DFT/TDDFT Study of Electronic and Optical Properties of Modified Indigofera Tinctoria Dyes as Solar Cell Sensitizers
DOI:
https://doi.org/10.48048/tis.2026.11890Keywords:
Indigofera tinctoria, Dye-sensitized solar cells, Density functional theory, Time-dependent DFT, Natural dyes, Light-harvesting efficiency, SDG 7 affordable and clean energy, SDG 13 climate actionAbstract
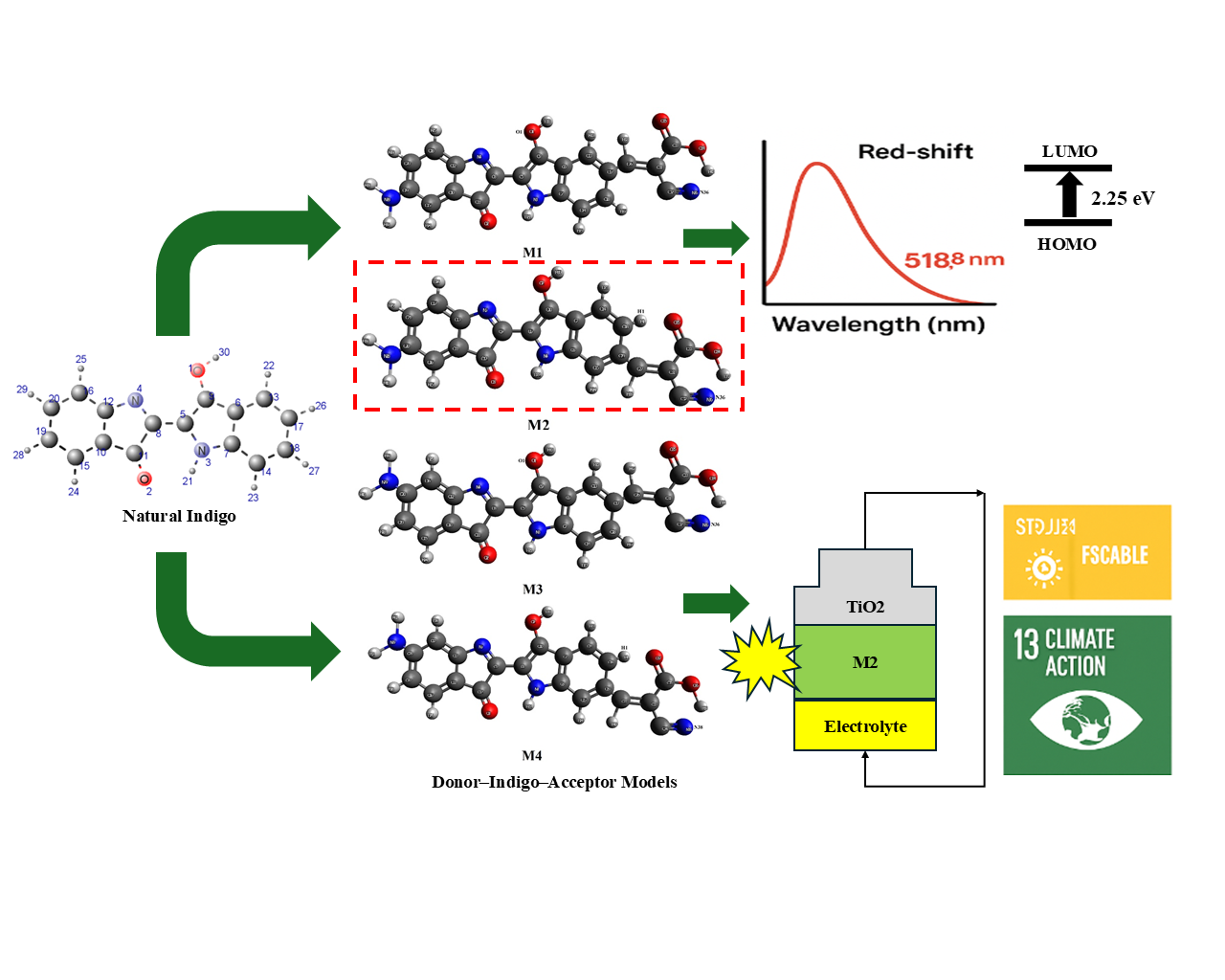
Renewable energy technology plays an important role in tssshe realization of sustainable development objectives, such as those by the United Nations (UN) on Sustainable Development Goals (SDGs). In this regard, the natural dye extracted from Indigofera tinctoria (indigo) is a potential source for low cost and eco-friendly photo-sensitizer which could be used in Dye-Sensitized Solar Cells (DSSCs). In this study, the electronic and optical characteristics of modified indigo derivatives are investigated for utilization in DSSCs using first-principles method. Four donor-indigo-acceptor (D-Indigo-A) architectures were developed by replacing the amine-type donating groups as well as the cyanoanistan acceptor conformations in the indigo scaffold. The DFT/TDDFT calculations at the B3LYP/def2-SVP level in DMF identified that these structure modifications brought about remarkable improvements in the optoelectronic properties, especially for M2. This derivative showed a red shift in absorption peak (518.8 nm), lower HOMO-LUMO energy gap (2.25 eV), high light-harvesting efficiency (74.72%), low exciton binding energy (0.28 eV) and suitable kinetics of electron injection/dye regeneration. These findings demonstrate the potential of these modified indigo derivatives as effective, environmentally friendly and low-cost natural sensitizers that correspond to SDG 7 (Affordable and Clean Energy) and SDG 13 (Climate Action).
HIGHLIGHTS
- Novel donor-indigo-acceptor (D-Indigo-A) dye models were designed by introducing amine donor groups and cyanoacrylate acceptors into natural Indigofera tinctoria.
- Density functional theory (DFT) and time-dependent DFT (TDDFT) simulations revealed significant improvements in electronic and optical properties compared to unmodified indigo.
- The M2 derivative exhibited a red-shifted absorption maximum (518.8 nm), a reduced HOMO-LUMO gap (2.25 eV), and the lowest exciton binding energy (0.28 eV), enhancing charge transfer and light harvesting.
- Modified indigo dyes demonstrated favorable free energy of electron injection and regeneration, indicating efficient electron mobility and suppressed recombination.
- This study highlights natural indigo derivatives as promising, eco-friendly, and cost-effective sensitizers for dye-sensitized solar cells, aligning with SDG 7 (Affordable and Clean Energy).
GRAPHICAL ABSTRACT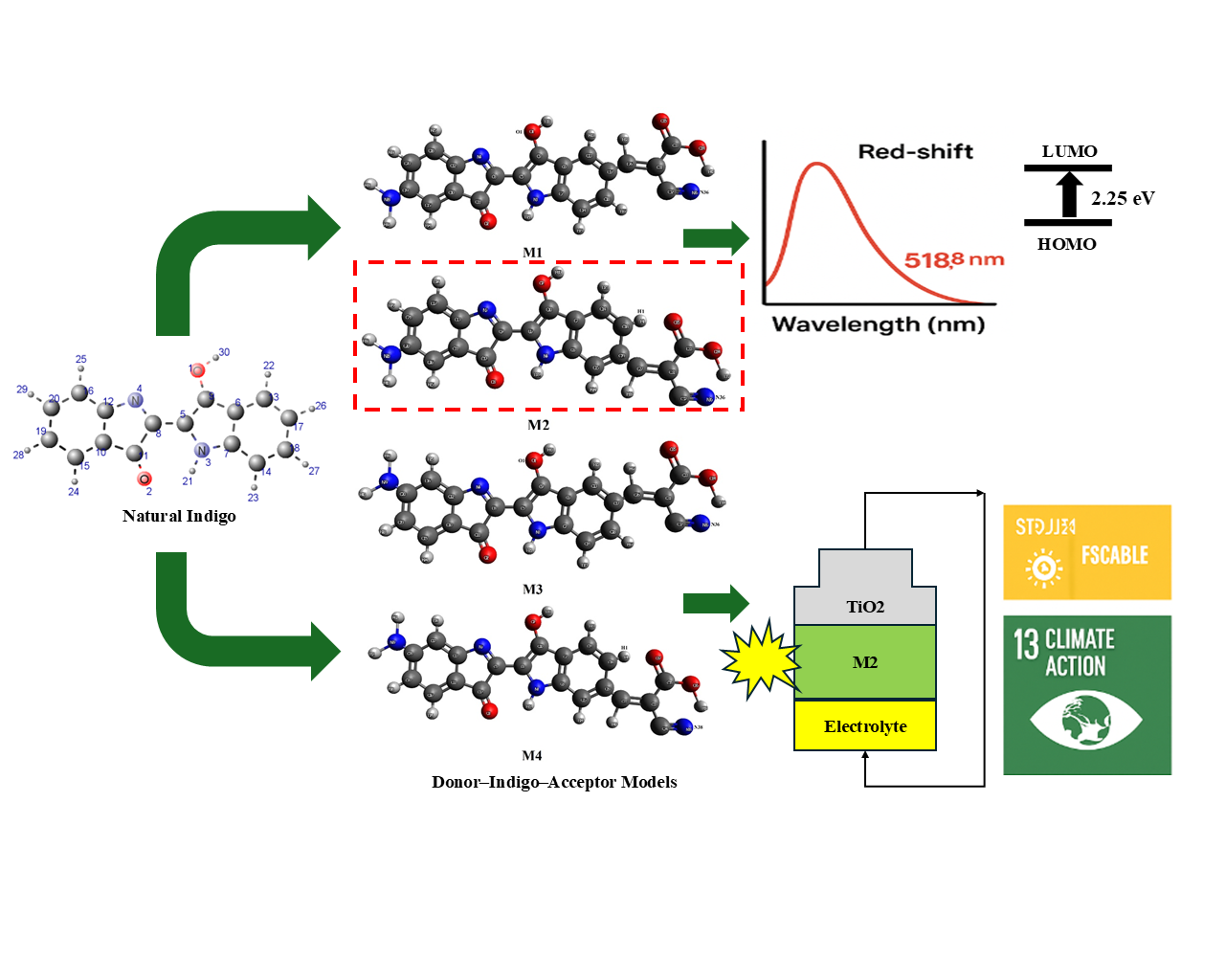
Downloads
References
W Wei, DJ Stacchiola and YH Hu. 3D graphene from CO2 and k as an excellent counter electrode for dye-sensitized solar cells. International Journal of Energy Research 2017; 41(15), 2502-2508.
B O’Regan and M Gratzel, A low-cost, high-efficiency solar cell based on dye -sensitized colloidal TiO2 films. Nature 1991; 353, 737-740.
S Huang, H Wang, Y Zhang, S Wang, Z Chen, Z Hu and X Qian. Prussian blue-derived synthesis of uniform nanoflakes-assembled nis2 hierarchical microspheres as highly efficient electrocatalysts in dye-sensitized solar cells. RSC Advances 2018; 11, 5992-6000.
N Canever, F Hughson, T Macdonald and T Nann. Electrospinning of photocatalytic electrodes for dye-sensitized solar cells. Journal of Visualized Experiments: Jove 2017; 124, 55309
L Kavan, J Yum and M Grätzel. Graphene nanoplatelets outperforming platinum as the electrocatalyst in co-bipyridine-mediated dye-sensitized solar cells. Nano Letters 2011; 11(12), 5501-5506.
L Heiniger, F Giordano, T Moehl and M Grätzel. Mesoporous TiO2 beads offer improved mass transport for cobalt‐based redox couples leading to high efficiency dye‐sensitized solar cells. Advanced Energy Materials 2014; 4(12), 1400168.
TS Senthil, N Muthukumarasamy and M Kang. Zno nanorods based dye sensitized solar cells sensitized using natural dyes extracted from beetroot, rose and strawberry. Bulletin of the Korean Chemical Society 2014; 35(4), 1050-1056.
T Purnomo, A Nayan, M Sayuthi, N Islami and A Rahman. Fabrication of dye sensitized solar cell using cassava leaf extract and red dragon fruit. Transmisi 2023; 19(2), 84-91.
Y Tiandho, P Purwati, M Marlina, M Mahjur and W Kurniawan. Dye sensitized solar cell (dssc) with areca extract as a natural dye sensitized. Konstan-Jurnal Fisika Dan Pendidikan Fisika 2022; 7(1), 18-24.
C Qin, W Wong and L Han. Squaraine dyes for dye‐sensitized solar cells: recent advances and future challenges. Chemistry - An Asian Journal 2013; 8(8), 1706-1719.
HY Chen, CW Lee, SH Chuang, HP Lu, EWG Diau and CY Yeh. Porphyrin‐perylene dyes for dye‐sensitized solar cells. Journal of the Chinese Chemical Society 2010; 57(5B), 1141-1146.
C Cao, T Yang and G Chen. Hibiscus leachate dye-based low-cost and flexible dye-sensitized solar cell prepared by screen printing. Materials 2021; 14(11), 2748.
E Barea, R Caballero, L López‐Arroyo, A Guerrero, P Cruz and F Langa. Triplication of the photocurrent in dye solar cells by increasing the elongation of the π‐conjugation in Zn‐porphyrin sensitizers. ChemPhysChem 2011; 12(5), 961-965.
P Kumar, K Sakthivel and V Balasubramanian. Microwave assisted biosynthesis of rice shaped ZnO nanoparticles using amorphophallus konjac tuber extract and its application in dye sensitized solar cells. Materials Science-Poland 2017; 35(1), 111-119.
L Yu, K Fan, T Duan, X Chen, R Li and T Peng. Efficient panchromatic light harvesting with co-sensitization of zinc phthalocyanine and bithiophene-based organic dye for dye-sensitized solar cells. ACS Sustainable Chemistry & Engineering 2014; 2(4), 718-725.
B Ranamagar, I Abiye, H Karki, Y Lan and F Abebe. Performance of rhodamine-sensitized solar cells fabricated with silver nanoparticles. Advances in Nanoparticles 2023; 12(2), 68-79.
N Li, Q Wang, J Zhou, S Li, J Liu and H Chen. Insight into the progress on natural dyes: sources, structural features, health effects, challenges, and potential. Molecules 2022; 27(10), 3291.
M Al-Fatimi. Traditional knowledge of wild plants on traditional tools, materials, products and economic practices in southern yemen. Journal of Ethnobiology and Ethnomedicine 2024; 20(1), 62.
L Zhang, L Wang, A Cunningham, Y Shi and Y Wang. Island blues: Indigenous knowledge of indigo-yielding plant species used by hainan miao and li dyers on Hainan Island, China. Journal of Ethnobiology and Ethnomedicine 2019; 15(1), 31.
R Agustarini, Y Heryati, Y Adalina, W Adinugroho, D Yuniati and R Fambayune. The development of Indigofera spp. as a source of natural dyes to increase community incomes on Timor Island, Indonesia. Economies 2022; 10(2), 49.
C Wu, M Chung, H Tsai, C Tan, S Chen and C Chang. Fluorene‐containing organic photosensitizers for dye‐sensitized solar cells. ChemPlusChem 2012; 77(9), 832-843.
I Imelda, H Aziz, A Santoni and U Nofitri. The modification of cyanidin based dyes to improve the performance of dye-sensitized solar cells (dsscs). Rasayan Journal of Chemistry 2020; 13(01), 121-130.
C Lopez, A Manova, C Hoppe, M Dreja, P Schmiedel and M Jobe. Combined UV-vis-absorbance and reflectance spectroscopy study of dye transfer kinetics in aqueous mixtures of surfactants. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2018; 550, 74-81.
S Dindorkar and A Yadav. Insights from density functional theory on the feasibility of modified reactive dyes as dye sensitizers in dye-sensitized solar cell applications. Solar 2022; 2(1), 12-31.
H Liu, L Liu, Y Fu, E Liu and B Xue. Theoretical design of d-π-a-a sensitizers with narrow band gap and broad spectral response based on boron dipyrromethene for dye-sensitized solar cells. Journal of Chemical Information and Modeling 2019; 59(5), 2248-2256.
G Bodedla, K Thomas, M Fan and K Ho. Effect of donors on photophysical, electrochemical and photovoltaic properties of benzimidazole‐branched dyes. ChemistrySelect 2017; 2(9), 2807-2814.
C Sakong, HJ Kim, SH Kim, JW Namgoong, JH Park, JH Ryu, B Kim, MJ Ko and JP Kim. Synthesis and applications of new triphenylamine dyes with donor–donor–(bridge)–acceptor structure for organic dye-sensitized solar cells. New Journal of Chemistry 2012; 36(10), 2025-2032.
A Kargeti, R Rohilla, A Raghav, A Bheemaraju and T Rasheed. Effect of extended conjugation on the photosensitizers for dssc: DFT and TD-DFT study. International Journal of Convergence in Healthcare 2023; 3(2), 1-7.
Y Zhang, H Cheema, L McNamara, LA Hunt, NI Hammer and JH Delcamp. Ullazine donor-π bridge-acceptor organic dyes for dye-sensitized solar cells. Chemistry - A European Journal 2018; 24(22), 5939-5949.
L Zhu, H Yang, C Zhong and CM Li. Modified triphenylamine‐dicyanovinyl‐based donor-acceptor dyes with enhanced power conversion efficiency of p‐type dye‐sensitized solar cells. Chemistry, An Asian Journal 2012; 7(12), 2791-2795.
E Özelci, M Aas, A Jonáš and A Kıraz. Optofluidic fret microlasers based on surface-supported liquid microdroplets. Laser Physics Letters 2014; 11(4), 045802.
T Ilahi, T Izhar, M Ali, U Noor, M Siddique, EM Khan, R Yousaf, A Ali and B Khan. Comprehensive design analysis of economical e-bike charger with IoT-empowered system for real-time parameter monitoring. Journal of Advanced Transportation 2024; 2024(1), 2387983.
N Mansouri, A Mnassri, S Nasri, M Ali, A Lashab, JC Vasquez and JM Guerrero. Control and optimization of hydrogen hybrid electric vehicles using GPS-based speed estimation. Electronics 2025; 14(1), 110.
A Gulraiz, SSH Zaidi, M Ashraf, M Ali, A Lashab, JM Guerrero and B Khan. Impact of photovoltaic ingress on the performance and stability of low voltage grid-connected microgrids. Results in Engineering 2025; 26, 105030.
A Gulraiz, SSH Zaidi, H Gulraiz, BM Khan, M Ali and B Khan. Leveraging dense layer hybrid graph neural networks for managing overvoltage in PV-dominated distribution systems. Results in Engineering 2025; 27, 106169.
M Ali, Y Guan, JC Vasquez, JM Guerrero, FD Wijaya and AP Perdana. Microgrids for energy access in remote and islanded communities under natural disasters - Context of Lombok Island Indonesia. Renewable Energy Focus 2025; 54, 100705.
Y Sekkat, A Fitri, O Britel, AT Benjelloun, M Mcharfi and M Benzakour. Dft/tddft investigation of the effect of electron acceptor groups on photovoltaic performance using phenothiazine‐based d‐ai‐π‐a dyes for dssc applications. ChemistrySelect 2024; 9(36), e202403042.
G Selopal, H Wu, J Lu, Y Chang, M Wang and A Vomiero. Metal-free organic dyes for tio2 and zno dye-sensitized solar cells. Scientific Reports 2016; 6(1), 18756.
KB Ingole, SS Deshmukh, TS Verma, S Krishnamurty, K Krishnamoorthy and J Nithyanandhan. Triazatruxene amine donor-based visible-light-responsive unsymmetrical squaraine dyes for dye-sensitized solar cells. ACS Applied Energy Materials 2024; 7(18), 7982-7991.
P Pierrat, C Cebrián, M Beley, PC Gros, I Torres‐Moya, P Prieto and S Hesse. Synthesis and properties of new multiple tcne adducts from dialkynyl‐n‐(het)arylpyrroles, European Journal of Organic Chemistry 2019; 2019(27), 4341-4348.
M AM Al‐Alwani, ABSA Al‐Mashaan and MF Abdullah. Performance of the dye‐sensitized solar cells fabricated using natural dyes from ixora coccinea flowers and cymbopogon schoenanthus leaves as sensitizers. International Journal of Energy Research 2019; 43(13), 7229-7239.
Y Yuan and C Wan. Dual application of waste grape skin for photosensitizers and counter electrodes of dye-sensitized solar cells. Nanomaterials 2022; 12(3), 563.
YS Yen, JS Ni, WI Hung, CY Hsu, H Chou and J Lin. Naphtho[2,3-c][1,2,5]thiadiazole and 2h-naphtho[2,3-d][1,2,3]triazole-containing d–a−π–a conjugated organic dyes for dye-sensitized solar cells. ACS Applied Materials & Interfaces 2016; 8(9), 6117-6126.
K Galappaththi, A Lim, P Ekanayake and MI Petra. Cyanidin-based novel organic sensitizer for efficient dye-sensitized solar cells: DFT/TDDFT study. International Journal of Photoenergy 2017; 2017(1), 8564293.
S Zimosz, A Slodek, P Gnida, A Glinka, M Ziółek, D Zych, AK Pająk, M Vasylieva and E Schab-Balcerzak. New d−π–d−π–a systems based on phenothiazine derivatives with imidazole structures for photovoltaics. The Journal of Physical Chemistry C 2022; 126(21), 8986-8999.
JM Cole, MA Blood-Forsythe, TC Lin, P Pattison, Y Gong and Á Vázquez‐Mayagoitia. Discovery of S···C≡N intramolecular bonding in a thiophenylcyanoacrylate-based dye: Realizing charge transfer pathways and dye···TiO2 anchoring characteristics for dye-sensitized solar cells. ACS Applied Materials & Interfaces 2017; 9(31), 25952-25961.
S Fantacci and F Angelis. Ab initio modeling of solar cell dye sensitizers: The hunt for red photons continues. European Journal of Inorganic Chemi.stry 2019; 2019(6), 743-750.
JK Roy, S Kar and J Leszczynski. Electronic structure and optical properties of designed photo-efficient indoline-based dye-sensitizers with d-a-π-a framework. The Journal of Physical Chemistry C 2019; 123(6), 3309-3320.
Z Yang, D Wang, X Bai, C Shao and D Cao. Designing triphenylamine derivative dyes for highly effective dye-sensitized solar cells with near-infrared light harvesting up to 1100 nm. RSC Advances 2014; 4(89), 48750-48757.
KB Ørnsø, CS Pedersen, JM Garcia-Lastra and KS Thygesen. Optimizing porphyrins for dye sensitized solar cells using large-scale ab initio calculations. Physical Chemistry Chemical Physics 2014; 16(30), 16246-16254.
K Ørnsø, J García‐Lastra and K Thygesen. Computational screening of functionalized zinc porphyrins for dye sensitized solar cells. Physical Chemistry Chemical Physics 2013; 15(44), 19478-19486.
H Ünal, D Gunceler and E Mete. A study of the density functional methods on the photoabsorption of bodipy dyes. Physical Chemistry Chemical Physics 2014; 278, 14-18.
L Yang, L Guo, Q Chen, H Sun, H Yan, Q Zeng, X Zhang, X Pan and S Dai. Substituent effects on zinc phthalocyanine derivatives: A theoretical calculation and screening of sensitizer candidates for dye-sensitized solar cells. Journal of Molecular Graphics and Modelling 2012; 38, 82-89.
NN Ghosh, A Chakraborty, S Pal, A Pramanik and P Sarkar. Modulating triphenylamine-based organic dyes for their potential application in dye-sensitized solar cells: A first principle theoretical study. Physical Chemistry Chemical Physics 2014; 16(46), 25280-25287.
E Nabil, AA Hasanein, RB Alnoman and M Zakaria. Optimizing the cosensitization effect of sq02 dye on bp-2 dye-sensitized solar cells: A computational quantum chemical study. Journal of Chemical Information and Modeling 2021; 61(10), 5098-5116.
F Cervantes-Navarro and D Glossman-Mitnik. Density functional theory study of indigo and its derivatives as photosensitizers for dye-sensitized solar cells. Journal of Photochemistry and Photobiology A: Chemistry 2013; 255, 24-26.
Basuki, Suyitno and B Kristiawan. Absorbance and electrochemical properties of natural indigo dye. AIP Conference Proceedings 2018; 1931(1), 030067.
MF Maahury, MF Maahury, YT Male, MA Martoprawiro, N Science and N Science. DFT study of leuco-indigo and indigo as active material in dyes-sensitized solar cell. Molekul 2020; 15(2), 114-120.
Z Ju, J Sun and Y Liu. Molecular structures and spectral properties of natural indigo and indirubin: Experimental and DFT studies. Molecules 2019; 24(21), 3831.
N Jiwalak, R Daengngern, T Rungrotmongkol, S Jungsuttiwong, S Namuangruk, N Kungwan and S Dokmaisrijan. A spectroscopic study of indigo dye in aqueous solution: A combined experimental and TD-DFT study. Journal of Luminescence 2018; 204, 568-572.
AK Rajan and L Cindrella. Studies on new natural dye sensitizers from Indigofera tinctoria in dye-sensitized solar cells. Optical Materials 2019; 88, 39-47.
P Venkatachalam, NG Joby and N Krishnakumar. Enhanced photovoltaic characterization and charge transport of TIO2 nanoparticles/nanotubes composite photoanode based on indigo carmine dye-sensitized solar cells. Journal of Sol-Gel Science and Technology 2013; 67(3), 618-628.
D Franchi, M Calamante, C Coppola, A Mordini, G Reginato, A Sinicropi and L Zani. Synthesis and characterization of new organic dyes containing the indigo core. Molecules 2020; 25(15), 3377.
S Suyitno, B Basuk and A Fery. Electricity generation and stability of dye-sensitized solar cells (DSSCs) employing indigofera as sensitizers. AIP Conference Proceedings 2024; 3124(1), 070020.
S Sreeja and B Pesala. Performance enhancement of betanin solar cells co-sensitized with indigo and lawsone: A comparative study. ACS Omega 2019; 4(19), 18023-18034.
F Rastegari, E Tohidinejad and G Mohammadi-Nejad. Effect of drought stress on indigo and seed yield of Indigofera tinctoria ecotypes. The Indian Journal of Agricultural Sciences 2020; 90(6), 1063-1067.
Y Selim and A Mohamed. Role of dyestuff in improving dye-sensitized solar cell performance. Renewable Energy and Sustainable Development 2017; 3(1), 79-82.
F Neese. Software update: The ORCA program system - Version 5.0. Wiley Interdisciplinary Reviews: Computational Molecular Science2022; 12(5), e1606.
FF Neese, F Wennmohs, U Becker and C Riplinger. The ORCA quantum chemistry program package. The Journal of Chemical Physics 2020; 152(22), L224108.
C Lee, W Yang and RG Parr. Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Physical Review B 1988; 37(2), 785.
AD Becke. Density-functional thermochemistry. III. The role of exact exchange. The Journal of Chemical Physics1993; 98(7), 5648-5652.
SH Vosko, L Wilk and M Nusair. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis. Canadian Journal of Physics 1980; 58(8), 1200-1211.
PJ Stephens, FJ Devlin, CF Chabalowski and MJ Frisch. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. Journal of Physical Chemistry 1994; 98, 11623-11627.
F Weigend and R Ahlrichs. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Physical Chemistry Chemical Physics 2005; 7(18), 3297-3305.
T Yanai, DP Tew and NC Handy. A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chemical Physics Letters 2004; 393(1), 51-57.
Y Tawada, T Tsuneda, S Yanagisawa, T Yanai and K Hirao. A long-range-corrected time-dependent density functional theory. The Journal of Chemical Physics 2004; 120(18), 8425-8433.
JD Chai and M Head-Gordon. Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Physical Chemistry Chemical Physics 2008; 10, 6615-6620.
F Weigend. Accurate coulomb-fitting basis sets for H to Rn. Physical Chemistry Chemical Physics 2006; 8, 1057-1065.
AV Marenich, CJ Cramer and DG Truhlar. Universal solvation model based on solute electron density and on a continuum model of the solvent. The Journal of Physical Chemistry B 2009; 113, 6378.
ED Glendening, JK Badenhoop, AE Reed, JE Carpenter, JA Bohmann, CM Morales and F Weinhold. NBO 7.0. madison, WI: Theoretical chemistry institute. University of Wisconsin, Wisconsin, United States, 2018.
ChemAxon Software, Available at: https://www.chemaxon.com, accessed May 2025.
Syafri, F Ahmad, A Wibawa Sakti, PA Putro, A Tinambunan and H Alatas. Effects of fused thiophene Π-bridge on the electronic and optical properties of modified theaflavin natural dye. Molecular Simulation 2024; 50(5), 367-378.
JM Juma, HVS Ali and NS Babu. TD-DFT investigations on optoelectronic properties of fluorescein dye derivatives in dye-sensitized solar cells (DSSCs). International Journal of Photoenergy 2019; 2019(1), 4616198.
CR Zhang, ZJ Liu, YH Chen, HS Chen, YZ Wu, WJ Feng and DB Wang. DFT and TD-DFT study on structure and properties of organic dye sensitizer TA-St-CA. Current Applied Physics 2010; 10(1), 77-83.
A Tripathi, A Ganjoo and P Chetti. Influence of internal acceptor and thiophene based π-spacer in D-A-π-A system on photophysical and charge transport properties for efficient DSSCs: A DFT insight. Solar Energy 2020; 209, 194-205.
T Manzoor, S Asmi, S Niaz and AH Pandith. Computational studies on optoelectronic and charge transfer properties of some perylene-based donor-p-acceptor systems for dye sensitized solar cell applications. International Journal of Quantum Chemistry 2017; 117(5), e25332.
MRSA Janjua, MU Khan, M Khalid, N Ullah, R Kalgaonkar, K Alnoaimi, N Baqader and S Jamil. Theoretical and conceptual framework to design efficient dye-sensitized solar cells (DSSCs): Molecular engineering by DFT method. Journal of Cluster Science 2020; 32(2), 243-253.
M Li, L Kou, L Diao, Q Zhang, Z Li, Q Wu, W Lu, D Pan and Z Wei. Theoretical study of WS-9-based organic sensitizers for unusual vis/NIR absorption and highly efficient dye-sensitized solar cells. The Journal of Physical Chemistry C 2015; 119(18), 9782-9790.
R Tarsang, V Promarak, T Sudyoadsuk, S Namuangruk, N Kungwan, P Khongpracha and S Jungsuttiwong. Triple-bond-modified anthracene sensitizers for dye-sensitized solar cell: A computational study. RSC Advances 2015; 5(48), 38130-38140.
AR Obasuyi, D Glossman-Mitnik and N Flores-Holguín. Theoretical modifications of the molecular structure of Aurantinidin and Betanidin dyes to improve their efficiency as dye‑sensitized solar cells. Journal of Computational Electronics 2020; 19(2), 507-515.
AB El-Meligy, N Koga, S Iuchi, K Yoshida, K Hirao, AH Mangood and AM El-Nahas. DFT/TD-DFT calculations of the electronic and optical properties of bis-N,N-dimethylaniline-based dyes for use in dye-sensitized solar cells. Journal of Photochemistry and Photobiology A: Chemistry 2018; 367, 332-346.
S Di Grande, I Ciofini, C Adamo, M Pagliai and G Cardini. Absorption spectra of flexible fluorescent probes by a combined computational approach: molecular dynamics simulations and time-dependent density functional theory. The Journal of Physical Chemistry A 2022; 126(47), 8809-8817.
W Sang-aroon, W Sang-aroon, S Saekow and V Amornkitbamrung. Density functional theory study on the electronic structure of Monascus dyes as photosensitizer for dye-sensitized solar cells. Journal of Photochemistry and Photobiology A: Chemistry 2012; 236, 35-40.
X Li, Y Hu, I Sanchez-Molina, Y Zhou, F Yu, SA Haque, W Wu, J Hua, H Tian and N Robertson. Insight into quinoxaline containing D–π–A dyes for dye-sensitized solar cells with cobalt and iodine based electrolytes: The effect of π-bridge on the HOMO energy level and photovoltaic performance. Journal of Materials Chemistry A 2015; 3(43), 21733-21743.
GC Dos Santos, EF Oliveira, FC Lavarda and LC da Silva-Filho. Designing new quinoline-based organic photosensitizers for dye-sensitized solar cells (DSSC): A theoretical investigation. Journal of Molecular Modeling 2019; 25(3), 75.
FM Mustafa, AA Abdel Khalek, A Mahboob and M Abdel-Latif. Designing efficient metal-free dye-sensitized solar cells: A detailed computational study. Molecules 2023; 28(17), 6177.
FA Yasseen and FA Al-Temimei. Electronic structures and photovoltaic properties of a novel phthalocyanine and titanium dioxide phthalocyanine for dye sensitized-solar cells. GSC Advanced Research and Reviews 2021; 6(3), 107-115.
A Amkassou and H Zgou. New dyes for DSSC containing thienylen-phenylene: A theoretical investigation. Materials Today: Proceedings 2019; 13, 569-578.
C Sun, Y Li, J Han, B Cao, H Yin and Y Shi. Enhanced photoelectrical properties of alizarin-based natural dye via structure modulation. Materials Today: Proceedings 2019; 185, 315-323.
A Islam, H Sugihara and H Arakawa. Molecular design of ruthenium(II) polypyridyl photosensitizers for efficient nanocrystalline TiO2 solar cells. Journal of Photochemistry and Photobiology A: Chemistry 2003; 158(2-3), 131-138.
CR Zhang, L Liu, JW Zhe, NZ Jin, Y Ma, LH Yuan, ML Zhang, YZ Wu, ZJ Liu and HS Chen. The role of the conjugate bridge in electronic structures and related properties of tetrahydroquinoline for dye sensitized solar cells. International Journal of Molecular Sciences 2013; 14, 5461-5481.
A Arunkumar, S Shanavas, R Acevedo and PM Anbarasan. Computational analysis on D–π–A based perylene organic efficient sensitizer in dye-sensitized solar cells. Optical and Quantum Electronics 2020; 52(3), 164.
A Mahmood, M HussainTahir, A Irfan, B Khalid and AG Al‐Sehemi. Computational designing of triphenylamine dyes with broad and red-shifted absorption spectra for dye-sensitized solar cells using multi-thiophene rings in π-spacer. Bulletin of the Korean Chemical Society 2015; 36, 2615-2620.
R Ghiasi, M Manoochehri and R Lavasani. DFT and TD-DFT study of benzene and borazines containing chromophores for DSSC materials. Russian Journal of Inorganic Chemistry 2016; 61(10), 1267-1273.
D Seo, KW Park, J Kim, J Hong and K Kwak. DFT computational investigation of tuning the electron donating ability in metal-free organic dyes featuring a thienylethynyl spacer for dye sensitized solar cells. Computational and Theoretical Chemistry 2016; 1081, 30-37.
A Fitri, AT Benjelloun, M Benzakour, M Mcharfi, M Hamidi and M Bouachrine. Theoretical design of thiazolothiazole-based organic dyes with different electron donors for dye-sensitized solar cells. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2014; 132, 232-238.
Z Demircioğlu, G Kaştaş, ÇA Kaştaş and R Frank. Spectroscopic, XRD, hirshfeld surface and DFT approach (chemical activity, ECT, NBO, FFA, NLO, MEP, NPA & MPA) of (E)-4-bromo-2-[(4-bromophenylimino) methyl]-6-ethoxyphenol. Journal of Molecular Structure 2019; 1191, 129-137.
CK Tai, YJ Chen, HW Chang, PL Yeh and BC Wang. DFT and TD-DFT investigations of metal-free dye sensitizers for solar cells: Effects of electron donors and p-conjugated linker. Computational and theoretical Chemistry 2019; 971, 42-50.
MN Arshad, M Khalid, M Asad, AM Asiri, MM Alotaibi, AAC Braga and A Khan. Donor moieties with D–π–a framing modulated electronic and nonlinear optical properties for non-fullerene-based chromophores. RSC Advances 2022; 12, 4209-4223.
S Isa, N Anhar, N Selamat, N Adib, M Muda and M Ramli. The effect of different remazol dye concentrations and soaking times in dye-sensitized solar cell. Applied Mechanics and Materials 2015; 815, 203-211.
JS Souza, LOM de Andrade and AS Polo. Nanomaterials for solar energy conversion: Dye-sensitized solar cells based on ruthenium (II) tris-heteroleptic compounds or natural dyes. In: F Souza and E Leite (Eds.). Nanoenergy. Springer, Cham, Switzerland, 2018.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






