Microwave-Assisted Synthesis and Antioxidant Evaluation of α,β-Unsaturated Ketones Incorporating a Pyrano[3,2-g] Chromene-2,6-dione Core via Claisen–Schmidt Condensation
DOI:
https://doi.org/10.48048/tis.2026.11668Keywords:
α,β-unsaturated ketones, Pyranochromones, Pyrano[3,2-g]chromene, Claisen–Schmidt condensation, Microwave-assisted synthesis, Green chemistry, Antioxidant activityAbstract
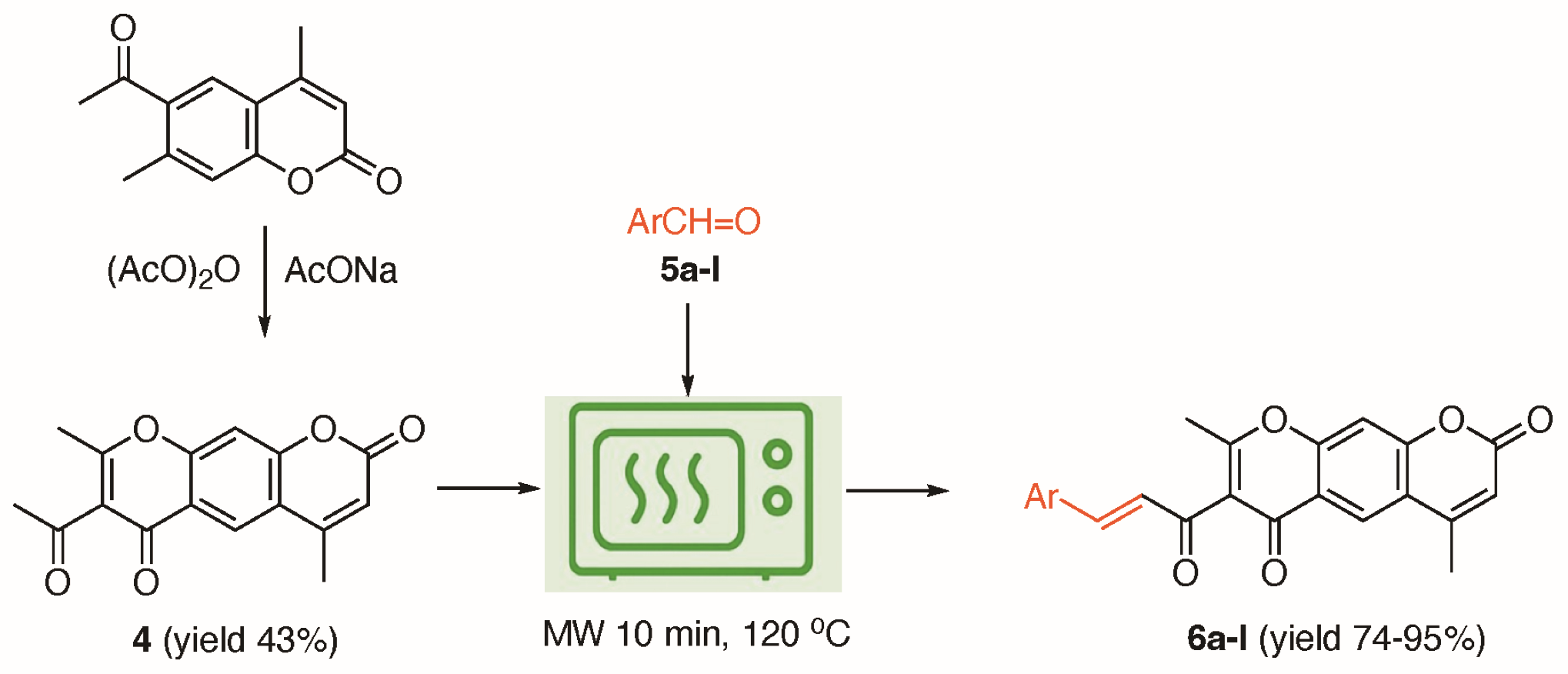
A green, rapid and high-yielding microwave-assisted synthesis of α,β-unsaturated ketones from pyrano[3,2-g]chromene derivatives was developed, followed by preliminary structure–activity relationship (SAR) evaluation. Inspired by the broad pharmacological potential of chromone-based scaffolds, these analogues were designed to enhance antioxidant properties. The key intermediate, 7-acetyl-4,8-dimethyl-2H,6H-pyrano[3,2-g]chromene-2,6-dione, was obtained via a Kostanecki–Robinson reaction and subsequently transformed through Claisen–Schmidt condensations with diverse aromatic aldehydes under microwave irradiation, affording twelve target compounds (6a–6l) in excellent yields (74% - 95%) within minutes. Structural identities were confirmed by spectroscopic and mass analyses. Antioxidant screening using the DPPH assay identified derivative 6 g as the most potent, exhibiting 86.7% radical scavenging at 50 µM, comparable to ascorbic acid (97.65%). While only a DPPH assay was performed, these results provide preliminary antioxidant insights that warrant further biological validation.
HIGHLIGHTS
- An efficient green synthesis affords novel α,β-unsaturated ketones with a pyrano[3,2-g]chromene-2,6-dione core.
- Microwave irradiation dramatically reduces reaction time to 10 min for the key step, delivering high yields (74% - 95%).
- Antioxidant potency is governed by phenolic substituents, with derivative 6g exhibiting strong activity (86.7% inhibition), rivaling ascorbic acid.
- A clear SAR emerges: para-hydroxy groups enhance activity, while ortho-substitution diminishes it due to intramolecular H-bonding.
GRAPHICAL ABSTRACT
Downloads
References
A Gaspar, EMPJ Garrido, F Borges and JMPJ Garrido. Biological and medicinal properties of natural chromones and chromanones. ACS Omega 2024; 9(20), 21706-21726.
LL Mao, Y Liu and JP Wan. An Update on the advances in chromone and the derivatives synthesis based on the key chromone annulation of o-hydroxyaryl enaminones. Chinese Chemical Letters 2024; 36(7), 110784.
M Lucas, M Freitas, AMS Silva, E Fernandes and D Ribeiro. Styrylchromones: Biological activities and structure-activity relationship. Oxidative Medicine and Cellular Longevity 2021; 2021, 2804521.
RS Ipe, S Kumar, F Benny, J Jayan, A Manoharan, ST Sudevan, G George, P Gahtori, H Kim and B Mathew. A concise review of the recent structural explorations of chromones as MAO-B inhibitors: Update from 2017 to 2023. Pharmaceuticals 2023; 16(9), 1310.
A Sobha, A Ganapathy, S Mohan, N Madhusoodanan, AD Babysulochana, K Alaganandan and SB Somappa. Novel small molecule-based Acetylcholinesterase (AChE) inhibitors: From biological perspective to recent developments. European Journal of Medicinal Chemistry Reports 2024; 12, 100237.
M Asim, MK Jastrzębski and AA Kaczor. Dual inhibitors of acetylcholinesterase and monoamine Oxidase-B for the treatment of Alzheimer’s disease. Molecules 2025; 30(14), 2975.
KM Khan, A Ahmad, N Ambreen, A Amyn, S Perveen, SA Khan and MI Choudhary. Design, synthesis and biological evaluation of pyrazole derivatives as anticancer agents. Letters in Drug Design & Discovery 2009; 6, 363-373.
GP Ellis. Chromenes, Chromanones, and Chromones: The chemistry of heterocyclic compounds. John Wiley & Sons, New York, 1977, p. 495-555.
W Baker. Molecular rearrangement of some o-acyloxyacetophenones and the mechanism of the production of 3-acylchromones. Journal of the Chemical Society 1933; 1933, 1381-1389.
S Tummanapalli, SK Punna, KC Gulipalli, S Endoori, S Bodige, AK Pommidi, S Medaboina, S Choppadandi, R Boya, VK Ganapathi, DY Mamindla, R Konakalla, GK Bodala, MR Bakangari, SD Kottam, D Jarikote, T Potewar and M Valluri. One-Pot Synthesis of Chromone-2-carboxylate Scaffold: An important pharmacophore with diverse biological properties. The Journal of Organic Chemistry 2023; 88(13), 8387-8399.
M Wang, L Cheng, J Ma, W Lu and J Wang. Base-Promoted difunctionalization of alkynes: One-Pot synthesis of polysubstituted chromones. European Journal of Organic Chemistry 2023; 26(32), e202300456.
MK Sharma, B Tiwaria and N Hussain. Pd-Catalyzed stereoselective synthesis of chromone C-Glycosides. Chemical Communications 2024; 60(36), 4838-4841.
Y Chen, T Deng, S Zhu, F Yin and H Zhu. Divergent synthesis of chromones and chromanones from diketones using an AgOTf/[Si]H system by switching hydrosilanes. Tetrahedron 2024; 162, 134087.
X Wang, M Peng, Y Wang, S Song, Y Xu, L Chen and F Yu. Eco-Friendly and efficient synthesis of 2-Hydroxy-3-Hydrazono-Chromones through α,β-C(sp2)–H Bond difunctionalization/chromone annulation reaction of O-Hydroxyaryl Enaminones, Water and Aryldiazonium Salts. Molecules 2025; 30(6), 1194.
S Song, M Peng, Z Zhang, H Hu, Y Wei, SJ Yan, Y Wang and F Yu. Divergent synthesis of 2-Chromonyl-3-hydrazono-chromones and 2-Alkoxy-3-hydrazono-chromones through switchable annulation reactions of O-Hydroxyphenylenaminones with aryldiazonium salts. Organic Letters 2024; 26(23), 4980-4985.
G Tiwari, A Khanna, VK Mishra and R Sagar. Recent developments on microwave-assisted organic synthesis of Nitrogen- and Oxygen-Containing Preferred Heterocyclic Scaffolds. RSC Advances 2023; 13, 32858-32892.
Y Baqi and AH Ismail. Microwave-assisted synthesis of Near-Infrared chalcone dyes: A systematic approach. ACS Omega 2025; 10(7), 7317-7326.
D Dotta, M Gastaldi, A Fin, N Barbero, C Barolo, F Cardano, F Rossi, F Brunelli, G Viscardi, GC Tron and P Quagliotto. Chalcone synthesis by Green Claisen–Schmidt reaction in cationic and nonionic micellar media. The Journal of Organic Chemistry 2025; 90(8), 2915-2926.
A Mondal and C Mukhopadhyay. Solvent-Free microwave reactions towards significant organic transformations: A green approach. Tetrahedron Green Chemistry 2024; 4, 100054.
AA Shahat, P Cos, N Hermans, S Apers, TD Bruyne, L Pieters, DV Berghe and AJ Vlietinck. Anticomplement and antioxidant activities of new acetylated flavonoid glycosides from Centaurium spicatum. Planta Medica 2003; 69(12), 1153-1156.
M Okawa, J Kinjo, T Nohara and M Ono. DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity of flavonoids obtained from some medicinal plants. Biological and Pharmaceutical Bulletin 2001; 24(10), 1202-1205.
MT Nguyen, VV Nguyen, QT Tran, DC Nguyen and TD Dong. The synthesis of some α,β-unsaturated ketones from derivatives of axetyl cumarines. Vietnam Journal of Chemistry 2009; 47(1), 22-27.
NT Nguyen. 2011, Synthesis and transformation of some of α,β-unsaturated ketones containing benzopyrone ring. Ph. D. Dissertation. Hanoi National University of Education, Hanoi, Vietnam.
TN Nguyen, GTT Pham and VT Ngo. Synthesis of some Bis(arylidenes) containing heterocyclic chromones and α-pyronochromones. Arkivoc 2024; 2024(8), 202412244.
VV Kouznetsov and LDVV Méndez. An update on the chemistry of 4H-Pyrans and 4H-Pyran-4-ones. Current Organic Chemistry 2020; 24(17), 1934-1973.
NA Al-Masoudi and YA Al-Soud. Enamine-mediated catalysis in organic synthesis: Recent advances. Journal of Chemical Research 2021; 45(7-8), 562-571.
D Díaz-Oviedo, MA García-Revilla and MA Vázquez. Microwave-assisted organic synthesis: A review of recent advancements and applications. Current Opinion in Green and Sustainable Chemistry 2023; 42, 100822.
D Tan, V Strukil, C Mottillo and T Friščić. Mechanochemical organic synthesis: A journey through time and space. Nature Reviews Chemistry 2023; 7, 51-56.
O Dhanya, P Navya, PVR Mumthas, MS Thasni and CKS Saleem. A review of microwave-assisted chalcone synthesis: Advancements over conventional methods and their pharmacological actions. International Journal of Pharmaceutical Sciences Review and Research 2025; 85(3), 158-165.
FMA Soliman, AF Mohamed, M Mohamed, NTA Dawood and LI Sadik. Microwave-assisted synthesis of novel chalcone derivatives and study of some of their antimicrobial activities. European Journal of Pharmaceutical and Medical Research 2018; 5(9), 1-10.
MR Ahmad, VG Sastry, N Bano and S Anwar. Synthesis of novel chalcone derivatives by conventional and microwave irradiation methods and their pharmacological activities. Arabian Journal of Chemistry 2016; 9, S931-S935.
A Rayar, MSI Veitía and C Ferroud. An efficient and selective microwave-assisted Claisen–Schmidt reaction for the synthesis of functionalized benzalacetones. SpringerPlus 2015; 4, 221.
KM Schaich, X Tian and J Xie. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. Journal of Functional Foods 2015; 14, 111-125.
AM Pisoschi, A Pop, F Iordache, L Stanca, G Predoi and AI Serban. Oxidative stress mitigation by antioxidants - An overview on their chemistry and influences on health status. European Journal of Medicinal Chemistry 2021; 209, 112891.
CC Shang, Y Zhang, C Sun and L Wang. Tactfully improve the antioxidant activity of 2′-hydroxychalcone with the strategy of substituent, solvent and intramolecular hydrogen bond effects. Journal of Molecular Liquids 2022; 362, 119748.
IR Ilyasov, VL Beloborodov, IA Selivanova and RP Terekhov. ABTS/PP decolorization assay of antioxidant capacity reaction pathways. International Journal of Molecular Sciences 2020; 21(3), 1131.
KM Schaich, X Tian and J Xie. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH and ORAC assays. Journal of Functional Foods 2015; 14, 111-125.
NB Sadeer, D Montesano, S Albrizio, G Zengin and MF Mahomoodally. The versatility of antioxidant assays in Food Science and Safety-Chemistry, applications, strengths and limitations. Antioxidants 2020; 9(8), 709.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






