Extraction Solvent Effects on Banana Blossom Bioactive Compounds: Enhanced Bioaccessibility via Gastrointestinal Digestion
DOI:
https://doi.org/10.48048/tis.2026.12055Keywords:
Banana blossoms, Bioactive compounds, Phenolic content, Flavonoid content, Aqueous extraction, Bioaccessibility, Gastrointestinal digestion, Agricultural byproducts, Banana blossoms, Bioactive compounds, Phenolic content, Flavonoid content, Aqueous extraction, Bioaccessibility, Gastrointestinal digestion, Agricultural byproductsAbstract
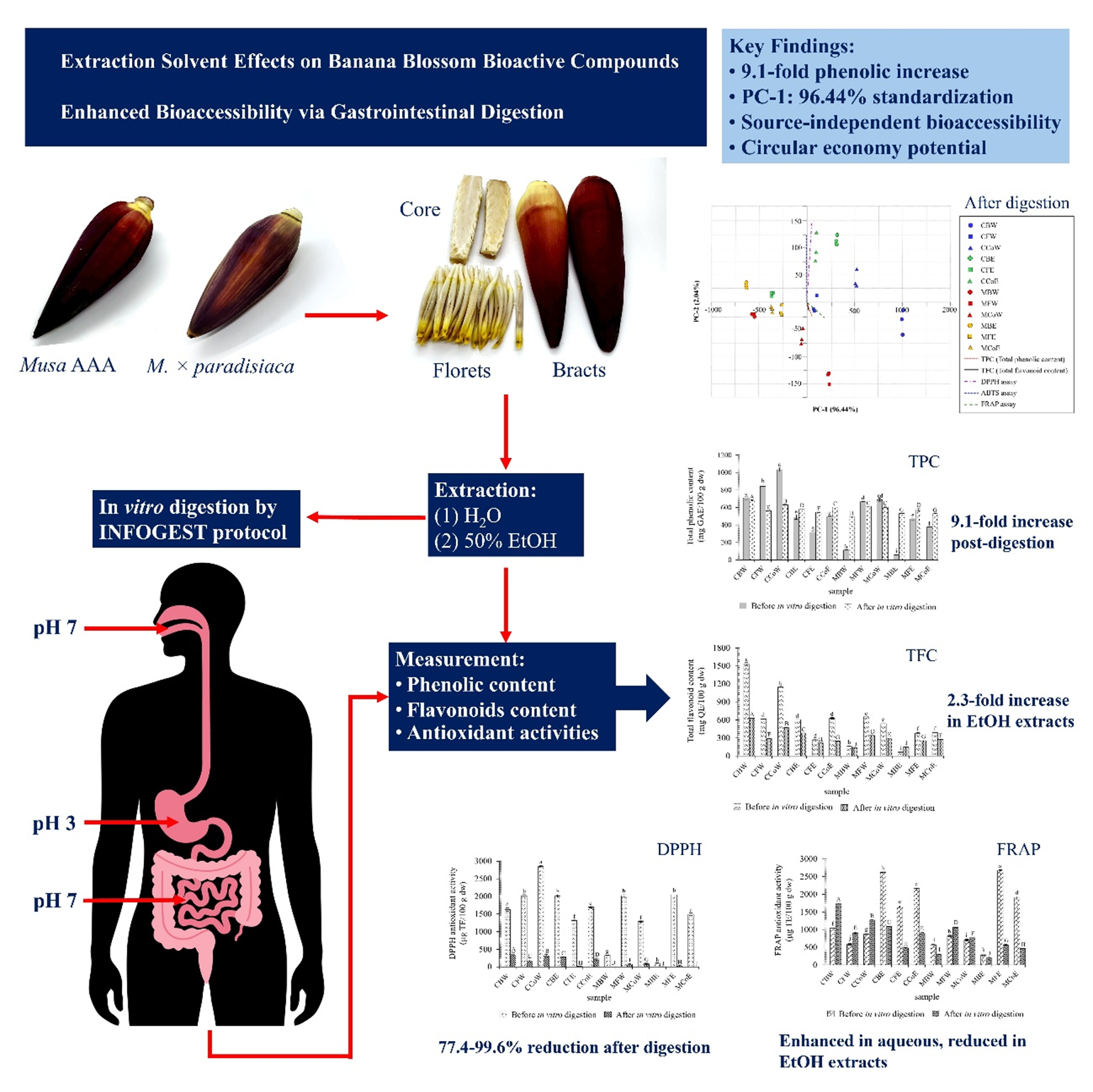
Banana blossoms are often discarded as agricultural byproducts, yet they contain bioactive compounds with potential health-promoting properties. The transformation of these underutilized materials into functional food ingredients represents an important circular economy strategy. Banana blossoms from the Musa AAA group and M. × paradisiaca cultivars were selected for analysis. Bioactive compounds and antioxidant capacity were evaluated in different anatomical parts (bracts, florets and core) through aqueous and 50% ethanol extraction. The extracts were assessed for bioaccessibility using in vitro gastrointestinal digestion following the INFOGEST protocol to determine their potential for developing functional foods. Bioactive content and antioxidant activities varied significantly across cultivars, anatomical parts, and extraction solvents. Musa AAA group showed superior performance, with aqueous core extracts achieving the highest phenolic content (1,033.57 ± 20.20 mg GAE/100 g) and aqueous bract extracts achieving the highest flavonoid content (1,529.23 ± 27.20 mg QE/100 g). Aqueous extraction was more effective than ethanol extraction. Gastrointestinal digestion produced contrasting effects: phenolic content increased substantially in certain extracts (9.1-fold in M. × paradisiaca bracts), while antioxidant activities decreased dramatically (77.4% - 99.6% reduction, p < 0.001). PCA confirmed that in vitro digestion resulted in standardized bioactive compound profiles regardless of source materials. PC-1 variance increased from 63.00% before digestion to 96.44% after digestion, demonstrating uniform bioaccessibility patterns across all samples. This study highlights banana blossoms as a promising source of bioactive compounds for functional food development, with aqueous extraction being effective and digestion ensuring uniform bioaccessibility. Their use can contribute to sustainable agricultural waste valorization.
HIGHLIGHTS
- First comprehensive evaluation of bioactive compound bioaccessibility in banana blossom anatomical parts
- Gastrointestinal digestion standardized bioactive profiles across different sources
- PC-1 explained 96.44% variance post-digestion vs. 63.00% pre-digestion
- Source selection became less critical for functional food applications
- Supports circular economy through valorization of banana blossom by-products
GRAPHICAL ABSTRACT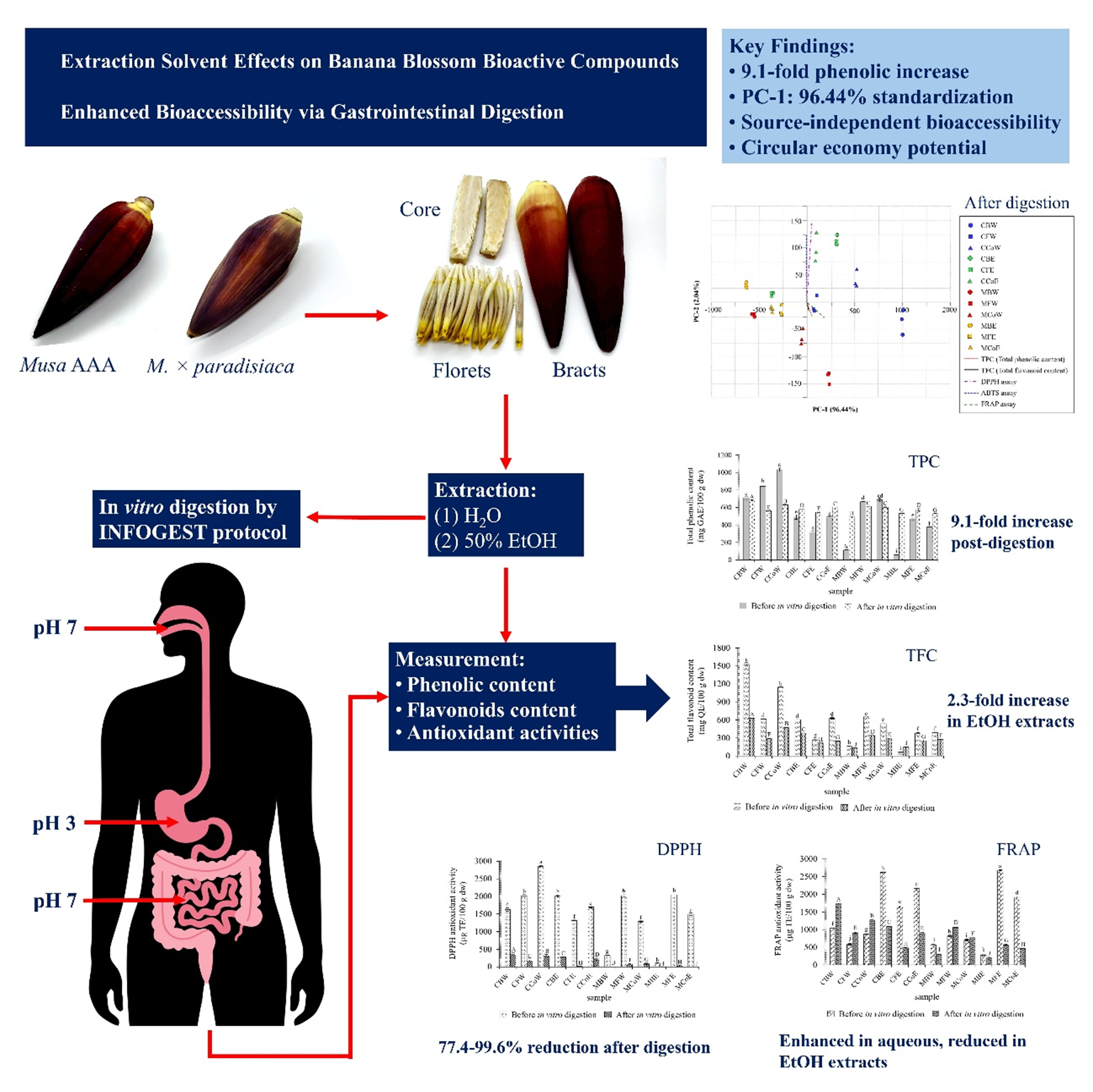
Downloads
References
M Al-Dairi, PB Pathare, R Al-Yahyai, H Jayasuriya and Z Al-Attabi. Evaluation of chemical quality attributes in bruised bananas during storage. LWT - Food Science and Technology 2024; 197, 115904.
V Voora. 2024. Global Market Report: Banana prices and sustainability. Institute for Sustainable Development (IISD). Available at: https://www.iisd.org/publications/report/global-market-report-banana, accessed August 2025.
B Tsegaye, S Jaiswal and AK Jaiswal. Food waste biorefinery: Pathway towards circular bioeconomy. Foods 2021; 10(6), 1174.
S Chiang, K Yang, Y Lai and C Chen. Evaluation of the in vitro biological activities of Banana flower and bract extracts and their bioactive compounds. International Journal of Food Properties 2021; 24(1), 1-16.
D Amornlerdpison, V Choommongkol, K Narkprasom and S Yimyam. Bioactive compounds and antioxidant properties of banana inflorescence in a beverage for maternal breastfeeding. Applied Sciences 2021; 11(1), 343.
G Buntuchai, P Pavadhgul, W Kittipichai and W Satheannoppakao. Traditional galactagogue foods and their connection to human milk volume in Thai breastfeeding mothers. Journal of Human Lactation 2017; 33(3), 552-559.
JM Carbonell-Capella, M Buniowska, FJ Barba, MJ Esteve and A Frígola. Analytical methods for determining bioavailability and bioaccessibility of bioactive compounds from fruits and vegetables: A review. Comprehensive Reviews in Food Science and Food Safety 2014; 13(2), 155-171.
ACS Pais, ER Coscueta, MM Pintado, AJD Silvestre and SAO Santos. Exploring the bioaccessibility and intestinal absorption of major classes of pure phenolic compounds using in vitro simulated gastrointestinal digestion. Heliyon 2024; 10(7), e28894.
JAM Pereira, CV Berenguer, CFP Andrade and JS Câmara. Unveiling the bioactive potential of fresh fruit and vegetable waste in human health from a consumer perspective. Applied Sciences 2022; 12(5), 2747.
M Palermo, N Pellegrini and V Fogliano. The effect of cooking on the phytochemical content of vegetables. Journal of the Science of Food and Agriculture 2014; 94(6), 1057-1070.
A Ribas-Agustí, O Martín-Belloso, R Soliva-Fortuny and P Elez-Martínez. Food processing strategies to enhance phenolic compounds bioaccessibility and bioavailability in plant-based foods. Critical Reviews in Food Science and Nutrition 2018; 58(15), 2531-2548.
Z Wang, X Yao, C Jia, B Xu, J Wang, J Liu and Z Jin. Identification and analysis of lignin biosynthesis genes related to fruit ripening and stress response in banana (Musa acuminata L. AAA group, cv. Cavendish). Frontiers in Plant Science 2023; 14, 1072086.
N Phacharapiyangkul, K Thirapanmethee, K Sa-ngiamsuntorn, U Panich, C Lee and MT Chomnawang. The ethanol extract of Musa sapientum Linn. peel inhibits melanogenesis through AKT signaling pathway. Cosmetics 2021; 8(3), 70.
S Thaweesang. Antioxidant activity and total phenolic compounds of fresh and blanching banana blossom (Musa ABB CV.Kluai “Namwa”) in Thailand. IOP Conference Series: Materials Science and Engineering 2019; 639, 012047.
S Thummajitsakul and K Silprasit. Kineticsof tyrosinase inhibition, antioxidant activity, total flavonoid dontent and analysis of Averrhoa bilimbi L. extracts and its fruit vinegar using FTIR and multivariate methods. Trends in Sciences 2023; 20(2), 3641.
L Lajoie, A Fabiano-Tixier and Chemat. Water as green solvent: Methods of solubilisation and extraction of natural products - past, present and future solutions. Pharmaceuticals 2022; 15(12), 1507.
NA ElNaker, M Daou, MA Ochsenkühn, SA Amin, AF Yousef and LF Yousef. A metabolomics approach to evaluate the effect of lyophilization versus oven drying on the chemical composition of plant extracts. Scientific Reports 2021; 11(1), 22679.
VL Singleton, R Orthofer and RM Lamuela-Raventós. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods in Enzymology 1999; 299, 152-178.
J Zhishen, T Mengcheng and W Jianming. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chemistry 1999; 64(4), 555-559.
W Brand-Williams, ME Cuvelier and C Berset. Use of a free radical method to evaluate antioxidant activity. LWT 1995; 28(1), 25-30.
R Re, N Pellegrini, A Proteggente, A Pannala, M Yang and C Rice-Evans. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine 1999; 26(9-10), 1231-1237.
IF Benzie and JJ Strain. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry 1996; 239(1), 70-76.
M Minekus, M Alminger, P Alvito, S Balance, T Bohn, C Bourlieu, M Corredig, D Dupont, C Dufour, L Egger, M Golding, S Karakaya, B Kirkhus, SL Feunteun, U Lesmes, A Macierzanka, A Mackie, S Marze, DJ McClements, O Menard, I Recio, CN Santos, RP Singh, GE Vegarud, MSJ Wickham, W Weitschies and A Brodkorb. A standardised static in vitro gastrointestinal digestion method suitable for food: An international consensus. Food & Function 2014; 5(6), 1113-1124.
M Li, Q Bai, J Zhou, TSP de Souza and HAR Suleria. In vitro gastrointestinal bioaccessibility, bioactivities and colonic fermentation of phenolic compounds in different Vigna beans. Foods 2022; 110, 3884.
S Ramírez-Bolaños, J Perez-Jimenez, J Diaz and L Robaina. A potential of banana flower and pseudo-stem as novel ingredients rich in phenolic compounds. International Journal of Food Science and Technology 2021; 56(11), 5601-5608.
TT Falowo, IP Ejidike, L Lajide and HS Clayton. Polyphenolic content of Musa acuminata and Musa paradisiaca bracts: Chemical composition, antioxidant and antimicrobial potentials. Biomedical and Pharmacology Journal 2021; 14(4), 1767-1780.
JS Boeing, EO Barizão, BCE Silva, PF Montanher, V de Cinque Almeida and JV Visentainer. Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: Application of principal component analysis. Chemistry Central Journal 2014; 8(1), 48.
M Li, C Ritzoulis, Q Du, Y Liu, Y Ding, W Liu and J Liu. Recent progress on protein-polyphenol complexes: Effect on stability and nutrients delivery of oil-in-water emulsion system. Frontiers in Nutrition 2021; 8, 765589.
A Cenci, Y Hueber, Y Zorrilla-Fontanesi, J van Wesemael, E Kissel, M Gislard, J Sardos, R Swennen, M Roux, SC Carpentier and M Rouard. Effect of paleopolyploidy and allopolyploidy on gene expression in banana. BMC Genomics 2016; 20(1), 244.
K Slámová, J Kapešová and K Valentová. “Sweet Flavonoids”: Glycosidase-catalyzed modifica¬tions. International Journal of Molecular Sciences 2018; 19(7), 2126.
X Luo, M Tian, Y Cheng, C Ji, S Hu, H Liu, J Lu and J Ren. Effects of simulated in vitro gastrointestinal digestion on antioxidant activities and potential bioaccessibility of phenolic compounds from K. coccinea fruits. Frontiers in Nutrition 2022; 9, 1024651.
OA Sánchez-Velázquez, M Mulero, EO Cuevas-Rodríguez, M Mondor, Y Arcand and AJ Hernández-Álvarez. In vitro gastrointestinal digestion impact on stability, bioaccessibility and antioxidant activity of polyphenols from wild and commercial blackberries (Rubus spp.). Food & Function 2021; 12(16), 7358-7378.
K Wojtunik-Kulesza, A Oniszczuk, T Oniszczuk, M Combrzynski, D Nowakowska and A Matwijczuk. Influence of in vitro digestion on composition, bioaccessibility and antioxidant activity of food polyphenols - A non-systematic review. Nutrients 2020; 12(5), 1401.
GL Chen, SG Chen, F Chen, YQ Xie, MD Han, CX Luo, YY Zhao and YQ Gao. Nutraceutical potential and antioxidant benefits of selected fruit seeds subjected to an in vitro gastrointestinal digestion. Journal of Functional Foods 2016; 20, 317-331.
G Mandalari, M Vardakou, R Faulks, C Bisignano, M Martorana, A Smeriglio and D Trombetta. Food matrix effects of polyphenol bioaccessibility from almond skin during simulated human digestion. Nutrients 2016; 8(9), 568.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






