Dynamic Expression of E2F1 and eEF1A1 During Lipopolysaccharide-Induced Activation of Primary Hepatic Stellate Cells
DOI:
https://doi.org/10.48048/tis.2026.11708Keywords:
E2F1, eEF1A1, HSCs, LPS, Antifibrosis, Activation, E2F1, eEF1A1, HSCs, Liver fibrosis, LPSAbstract
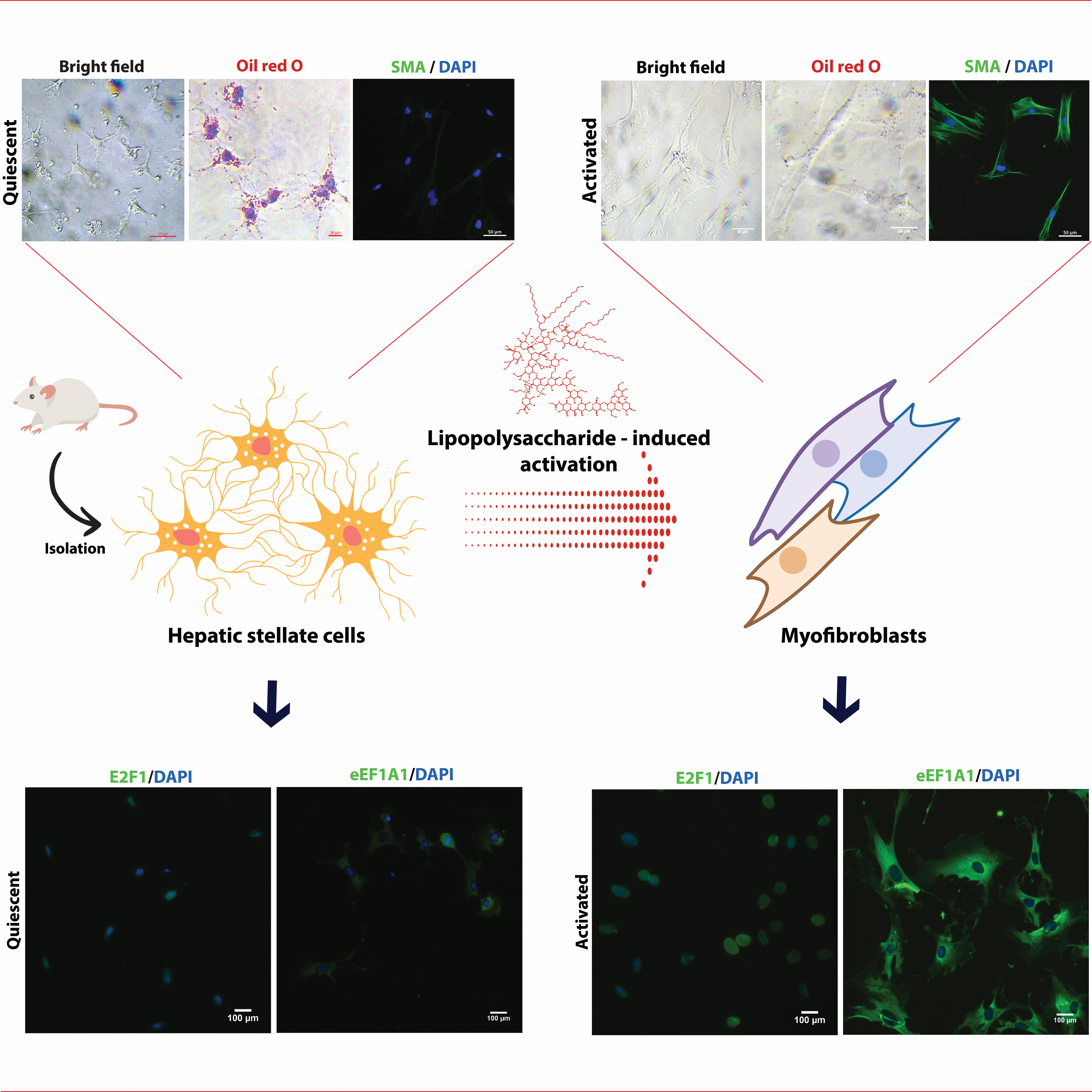
Hepatic stellate cells (HSCs) are the principal mediators of liver fibrosis. Upon liver injury, they undergo activation marked by phenotypic transformation, proliferation, and extracellular matrix deposition. However, the gene regulatory networks driving this activation remain incompletely understood. This study investigates the expression of E2F1 and eEF1A1 - 2 genes involved in cell proliferation and protein synthesis - in HSCs activated by lipopolysaccharide (LPS), an inflammatory stimulant used to model liver injury. Primary HSCs were isolated from Swiss mice and cultured in vitro. Cells were treated with 0, 100 or 200 ng/mL LPS for 24 h and assessed up to 7 days. Morphological changes were assessed microscopically, lipid content was evaluated via Oil Red O staining, and expression of α-SMA, E2F1, and eEF1A1 was measured by immunocytochemistry and qRT-PCR. LPS induced dose- and time-dependent HSC activation, characterized by morphological changes, loss of lipid droplets, and increased α-SMA expression. E2F1 expression increased progressively, peaking at Day 3 and remaining elevated through Day 7. eEF1A1 expression showed a biphasic pattern, with upregulation at Day 3 followed by a reduction at Day 7. Notably, 200 ng/mL LPS yielded the most pronounced gene expression changes. E2F1 and eEF1A1 are dynamically regulated during LPS-induced HSC activation, indicating their potential roles in fibrogenic processes. These findings offer new insights into the transcriptional and translational control of HSC biology and suggest novel targets for antifibrotic therapies.
HIGHLIGHTS
- LPS stimulation established an in vitro model of HSC activation
- E2F1 was progressively upregulated, peaking at Day 3 and sustained to Day 7 of activation
- eEF1A1 showed biphasic expression, with early upregulation (Day 3) followed by decline (Day 7)
- Findings suggest E2F1/eEF1A1 as regulators and targets in liver fibrogenesis
GRAPHICAL ABSTRACT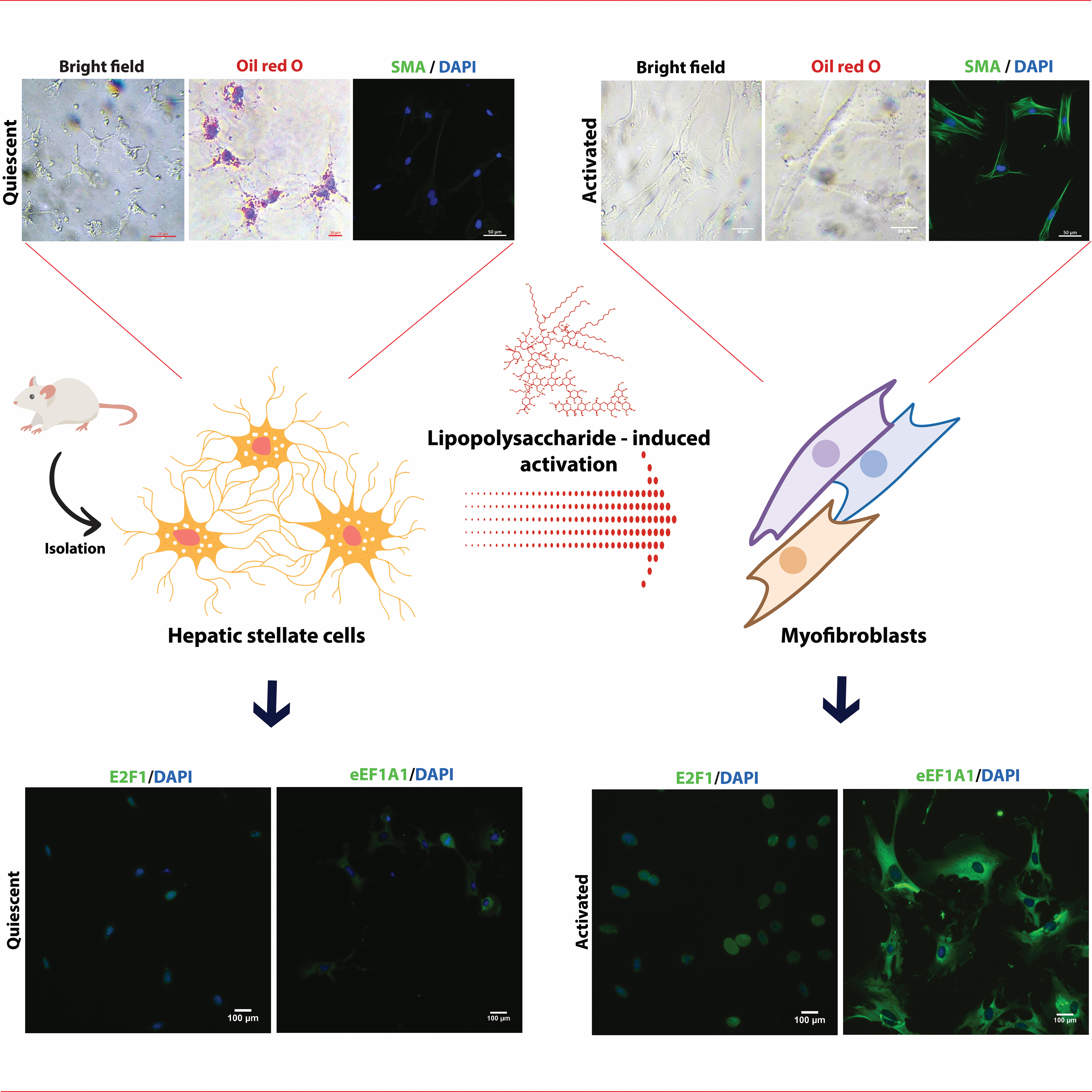
Downloads
References
Y Wang, M Wang, C Liu, M Hao, W Wang and Y Li. Global burden of liver cirrhosis 1990 - 2019 and 20 years forecast: Results from the global burden of disease study 2019. Annals of Medicine 2024; 56(1), 2328521.
EKJ Tham, DJH Tan, P Danpanichkul, CH Ng, N Syn and B Koh. The global burden of cirrhosis and other chronic liver diseases in 2021. Liver International 2025; 45(3), e70001.
HHY Kim, S Sakane, A Eguileor, RCG Weber, W Lee, X Liu, K Lam, K Ishizuka, SB Rosenthal, K Diggle, DA Brenner and T Kisseleva. The origin and fate of liver myofibroblasts. Cellular and Molecular Gastroenterology and Hepatology 2024; 17(1), 93-106.
DR Kamm and KS McCommis. Hepatic stellate cells in physiology and pathology. The Journal of Physiology 2022; 600(8), 1825-1837.
RM Wahid, NH Hassan, W Samy, AA Abdelhadi, SF Saadawy and SF Elsayed. Unraveling the hepatic stellate cells mediated mechanisms in aging’s influence on liver fibrosis. Scientific Reports 2024; 14(1), 13473.
HY Kim, SB Rosenthal, X Liu, C Miciano, X Hou and M Miller. Multi-modal analysis of human hepatic stellate cells identifies novel therapeutic targets for metabolic dysfunction-associated steatotic liver disease. Journal of Hepatolog 2025; 82(5), 882-897.
T Tsuchida and SL Friedman. Mechanisms of hepatic stellate cell activation. Nature Reviews Gastroenterology & Hepatology 2017; 14(7), 397-411.
Z Fang, M Lin, S Chen, H Liu, M Zhu, Y Hu, S Han, Y Wang, L Sun, F Zhu, C Xu and C Gong. E2F1 promotes cell cycle progression by stabilizing spindle fiber in colorectal cancer cells. Cellular & Molecular Biology Letters 2022; 27(1), 90.
A Mills and F Gago. On the need to tell apart fraternal twins eEF1A1 and eEF1A2, and their respective outfits. International Journal of Molecular Sciences 2021; 22(13), 6973.
RB Wilson, AM Kozlov, HH Tehrani, JS Twumasi-Ankrah, YJ Chen and MJ Borrelli. Elongation factor 1A1 regulates metabolic substrate preference in mammalian cells. Journal of Biological Chemistry 2024; 300(3), 105684.
R Farra, B Scaggiante, C Guerra, G Pozzato, M Grassi and F Zanconati. Dissecting the role of the elongation factor 1A isoforms in hepatocellular carcinoma cells by liposome-mediated delivery of siRNAs. International Journal of Pharmaceutics 2017; 525(2), 367-376.
I Mederacke, DH Dapito, S Affo, H Uchinami and RF Schwabe. High-yield and high-purity isolation of hepatic stellate cells from normal and fibrotic mouse livers. Nature Protocols 2015; 10(2), 305-315.
TM Dang, TV Le, HQ Do, VT Nguyen, AXL Holterman and LTT Dang. Optimization of the isolation procedure and culturing conditions for hepatic stellate cells obtained from mouse. Bioscience Reports 2021; 41(1), BSR20202514.
T Le Van, N Ho, T Dang Minh, and T Nhung. Chloroquine inhibiting the activation of primary hepatic stellate cells induced by lipopolysaccharide. Research Journal of Biotechnology 2023; 18(8), 54-61.
RV Modak and DM Zaiss. Isolation and culture of murine hepatic stellate cells. Bio-protocol 2019; 9(21), e3422-e3422.
D Knook, A Seffelaar and AJE De Leeuw. Fat-storing cells of the rat liver: Their isolation and purification. Experimental Cell Research 1982; 139(2), 468-471.
T Kisseleva, S Ganguly, R Murad, A Wang and DA Brenner. Regulation of hepatic stellate cell phenotypes in MASH. Gastroenterology 2025; 139(2), 468-471.
SK Meurer, S Weiskirchen, CG Tag and R Weiskirchen. Isolation, purification, and culture of primary murine hepatic stellate cells: An update. In: R Weiskirchen and SL Friedman (Eds.). Hepatic stellate cells: Methods and protocols. Humana, New York, 2023.
D Wang, B Wan, X Zhang, P Sun, S Lu and C Liu. Nuclear respiratory factor 1 promotes the growth of liver hepatocellular carcinoma cells via E2F1 transcriptional activation. BMC Gastroenterology 2022; 22(1), 198.
Y Yu, D Zhao, K Li, Y Cai, P Xu, R Li, J Li, X Chen, P Chen and G Cui. E2F1 mediated DDX11 transcriptional activation promotes hepatocellular carcinoma progression through PI3K/AKT/mTOR pathway. Cell Death & Disease 2020; 11(4), 1-16.
R Han, X Chen, Y Li, S Zhang, R Li and L Lu. MicroRNA-34a suppresses aggressiveness of hepatocellular carcinoma by modulating E2F1, E2F3, and Caspase-3. Cancer Management and Research 2019; 11, 2963.
Y Zhang, N Xu, J Xu, B Kong, B Copple and GL Guo. E2F1 is a novel fibrogenic gene that regulates cholestatic liver fibrosis through the Egr‐1/SHP/EID1 network. Hepatology 2014; 60(3), 919-930.
J Zhang, Y Zhao, G Pulivendala, Q Zhang, L Rui and J Gao. The non‐canonical ChREBPα activity suppresses the activation of hepatic stellate cells and liver fibrosis by antagonizing TGF‐β‐E2F1 axis. Advanced Science 2025; 12(29), e15032.
D Romaus-Sanjurjo, JM Saikia, HJ Kim, KM Tsai, GQ Le and B Zheng. Overexpressing eukaryotic elongation factor 1 alpha (eEF1A) proteins to promote corticospinal axon repair after injury. Cell Death Discovery 2022; 8(1), 390.
B Negrutskii and A El’Skaya. Eukaryotic translation elongation factor 1α: Structure, expression, functions, and possible role in aminoacyl-tRNA channeling. Progress in Nucleic Acid Research and Molecular Biology 1998; 60, 47-78.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






