Effect of Malolactic Fermentation Conditions on Bioactive Compounds and Antioxidant Capacity of Red Dragon Fruit (Hylocereus polyrhizus) Wine
DOI:
https://doi.org/10.48048/tis.2026.11904Keywords:
Antioxidant, Betacyanin, Lactobacillus plantarum, Malolactic fermentation, Polyphenols, Red dragon fruitAbstract
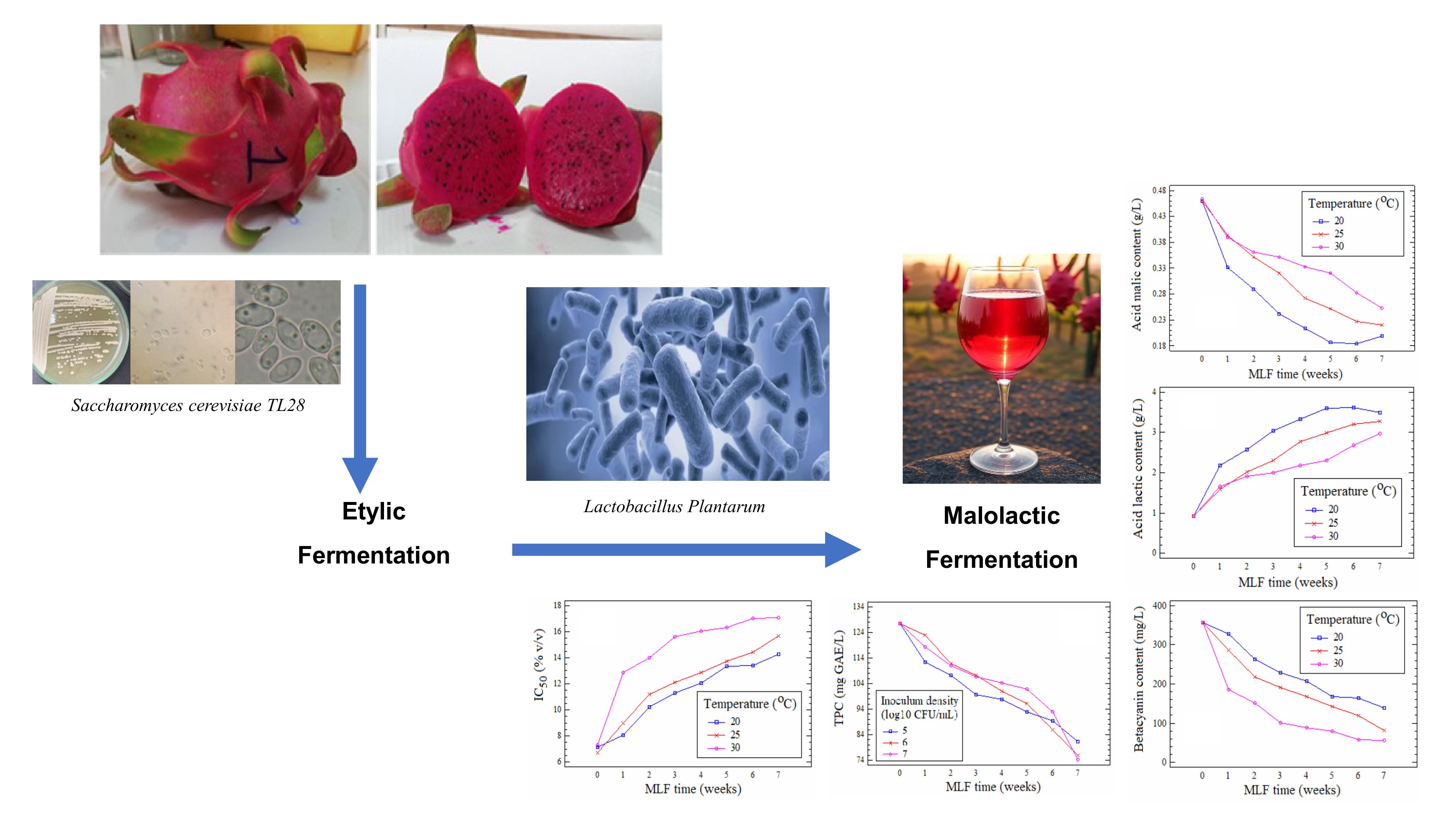
This study was conducted to evaluate the impact of malolactic fermentation (MLF) by Lactobacillus plantarum on the physicochemical and bioactive characteristics of red-fleshed dragon fruit (Hylocereus polyrhizus) wine. A 2-factor completely randomized design was applied, including 3 fermentation temperatures (20, 25 and 30 °C) and 3 bacterial inoculum levels (105, 106 and 107 CFU/mL). Parameters monitoring over 7 weeks included pH, malic acid, lactic acid, total polyphenol content, betacyanin, and antioxidant activity (IC50-DPPH). The results indicated that 20 °C combined with an inoculum of 106 CFU/mL represented the favorable condition, ensuring efficient malic acid degradation (0.16 g/L after 7 weeks), increased pH (4.06), while maintaining high levels of betacyanin (174 mg/L) and polyphenols (98.5 mg GAE/L), thereby preserving antioxidant capacity (IC50 = 13.1% v/v). These findings provide a scientific basis for optimizing the production process of red-fleshed dragon fruit wine, aiming to develop products with high sensory and bioactive value.
HIGHLIGHTS
- Evaluated the effects of temperature and inoculum density of Lactobacillus plantarum on malolactic fermentation in red-fleshed dragon fruit wine.
- Identified 20 °C with 106 CFU/mL inoculum as the favorable condition, achieving efficient malic acid degradation while preserving betacyanin and polyphenols.
- Demonstrated that betacyanin degradation followed a first-order kinetic model, strongly influenced by fermentation temperature and bacterial load.
- Showed that controlled MLF maintained antioxidant activity better than higher temperatures or excessive inoculum levels.
- Provided a scientific basis for optimizing MLF in tropical fruit wines to enhance bioactive value and functional potential.
GRAPHICAL ABSTRACT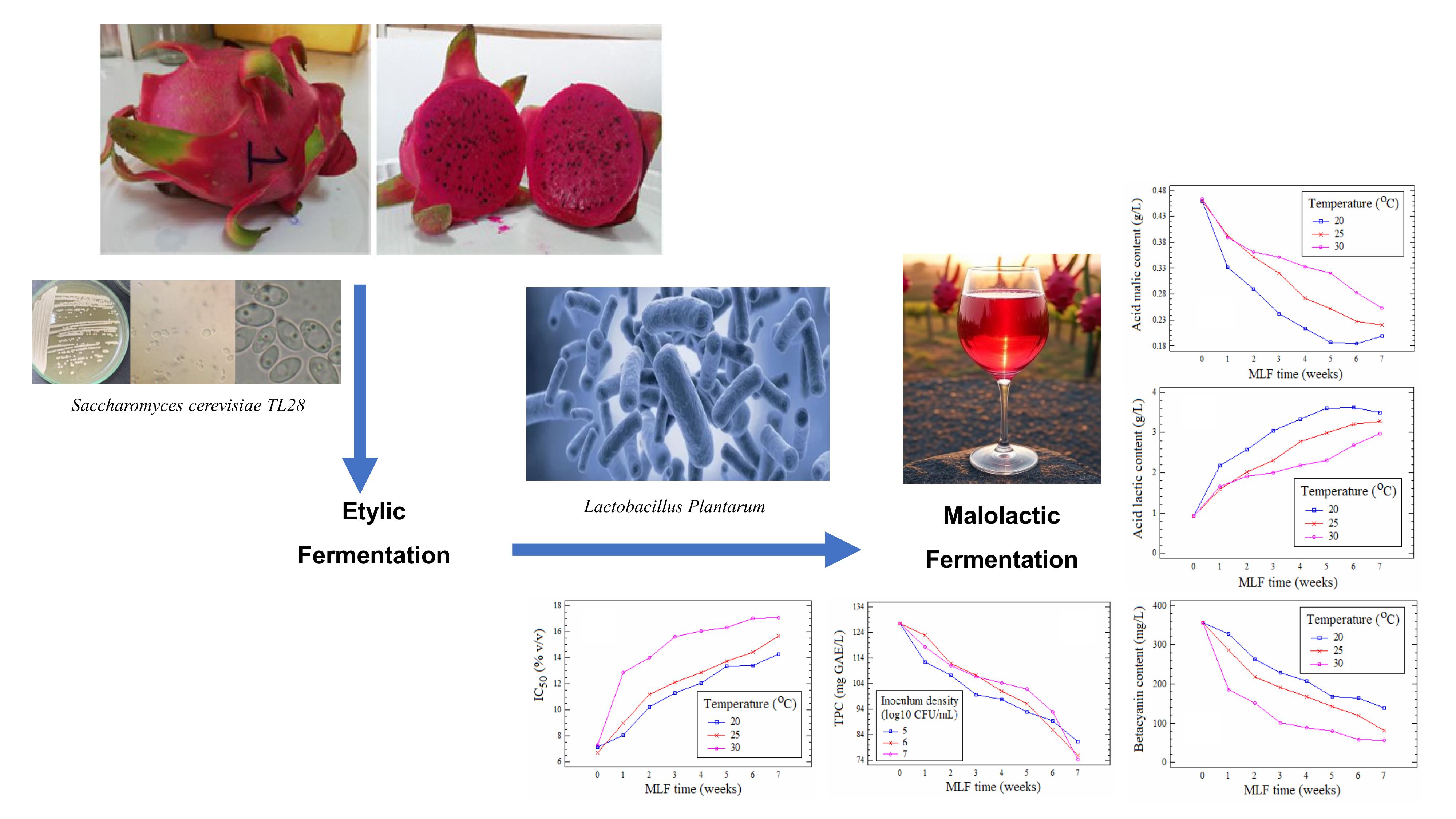
Downloads
References
TW Lim, CJ Lim, CA Liow, ST Ong, LH Lim, LP Pui, CP Tan and CW Ho. Studies on the storage stability of betacyanins from fermented red dragon fruit (Hylocereus polyrhizus) drink imparted by xanthan gum and carboxymethyl cellulose. Food Chemistry 2022; 393, 133404.
J Fu, L Wang, J Sun, N Ju and G Jin. Malolactic fermentation: New approaches to old problems. Microorganisms 2022; 10(12), 2363.
H Liu, C Zhang, B Zhang, W Jia, L He, Y Sun, J Li, P Zhou, X Guan and S Liu. A comparative study on the modification of polyphenolic, volatile, and sensory profiles of merlot wine by indigenous Lactiplantibacillus plantarum and Oenococcus oeni. Australian Journal of Grape and Wine Research 2024; 2024(1), 5602066.
YS Kumar, S Varakumar and O Reddy. Evaluation of antioxidant and sensory properties of mango (Mangifera indica L.) wine. CyTA-Journal of Food 2012; 10(1), 12-20.
C Prakitchaiwattana, K Boonin and P Kaewklin. De-acidification of fresh whole pineapple juice wine by secondary malolactic fermentation with lactic acid bacteria. International Food Research Journal 2017; 24(1), 223-231.
X Yuan, T Wang, L Sun, Z Qiao, H Pan, Y Zhong and Y Zhuang. Recent advances of fermented fruits: A review on strains, fermentation strategies, and functional activities. Food Chemistry: X 2024; 22, 101482.
M Du Toit, L Engelbrecht, E Lerm and S Krieger-Weber. Lactobacillus: The next generation of malolactic fermentation starter cultures - an overview. Food and Bioprocess Technology 2011; 4(6), 876-906.
M Succi, G Pannella, P Tremonte, L Tipaldi, R Coppola, M Iorizzo, SJ Lombardi and E Sorrentino. Sub-optimal pH preadaptation improves the survival of Lactobacillus plantarum strains and the malic acid consumption in wine-like medium. Frontiers in Microbiology 2017; 8, 470.
A Balmaseda, N Rozes, A Bordons and C Reguant. Characterization of malolactic fermentation by Lactiplantibacillus plantarum in red grape must. LWT 2024; 199, 116070.
DL Hawkins, J Ryder, SA Lee, K Parish‐Virtue, B Fedrizzi, MR Goddard and SJ Knight. Mixed yeast communities contribute to regionally distinct wine attributes. FEMS Yeast Research 2023; 23, foad005.
H Erten, H Tanguler, T Cabaroglu and A Canbas. The influence of inoculum level on fermentation and flavour compounds of white wines made from cv. Emir. Journal of the Institute of Brewing 2006; 112(3), 232-236.
M Ramírez and R Velázquez. The yeast Torulaspora delbrueckii: An interesting but difficult-to-use tool for winemaking. Fermentation 2018; 4(4), 94.
R Rezgui, R Walia, J Sharma, D Sidhu, K Alshagadali, S Ray Chaudhuri, A Saeed and P Dey. Chemically defined lactobacillus plantarum cell-free metabolites demonstrate cytoprotection in HepG2 cells through nrf2-dependent mechanism. Antioxidants 2023; 12(4), 930.
X Jiang, Y Lu and SQ Liu. Effects of different yeasts on physicochemical and oenological properties of red dragon fruit wine fermented with Saccharomyces cerevisiae, Torulaspora delbrueckii and Lachancea thermotolerans. Microorganisms 2020; 8(3), 315.
L Fu, BT Xu, XR Xu, RY Gan, Y Zhang, EQ Xia and HB Li. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chemistry 2011; 129(2), 345-350.
YM Wong and LF Siow. Effects of heat, pH, antioxidant, agitation and light on betacyanin stability using red-fleshed dragon fruit (Hylocereus polyrhizus) juice and concentrate as models. Journal of Food Science and Technology 2015; 52(5), 3086-3092.
S Sharma, K Hullatti, S Kumar and K Tiwari. Comparative antioxidant activity of Cuscuta reflexa and Cassytha filiformis. Journal of Pharmacy Research 2012; 5(1), 441-443.
MV Leal-Sánchez, R Jiménez-Díaz, A Maldonado-Barragán, A Garrido-Fernández and JL Ruiz-Barba. Optimization of bacteriocin production by batch fermentation of Lactobacillus plantarum LPCO10. Applied and Environmental Microbiology 2002; 68(9), 4465-4471.
A Karampatea, A Skendi, M Manoledaki and E Bouloumpasi. Predicting organic acid variation in white wine malolactic fermentation using a logistic model. Fermentation 2025; 11(5), 288.
PT Nguyen and HT Nguyen. Environmental stress for improving the functionality of lactic acid bacteria in malolactic fermentation. The Microbe 2024; 4, 100138.
F Kösebalaban and M Özilgen. Kinetics of wine spoilage by acetic acid bacteria. Journal of Chemical Technology & Biotechnology 1992; 55(1), 59-63.
D Mamlouk and M Gullo. Acetic acid bacteria: Physiology and carbon sources oxidation. Indian Journal of Microbiology 2013; 53(4), 377-384.
W Beaman. 2011, Effect of cell density and growth phase on malolactic fermentation by Oenococcus oeni. Honor’s Thesis. Cornell University, New York.
S Krieger-Weber, JM Heras and C Suarez. Lactobacillus plantarum, a new biological tool to control malolactic fermentation: A review and an outlook. Beverages 2020; 6(2), 23.
A Lonvaud-Funel. Lactic acid bacteria in the quality improvement and depreciation of wine. Antonie Van Leeuwenhoek 1999; 76(1), 317-331.
O Lucio, I Pardo, JM Heras, S Krieger and S Ferrer. Influence of yeast strains on managing wine acidity using Lactobacillus plantarum. Food Control 2018; 92, 471-478.
CAN Alves, ACTCT Biasoto, G da Silva Nunes, HO do Nascimento, RF do Nascimento, IG Oliveira, PC de Souza Leão and LB de Vasconcelos. Exploring the impact of elevated pH and short maceration on the deterioration of red wines: Physical and chemical perspectives. OENO One 2025; 59(1), 8111.
M De Angelis, R Di Cagno, C Huet, C Crecchio, PF Fox and M Gobbetti. Heat shock response in Lactobacillus plantarum. Applied and Environmental Microbiology 2004; 70(3), 1336-1346.
P Filannino, G Cardinali, CG Rizzello, S Buchin, M De Angelis, M Gobbetti and R Di Cagno. Metabolic responses of Lactobacillus plantarum strains during fermentation and storage of vegetable and fruit juices. Applied and Environmental Microbiology 2014; 80(7), 2206-2215.
JY Da, MS Xi, HL Li, MM Liu, CH Zhou, ZY Li, YJ Song, S Zhou, TC Zhang and XG Luo. Transcriptome analysis and functional gene identification reveals potential mechanisms of heat stress response of Lactiplantibacillus plantarum CGMCC8198. Food Bioscience 2023; 51, 102202.
C Butzke. Preventing refermentation, Available at: https://www.extension.purdue.edu/extmedia/FS/FS-56-W.pdf, accessed May 2025.
IS Pretorius. Conducting wine symphonics with the aid of yeast genomics. Beverages 2016; 2(4), 36.
A Lonvaud-Funel. Effects of malolactic fermentation on wine quality. In: AG Reynolds (Ed.). Managing wine quality. Woodhead Publishing, Sawston, United Kingdom, 2010, p. 60-92.
N Brizuela, EE Tymczyszyn, LC Semorile, DV La Hens, L Delfederico, A Hollmann and B Bravo-Ferrada. Lactobacillus plantarum as a malolactic starter culture in winemaking: A new (old) player? Electronic Journal of Biotechnology 2019; 38, 10-18.
F Sun, S Liu, X Che, G Wang, X Wang, Y Li, S Zhang and H Chen. High-density fermentation of Lactobacillus plantarum P6: Enhancing cell viability via sodium alginate enrichment. Foods 2024; 13(21), 3407.
T Zotta, M Giavalisco, E Parente, G Picariello, F Siano and A Ricciardi. Selection of Lactiplantibacillus strains for the production of fermented table olives. Microorganisms 2022; 10(3), 625.
G Paventi, C Di Martino, F Coppola and M Iorizzo. β-glucosidase activity of Lactiplantibacillus plantarum: A key player in food fermentation and human health. Foods 2025; 14(9), 1451.
X Bai, X Chen, X Li, F Tan, FE Sam and Y Tao. Wine polyphenol oxidation mechanism and the effects on wine quality: A review. Comprehensive Reviews in Food Science and Food Safety 2024; 23(6), e70035.
S Paramithiotis, V Stasinou, A Tzamourani, Y Kotseridis and M Dimopoulou. Malolactic fermentation - theoretical advances and practical considerations. Fermentation 2022; 8(10), 521.
A Tzachristas, K Pasvanka, A Calokerinos and C Proestos. Polyphenols: Natural antioxidants to be used as a quality tool in wine authenticity. Applied Sciences 2020; 10(17), 5908.
I Sadowska-Bartosz and G Bartosz. Biological properties and applications of betalains. Molecules 2021; 26(9), 2520.
L Aztatzi-Rugerio, SY Granados-Balbuena, Y Zainos-Cuapio, E Ocaranza-Sánchez and M Rojas-López. Analysis of the degradation of betanin obtained from beetroot using Fourier transform infrared spectroscopy. Journal of Food Science and Technology 2019; 56(8), 3677-3686.
M Das, A Saeid, MF Hossain, GH Jiang, JB Eun and M Ahmed. Influence of extraction parameters and stability of betacyanins extracted from red amaranth during storage. Journal of Food Science and Technology 2019; 56(2), 643-653.
E Janiszewska-Turak, M Walczak, K Rybak, K Pobiega, M Gniewosz, Ł Woźniak and D Witrowa-Rajchert. Influence of fermentation beetroot juice process on the physico-chemical properties of spray dried powder. Molecules 2022; 27(3), 1008.
S Wardani, M Cahyanto, E Rahayu and T Utami. The effect of inoculum size and incubation temperature on cell growth, acid production and curd formation during milk fermentation by Lactobacillus plantarum Dad 13. International Food Research Journal 2017; 24(3), 921.
J Samoticha, A Wojdyło, J Chmielewska and J Oszmiański. The effects of flash release conditions on the phenolic compounds and antioxidant activity of Pinot noir red wine. European Food Research and Technology 2017; 243(6), 999-1007.
SA Ordoudi, F Mantzouridou, E Daftsiou, C Malo, E Hatzidimitriou, N Nenadis and MZ Tsimidou. Pomegranate juice functional constituents after alcoholic and acetic acid fermentation. Journal of Functional Foods 2014; 8, 161-168.
ZE Mousavi, SM Mousavi, SH Razavi, M Hadinejad, Z Emam-Djomeh and M Mirzapour. Effect of fermentation of pomegranate juice by Lactobacillus plantarum and Lactobacillus acidophilus on the antioxidant activity and metabolism of sugars, organic acids and phenolic compounds. Food Biotechnology 2013; 27(1), 1-13.
MA Varo, MP Serratosa, J Martín-Gómez, L Moyano and J Mérida. Influence of fermentation time on the phenolic compounds, vitamin C, color and antioxidant activity in the winemaking process of blueberry (Vaccinium corymbosum) wine obtained by maceration. Molecules 2022; 27(22), 7744.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






