabaghnejad and S Pezeshki. Comparing the association of 2 metabolic syndrome definitions, NCEP ATP III and IDF, with the risk of developing atherosclerotic cardiovascular disease: An analytical cross-sectional study. Endocrinology, Diabetes & Metabolism 2024; 7(1), e468.
B Ulambayar, AS Ghanem, NM Chau, EV Faludi, M Móré and AC Nagy. Evaluation of cardiovascular disease risk in patients with type 2 diabetes mellitus using clinical laboratory markers. Journal of Clinical Medicine 2024; 13(12), 3561.
MA Mengstie, EC Abebe, TA Dejenie, MA Seid and AA Teshome. Frequency and correlates of poor glycemic control in patients with type 2 diabetes at Jimma Medical Centre, Ethiopia: A cross-sectional study. Pan African Medical Journal 2024; 47, 7.
S Padhi, AK Nayak and A Behera. Type II diabetes mellitus: A review on recent drug-based therapeutics. Biomedicine & Pharmacotherapy 2020; 131, 110708.
RH Nelson. Hyperlipidemia as a risk factor for cardiovascular disease. Primary Care 2013; 40(1), 195-211.
N Thongtang, R Sukmawan, EJB Llanes and ZV Lee. Dyslipidemia management for primary prevention of cardiovascular events: Best in-clinic practices. Preventive Medicine Reports 2022; 27, 101819.
W Aekplakorn, S Chariyalertsak, S Bumrerraj, S Assanangkornchai, S Taneepanichskul, N Neelapaichit, B Ongphiphadhanakul and W Nitiyanant. Diabetes trends and determinants among Thai adults from 2004 to 2020. Scientific Reports 2025; 15, 31620.
LL de Oliveira, AC de Assis, VZ Giraldez, TL Scudeler and PR Soares. Dyslipidemia: A narrative review on pharmacotherapy. Pharmaceuticals 2024; 17(3), 289.
M Abdul-Ghani, RA DeFronzo, S Del Prato, R Chilton, R Singh and REJ Ryder. Cardiovascular disease and type 2 diabetes: Has the dawn of a new era arrived? Diabetes Care 2017; 40(7), 813-820.
S Vinzio, E Andrès, AE Perrin, JL Schlienger and B Goichot. Glibenclamide-induced acute haemolytic anaemia revealing a G6PD-deficiency. Diabetes Research and Clinical Practice 2004; 64(3), 181-183.
F Mach, C Baigent, AL Catapano, KC Koskinas, M Casula, L Badimon, MJ Chapman, GG De Backer, V Delgado, BA Ference, IM Graham, A Halliday, U Landmesser, B Mihaylova, TR Pedersen, G Riccardi, DJ Richter, MS Sabatine, MR Taskinen, L Tokgozoglu, O Wiklund and ESC Scientific Document Group. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. European Heart Journal 2020; 41(1), 111-188.
J Pedro-Botet, JM Núñez-Cortés, JA Flo res and J Rius. Muscle symptoms related with statin therapy in general practice. Atherosclerosis 2015; 241(1), e197.
M Mudra, N Ercan-Fang, L Zhong, J Furne and M Levitt. Influence of mulberry leaf extract on the blood glucose and breath hydrogen response to ingestion of 75 g sucrose by type 2 diabetic and control subjects. Diabetes Care 2007; 30(5), 1272-1274.
S Anchalee, B Akkarach, K Wiroje, S Tipanee and P Patcharanee. The effect of mulberry fruits consumption on lipid profiles in hypercholesterolemic subjects: A randomized controlled trial. Journal of Pharmacy and Nutrition Sciences 2016; 6(1), 7-14.
P Aramwit, N Bang and T Srichana. The properties and stability of anthocyanins in mulberry fruits. Food Research International 2010; 43(4), 1093-1097.
YR Esti Wulandari, VD Prasasty, A Rio and C Geniola. Determination of 1-deoxynojirimycin content and phytochemical profiles from young and mature mulberry leaves of Morus spp. OnLine Journal of Biological Sciences 2019; 19(2), 124-131.
L Qu, X Liang, G Tian, G Zhang, Q Wu, X Huang, Y Cui, Y Liu, Z Shen, G Ma, H Lu, Y Li, H Jiang, X Yang, G Zhang and C Yang. Efficacy and safety of mulberry twig alkaloids tablet for treatment of type 2 diabetes: A randomized, double-blind, placebo-controlled multicenter clinical study. Chinese Journal of Integrative Medicine 2022; 28(4), 304-311.
M Taghizadeh, A Mohammad Zadeh, Z Asemi, AH Farrokhnezhad, MR Memarzadeh, Z Banikazemi, M Shariat, and R Shafabakhsh. Morus Alba leaf extract affects metabolic profiles, biomarkers inflammation and oxidative stress in patients with type 2 diabetes mellitus: A double-blind clinical trial. Clinical Nutrition ESPEN 2022; 49, 68-73.
W Phimarn, K Wichaiyo, K Silpsavikul, B Sungthong and K Saramunee. A meta-analysis of efficacy of Morus alba Linn. to improve blood glucose and lipid profile. European Journal of Nutrition 2017; 56(4), 1509-1521.
SO Shin, HJ Seo, H Park and HJ Song. Effects of mulberry leaf extract on blood glucose and serum lipid profiles in patients with type 2 diabetes mellitus: A systematic review. European Journal of Integrative Medicine 2016; 8(5), 602-608.
M Tornaghi, A Ali Redha, M Rondanelli and S Perna. Exploring the impact of mulberry fruits on metabolic syndrome: A systematic review of current evidence. PharmaNutrition 2023; 26, 100363.
X Chen, MH Sohouli, M Nateghi, E Melekoglu and S Fatahi. Impact of mulberry consumption on cardiometabolic risk factors: A systematic review and meta-analysis of randomized-controlled trials. Journal of Clinical Pharmacy and Therapeutics 2022; 47(12), 1982-1993.
MJ Page, JE McKenzie, PM Bossuyt, I Boutron, TC Hoffmann, CD Mulrow, L Shamseer, JM Tetzlaff, EA Akl, SE Brennan, R Chou, J Glanville, JM Grimshaw, A Hróbjartsson, MM Lalu, T Li, EW Loder, E Mayo-Wilson, S McDonald, LA McGuinness, LA Stewart, J Thomas, AC Tricco, VA Welch, P Whiting and D Moher. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021; 372, n71.
JPT Higgins, J Savović, MJ Page, RG Elbers and JAC Sterne. Assessing risk of bias in a randomized trial. In: JPT Higgins, J Thomas, J Chandler, M Cumpston, T Li, MJ Page and VA Welch (Eds.). Cochrane handbook for systematic reviews of interventions, version 6.5. The Cochrane Collaboration, London, 2024.
JPT Higgins, T Li and JJ Deeks. Choosing effect measures and computing estimates of effect. In: JPT Higgins, J Thomas, J Chandler, M Cumpston, T Li, MJ Page and VA Welch (Eds.). Cochrane handbook for systematic reviews of interventions, version 6.5. The Cochrane Collaboration, London, 2024.
JPT Higgins, SG Thompson, JJ Deeks and DG Altman. Measuring inconsistency in meta-analyses. BMJ 2003; 327(7414), 557-560.
MJ Page, JPT Higgins and JAC Sterne. Assessing risk of bias due to missing evidence in a meta-analysis. In: JP Higgins, T James, J Chandler, M Cumpston, T Li, M Page and VA Welch (Eds.). Cochrane handbook for systematic reviews of interventions, version 6.5. The Cochrane Collaboration, London, 2024.
HJ Schünemann, JPT Higgins, GE Vist, P Glasziou, EA Akl, N Skoetz and GH Guyatt. Completing ‘summary of findings’ tables and grading the certainty of the evidence. In: JPT Higgins, J Thomas, J Chandler, M Cumpston, T Li, MJ Page and VA Welch (Eds.). Cochrane handbook for systematic reviews of interventions, version 6.5. The Cochrane Collaboration, London, 2024.
PS Thondre, I Butler, J Tammam, I Achebe, E Young, M Lane and A Gallagher. Understanding the impact of different doses of Reducose® mulberry leaf extract on blood glucose and insulin responses after eating a complex meal: Results from a double-blind, randomised, crossover trial. Nutrients 2024; 16, 1670.
PS Thondre, H Lightowler, L Ahlstrom and A Gallagher. Mulberry leaf extract improves glycaemic response and insulaemic response to sucrose in healthy subjects: Results of a randomized, double blind, placebo-controlled study. Nutrition & Metabolism 2021; 18, 41.
DJ Mela, XZ Cao, S Govindaiah, H Hiemstra, R Kalathil, L Lin, J Manoj, T Mi and C Verhoeven. Dose-response efficacy of mulberry fruit extract for reducing post-prandial blood glucose and insulin responses: randomised trial evidence in healthy adults. British Journal of Nutrition 2023; 129(5), 771-778.
DJ Mela, M Alssema, H Hiemstra, AR Hoogenraad and T Kadam. Effect of low-dose mulberry fruit extract on postprandial glucose and insulin responses: A randomized pilot trial in individuals with type 2 diabetes. Nutrients 2024; 16, 2177.
DJ Mela, HM Boers, T Kadam, H Hiemstra, R Kalathil and JWMS Ten Hoorn. Effect of mulberry fruit extract on post-prandial glycaemic and insulinemic responses to different rice types: A randomised trial in healthy adults. British Journal of Nutrition 2023; 130(2), 2088-2094.
DJ Mela, XZ Cao, R Dobriyal, MI Fowler, L Lin, M Joshi, TJP Mulder PG Murray HPF Peters, MA Vermeer and Z Zhang. The effect of 8 plant extracts and combinations on post-prandial blood glucose and insulin responses in healthy adults: a randomized controlled trial. Nutrition & Metabolism 2020; 17, 51.
N Gheldof, C Francey, A Rytz, L Egli, F Delodder, L Bovetto, N Piccardi and C Darimont. Effect of different nutritional supplements on glucose response of complete meals in two crossover studies. Nutrients 2022; 14, 2674.
S Park, O Kwon and JY Kim. Effect of Mori ramulus on the postprandial blood glucose levels and inflammatory responses of healthy subjects subjected to an oral high-fat/sucrose challenge: A double-blind, randomized, crossover clinical trial. Biomedicine & Pharmacotherapy 2022; 146, 112552.
R Wang, Y Li, W Mu, Z Li, J Sun, B Wang, Z Zhong, X Luo, C Xie and Y Huang. Mulberry leaf extract reduces the glycemic indexes of 4 common dietary carbohydrates. Medicine 2018; 97(34), e11996.
P Sukriket, S Lookhanumarnjao and A Bumrungpert. The effect of mulberry leaf tea on postprandial glycemic control and insulin sensitivity: A randomized, placebo-controlled crossover study. Journal of Pharmacy and Nutrition Sciences 2016; 6(1), 33-37.
M Takahashi, Y Mineshita, J Yamagami, C Wang, K Fujihira, Y Tahara, H Kim, T Nakaoka and S Shibata. Effects of the timing of acute mulberry leaf extract intake on postprandial glucose metabolism in healthy adults: A randomised, placebo-controlled, double-blind study. European Journal of Clinical Nutrition 2023; 77(4), 468-473.
DM Riche, KD Riche, HE East, EK Barrett and WL May. Impact of mulberry leaf extract on type 2 diabetes (Mul-DM): A randomized, placebo-controlled pilot study. Complementary Therapies in Medicine 2017; 32, 105-108.
W Parklak, M Chottidao, N Munkong, S Komindr, S Monkhai, B Wanikorn, N Makaje, K Kulprachkarn, H Chuljerm and S Somnuk. Nutraceutical properties of Thai mulberry (Morus alba L.) and their effects on metabolic and cardiovascular risk factors in individuals with obesity: A randomized, single-blind crossover trial. Nutrients 2024; 16, 4336.
A Asai, K Nakagawa, O Higuchi, T Kimura, Y Kojima, J Kariya, T Miyazawa and S Oikawa. Effect of mulberry leaf extract with enriched 1‐deoxynojirimycin content on postprandial glycemic control in subjects with impaired glucose metabolism. Journal of Diabetes Investigation 2011; 2(4), 318-323.
T Thaipitakwong. 2020, Efficacy and safety of mulberry leaves on glycemic control in patients with obesity and patients with type 2 diabetes. Ph. D. Dissertation. Chulalongkorn University, Bangkok, Thailand.
JY Kim, HM Ok, J Kim, SW Park, SW Kwon and O Kwon. Mulberry leaf extract improves postprandial glucose response in prediabetic subjects: A randomized, double-blind placebo-controlled trial. Journal of Medicinal Food 2015; 18(3), 306-313.
HI Jeong, S Jang and KH Kim. Morus alba L. for blood sugar management: A systematic review and meta-analysis. Evidence‐Based Complementary and Alternative Medicine 2022; 2022(1), 9282154.
KS Hershon, BR Hirsch and O Odugbesan. Importance of postprandial glucose in relation to A1C and cardiovascular disease. Clinical Diabetes 2019; 37(3), 250-259.
H Haller. The clinical importance of postprandial glucose. Diabetes Research and Clinical Practice 1998; 40, S43-S49.
N den Braber, MMR Vollenbroek-Hutten, SEM Teunissen, MM Oosterwijk, KDR Kappert and GD Laverman. The contribution of postprandial glucose levels to hyperglycemia in type 2 diabetes calculated from continuous glucose monitoring data: Real-world evidence from the DIALECT-2 cohort. Nutrients 2024; 16, 3557.
American Diabetes Association. Postprandial blood glucose. Diabetes Care 2001; 24(4), 775-778.
Q Qiao, K Pyörälä, M Pyörälä, A Nissinen, J Lindström, R Tilvis and J Tuomilehto. Two-hour glucose is a better risk predictor for incident coronary heart disease and cardiovascular mortality than fasting glucose. European Heart Journal 2002; 23(16), 1267-1275.
F Cavalot, A Petrelli, M Traversa, K Bonomo, E Fiora, M Conti, G Anfossi, G Costa and M Trovati. Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: Lessons from the San Luigi Gonzaga Diabetes Study. Journal of Clinical Endocrinology & Metabolism 2006; 91(3), 813-819.
IH Cho, R Cheng, CW Jung, TH Won, D Wang, HH Jang, IG Hwang and SW Kwan. Effects of 1-Deoxynojirimycin on glycemic control: A systematic review and meta-analysis. NFS Journal 2025; 38, 100210.
N Asano, T Yamashita, K Yasuda, K Ikeda, H Kizu, Y Kameda, A Kato, RJ Nash, HS Lee and KS Ryu. Polyhydroxylated alkaloids isolated from mulberry trees (Morus alba L.) and silkworms (Bombyx mori L.). Journal of Agricultural and Food Chemistry 2001; 49(9), 4208-4213.
N Asano. Sugar-mimicking glycosidase inhibitors: Bioactivity and application. Cellular and Molecular Life Sciences 2009; 66(9), 1479-1492.
E Standl and O Schnell. Alpha-glucosidase inhibitors 2012 - cardiovascular considerations and trial evaluation. Diabetes and Vascular Disease Research 2012; 9(3), 163-169.
C Liu, W Xiang, Y Yu, ZQ Shi, XZ Huang and L Xu. Comparative analysis of 1-deoxynojirimycin contribution degree to α-glucosidase inhibitory activity and physiological distribution in Morus alba L. Industrial Crops and Products 2015; 70, 309-315.
LW Chang, LJ Juang, BS Wang, MY Wang, HM Tai, WJ Hung, YJ Chen and MH Huand. Antioxidant and antityrosinase activity of mulberry (Morus alba L.) twigs and root bark. Food and Chemical Toxicology 2011; 49(3), 785-790.
HM Boers, JS Ten Hoorn and DJ Mela. A systematic review of the influence of rice characteristics and processing methods on postprandial glycaemic and insulinaemic responses. British Journal of Nutrition 2015; 114(7), 1035-1045.
J Ju, J Li, Q Lin and H Xu. Efficacy and safety of berberine for dyslipidaemias: A systematic review and meta-analysis of randomized clinical trials. Phytomedicine 2018; 50, 25-34.
JJ Deeks, JPT Higgins, DG Altman, JE McKenzie and AA Veroniki. Analysing data and undertaking meta-analyses. In: JPT Higgins, J Thomas, J Chandler, M Cumpston, T Li, MJ Page and VA Welch (Eds.). Cochrane handbook for systematic reviews of interventions, version 6.5. The Cochrane Collaboration, London, 2024.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






