New Synthetic Routes, Wound Healing and Hemostatic Properties of N-Guanidinium Chitosan
DOI:
https://doi.org/10.48048/tis.2026.12110Keywords:
Chitosan, Modification, Guanidine, Acetonitrile, N-guanidinium chitosan, Wound healing, Hemostatic propertiesAbstract
The development of novel multifunctional biomaterials remains one of the most pressing challenges in modern materials science. Polymer derivatives of guanidine are in high demand due to their broad spectrum of pharmacological activities. In recent years, various methods have been developed to introduce guanidine groups into the chitosan structure. However, most of the known guanidination methods are carried out in the presence of acids, which leads to the formation of side reactions in the reaction medium. In this study, we present a method for guanidination of chitosan in an aprotic solvent medium - acetonitrile. It has been demonstrated that by varying the molar ratio of the guanidinating reagent, it is possible to obtain N-guanidinium chitosan derivatives with a degree of substitution ranging from 0.16 to 0.45. Notably, the use of acetonitrile prevents hydrolysis of the chitosan macromolecular backbone, and an increase in the number of guanidine groups leads to an increase in the molecular weight of the synthesized N-guanidinium chitosan derivatives. The structure and properties of the obtained chitosan derivatives were confirmed by physico-chemical methods (elemental analysis, FTIR, NMR, XRD, SEM, DLS, etc.). The presence of guanidine groups in the chitosan structure conferred broad pH-range solubility to the synthesized compounds. Results from a comparative in vivo analysis demonstrated that 0.5% solutions of N-guanidinium chitosan exhibited excellent wound healing properties, attributed to the synergistic interaction between the polysaccharide backbone and the guanidine moieties. Pharmacological evaluations confirmed that the introduction of guanidine groups into the chitosan structure enhances its hemostatic efficacy compared to native chitosan.
HIGHLIGHTS
- This study chitosan was subjected to guanylation in an aprotic solvent medium.
- Guanylated chitosan derivatives with diverse physico-chemical properties and solubility across a broad pH range were synthesized.
- The N-guanidinium chitosan samples demonstrated effective wound-healing and hemostatic properties.
- Findings suggest indicate the potential for developing new multifunctional polymeric materials based on chitosan.
GRAPHICAL ABSTRACT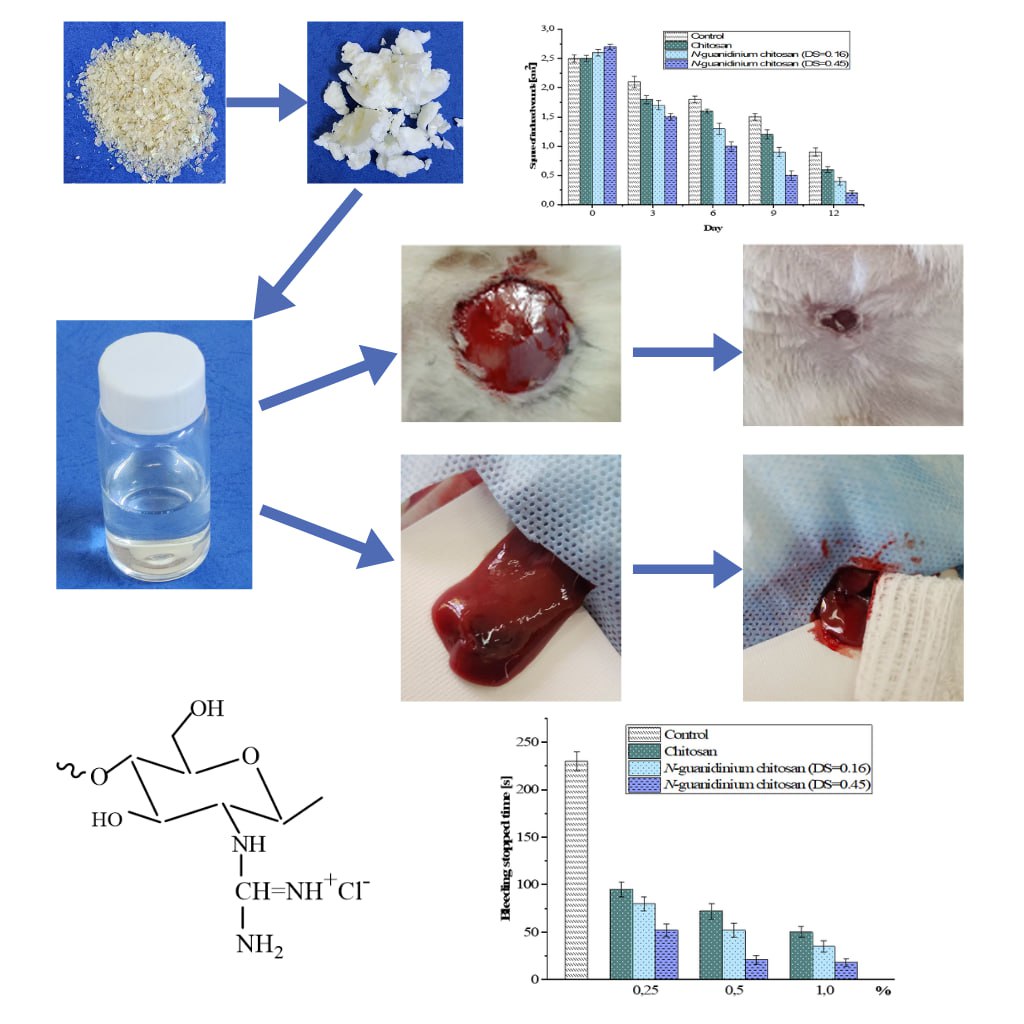
Downloads
References
MK Yazdi, F Seidi, A Hejna, P Zarrintaj, N Rabiee, J Kucinska-Lipka, MR Saeb and SA Bencherif. Tailor-made polysaccharides for biomedical applications. ACS Applied Bio Materials 2024; 7(7), 4193-4230.
ZU Arif. The role of polysaccharide-based biodegradable soft polymers in the healthcare sector. Advanced Industrial and Engineering Polymer Research 2025; 8(1), 132-156.
Q Meng, Y Sun, H Cong, H Hu and FJ Xu. An overview of chitosan and its application in infectious diseases. Drug Delivery and Translational Research 2021; 11, 1340-1351.
H Yadav, R Malviya and N Kaushik. Chitosan in biomedicine: A comprehensive review of recent developments. Carbohydrate Polymer Technologies and Applications 2024; 8, 100551.
A Sidarenka, A Kraskouski, V Savich, O Akhmedov, V Nikalaichuk, A Herasimovich, K Hileuskaya and V Kulikouskaya. Design of sponge-like chitosan wound dressing with immobilized bacteriophages for promoting healing of bacterially infected wounds. Journal of Polymers and the Environment 2024; 32, 3893-3909.
F Fitriagustiani, AZ Mustopa, S Budiarti, MF Warsito, RD Pratiwi, DF Agustiyanti and M Nurfatwa. Formulation and characterization of alginate coated chitosan nanoparticles as therapeutic protein for oral delivery system. Trends in Sciences 2022; 19(18), 5797.
L Chen, Y Xie, X Chen, H Li, Y Lu, H Yu and D Zheng. O-carboxymethyl chitosan in biomedicine: A review. International Journal of Biological Macromolecules 2024; 275(2), 133465
MES Miranda, C Marcolla, CA Rodrígues, HM Wilhelm, MR Sierakowski, TMB Bresolin and RA de Freitas. Chitosan and N-carboxymethylchitosan: I. The role of N-carboxymethylation of chitosan in the thermal stability and dynamic mechanical properties of its films. Polymer International 2006; 55, 961-969.
S Kalliola, E Repo, V Srivastava, F Zhao, JP Heiskanen, JA Sirviö, H Liimatainen and M Sillanpää. Carboxymethyl chitosan and its hydrophobically modified derivative as pH-switchable emulsifiers. Langmuir 2018; 34, 2800-2806.
C Ardean, NS Nemeş, A Negrea, M Ciopec, N Duteanu, P Negrea, M Ciopec, N Duteanu, P Negrea, D Duda-Seiman and V Musta. Factors influencing the antibacterial activity of chitosan and chitosan modified by functionalization. International Journal of Molecular Sciences 2021; 22(14), 7449.
H Tan, R Ma, C Lin, Z Liu and T Tang. Quaternized chitosan as an antimicrobial agent: antimicrobial activity, mechanism of action and biomedical applications in orthopedics. International Journal of Molecular Sciences 2013; 14(1), 1854-1869.
W Sajomsang, P Gonil and S Tantayanon. Antibacterial activity of quaternary ammonium chitosan containing mono or disaccharide moieties: Preparation and characterization. International Journal of Biological Macromolecules 2009; 44(5), 419-427.
T Ishikawa. Guanidine chemistry. Chemical and Pharmaceutical Bulletin 2010; 58(12), 1555-1564.
P Pham, S Oliver and C Boyer. Design of antimicrobial polymers. Macromolecular Chemistry and Physics 2023; 224(3), 2200226.
J Zhang, L Hu, Q Zhang, C Guo, C Wu, Y Shi, R Shu and L Tan. Polyhexamethylene guanidine hydrochloride modified sodium alginate nonwoven with potent antibacterial and hemostatic properties for infected full-thickness wound healing. International Journal of Biological Macromolecules 2022; 209, 2142-2150.
AR Gomes, CL Varela, AS Pires, EJ Tavares-da-Silva and FMF Roleira. Synthetic and natural guanidine derivatives as antitumor and antimicrobial agents: A review. Bioorganic Chemistry 2023; 138, 106600.
FV Drozdov and VM Kotov. Guanidine: A simple molecule with great potential: From catalysts to biocides and molecular glues. INEOS Open 2020; 3, 200-213.
OR Akhmedov, SA Shomurotov and AS Turaev. Comparative studies of the chemical interaction of guanidine with dialdehyde cellulose and pectin. Russian Journal of Bioorganic Chemistry 2023; 49(7), 1587-1595
HE Salama, GR Saad and MW Sabaa. Synthesis, characterization, and biological activity of cross-linked chitosan biguanidine loaded with silver nanoparticles. Journal of Biomaterials Science, Polymer Edition 2016; 27(18), 1880-1898.
A Salama and P Hesemann. New N-guanidinium chitosan/silica ionic microhybrids as efficient adsorbent for dye removal from waste water. International Journal of Biological Macromolecules 2018; 111, 762-768
OR Akhmedov, SA Shomurotov, AS Turaev and AV Sidarenka. Dependence of antimicrobial effects of guanidine-containing pectin derivatives from some structural characteristics. Drug Development and Registration 2022; 11(2), 38-45.
OR Akhmedov, SA Shomurotov and AS Turaev. Features of synthesis and antimicrobial properties of guanidine-containing carboxymethylcellulose derivatives. Russian Journal of Bioorganic Chemistry 2022; 48, 1379-1386.
X Zhao, JX He and YZ Zhan. Synthesis and characterization of chitosan biguanidine hydrochloride under microwave irradiation. Polymer Journal 2009; 41, 1030-1035.
LAB Rawlinson, SM Ryan, G Mantovani, JA Syrett, DM Haddleton and DJ Brayden. Antibacterial effects of poly(2-(dimethylamino ethyl)methacrylate) against selected gram-positive and gram-negative bacteria. Biomacromolecules 2010; 11(2), 443-453.
C de l’Europe. European Convention for the protection of vertebrate animals used for experimental and other scientific purposes. Official Journal 1986; L222, 37-87.
K Khainskaya, K Hileuskaya, V Nikalaichuk, A Ladutska, O Akhmedov, N Abrekova, L You, P Shao and M Odonchimeg. Chitosan-gallic acid conjugate with enhanced functional properties and synergistic wound healing effect. Carbohydrate Research 2025; 553, 109496.
Y Li, Q Leng, X Pang, H Shi, Y Liu, S Xiao, L Zhao, P Zhou and S Fu. Therapeutic effects of EGF-modified curcumin/chitosan nano-spray on wound healing. Regenerative Biomaterials 2021; 8(2), 1-9.
J Kumirska, M Czerwicka, Z Kaczyński, A Bychowska, K Brzozowski, J Thöming and P Stepnowski. Application of spectroscopic methods for structural analysis of chitin and chitosan. Marine Drugs 2010; 8, 1567-1636.
MS Joshi, TB Ferguson, FK Johnson and JR Lancaster. Receptor-mediated activation of nitric oxide synthesis by arginine in endothelial cells. Proceedings of the National Academy of Sciences 2007; 104(24), 9982-9987.
Y Zhou, G Liu, H Huang and J Wu. Advances and impact of arginine-based materials in wound healing. Journal of Materials Chemistry B 2021; 9, 6738-6750.
MB Witte and A Barbul. Arginine physiology and its implication for wound healing. Wound Repair and Regeneration 2003; 11(6), 419-423.
HP Shi, D Most, DT Efron, MB Witte and A Barbul. Supplemental L-arginine enhances wound healing in diabetic rats. Wound Repair and Regeneration 2003; 11(3), 198-203.
DJ Yang, SH Moh, DH Son, S You, AW Kinyua, CM Ko, M Song, J Yeo, YH Choi and KW Kim. Gallic acid promotes wound healing in normal and hyperglucidic conditions. Molecules 2016; 21(7), 899.
P Feng, Y Luo, C Ke, H Qiu, W Wang, Y Zhu, R Hou, L Xu and S Wu. Chitosan-based functional materials for skin wound repair: Mechanisms and applications. Frontiers in Bioengineering and Biotechnology 2021; 9, 650598.
Y Fang, Y Lin, L Wang, Y Weng, Q Chen and H Liu. Clotting blood into an adhesive gel by hemostatic powder based on cationic/anionic polysaccharides and laponite. Biomacromolecules 2024; 25(6), 3335-3344.
K Kaminski, K Szczubiałka, K Zazakowny, R Lach and M Nowakowska. Chitosan derivatives as novel potential heparin reversal agents. Journal of Medicinal Chemistry 2010; 53(10), 4141-4147.
KY Chen, TH Lin, CY Yang, YW Kuo and U Lei. Mechanics for the adhesion and aggregation of red blood cells on chitosan. Journal of Mechanics 2018; 34(5), 725-732.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






