Rapid On-Site Detection of Hepatitis A and Norovirus in Fresh Food Using Duplex RT-RPA and Lateral Flow Assay
DOI:
https://doi.org/10.48048/tis.2026.11645Keywords:
Food safety, Foodborne viruses, Hepatitis A virus, Lateral flow test, Norovirus, Rapid detection, RT-RPA-LFAAbstract
Foodborne viruses, such as hepatitis A virus (HAV) and norovirus (NoV), are major public health threats, highlighting the need for rapid, affordable, and user-friendly detection methods. This study introduces a new method that simplifies food extraction and uses duplex RT-RPA combined with a lateral flow assay (LFA) to detect HAV and NoV simultaneously in fresh foods like fresh shellfish, meats, and produce. The RT-RPA conditions and LFA strip design were optimized to improve sensitivity and accuracy. The method was validated for cross-reactivity, sensitivity, and accuracy through interlaboratory testing. It achieved a detection limit of 104 RNA copies per reaction in purified RNA for both HAV and NoV, with no cross-reactivity observed. Among 200 fresh food samples tested, 76.5% (153/200) were contaminated, and 45% (90/200) showed co-contamination with HAV and NoV. Additionally, blind testing by a non-expert achieved 96% accuracy. Compared to existing molecular methods, the main advantages of RT-RPA over RT-PCR and RT-LAMP are its speed, low-temperature operation, robustness and simplicity. Combining RT-RPA with LFA offers a practical and efficient solution for on-site detection of foodborne viruses, with the potential to enhance food safety and reduce virus transmission.
HIGHLIGHTS
- Duplex RT-RPA-LFA detects HAV and NoV in fresh foods within 2 h.
- High sensitivity with a detection limit of 104 RNA copies per reaction in purified RNA for both viruses.
- Validated with 96% accuracy in blind testing by non-experts.
- 5% of tested foods contaminated; 45% co-contaminated with HAV and NoV.
- Practical, on-site method enhances food safety and reduces virus transmission.
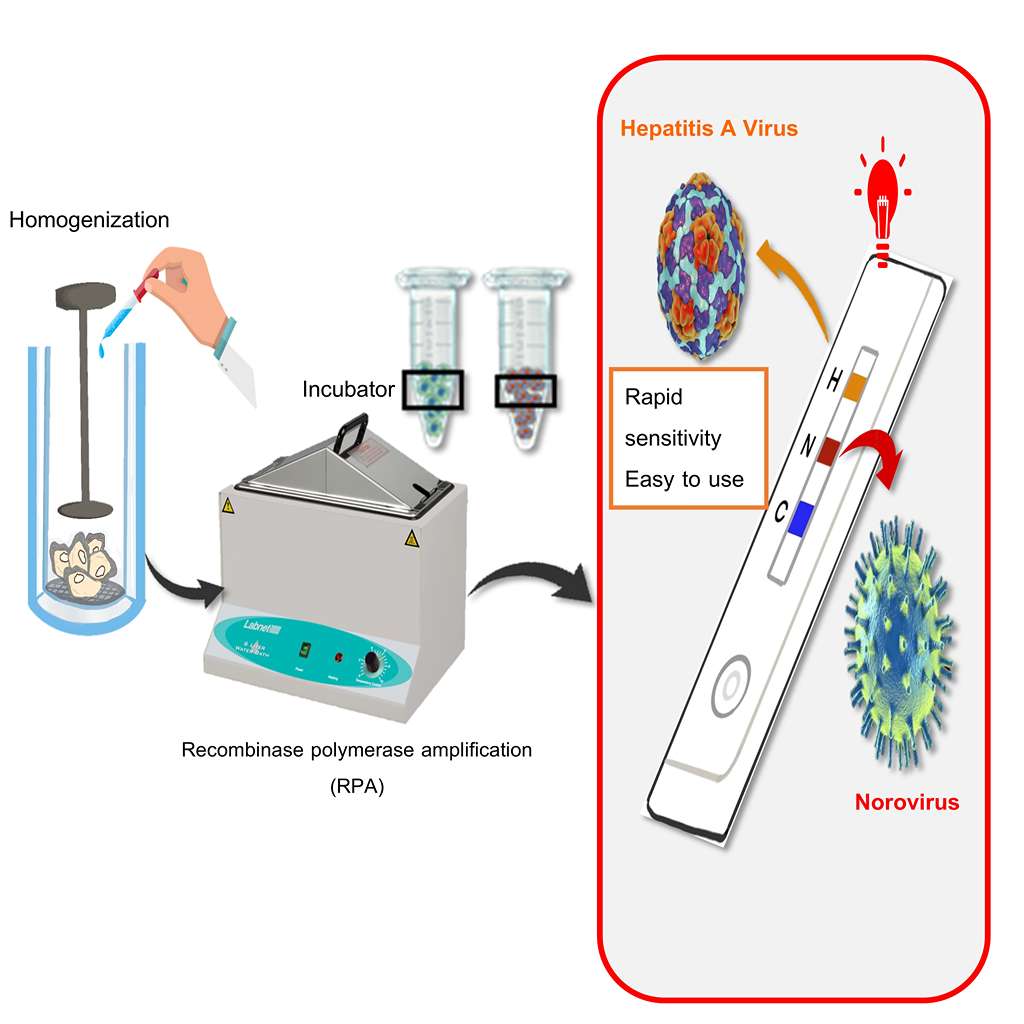
GRAPHICAL ABSTRACT
Downloads
References
Food safety, Available at: https://www.who.int/
news-room/fact-sheets/detail/food-safety, accessed October 2024.
G Cao, W Jing, J Liu and M Liu. The global trends and regional differences in incidence and mortality of hepatitis A from 1990 to 2019 and implications for its prevention. Hepatology International 2021; 15(5), 1068-1082.
KB Carlson, A Dilley, T O’Grady, JA Johnson, B Lopman and E Viscidi. A narrative review of norovirus epidemiology, biology, and challenges to vaccine development. npj Vaccines 2024; 9(1), 94.
X Zhang, C Chen, Y Du, D Yan, D Jiang, X Liu, M Yang, C Ding, L Lan, R Hecht and S Yang. Global burden and trends of norovirus-associated diseases from 1990 to 2019: An observational trend study. Frontiers in Public Health 2022; 10, 905172.
AF Athiyyah, S Wardhani, A Darma, RG Ranuh, D Raharjo, T Shirakawa and SM Sudarmo. The clinical epidemiology of norovirus infection in children with diarrhea at regional public hospital Dr. Soetomo. Jurnal Berkala Epidemiologi 2020; 8(3), 200-207.
M Bellou, P Kokkinos and A Vantarakis. Shellfish-borne viral outbreaks: A systematic review. Food and Environmental Virology 2013; 5(1), 13-23.
H Bernard, M Faber, H Wilking, S Haller, M Höhle, A Schielke, T Ducomble, C Siffczyk, SS Merbecks, G Fricke, O Hamouda, K Stark and D Werber. Large multistate outbreak of norovirus gastroenteritis associated with frozen strawberries, Germany, 2012. Euro Surveillance: Bulletin Europeen Sur Les Maladies Transmissibles 2014; 19(8), 20719.
S Ethelberg, M Lisby, B Bottiger, AC Schultz, A Villif, T Jensen, KE Olsen, F Scheutz, C Kjelso and L Muller. Outbreaks of gastroenteritis linked to lettuce, Denmark, January 2010. Euro Surveillance: Bulletin Europeen Sur Les Maladies Transmissibles 2010; 15(6), 19484.
E Severi, L Verhoef, L Thornton, BR Guzman-Herrador, M Faber, L Sundqvist, R Rimhanen-Finne, AM Roque-Afonso, SL Ngui, F Allerberger, A Baumann-Popczyk, L Muller, K Parmakova, V Alfonsi, L Tavoschi, H Vennema, M Fitzgerald, M Myrmel, M Gertler, J Ederth, M Kontio, C Vanbockstael, S Mandal, M Sadkowska-Todys, ME Tosti, B Schimmer, J O Gorman, K Stene-Johansen, JJ Wenzel, G Jones, K Balogun, AR Ciccaglione, L O’Connor, L Vold, J Takkinen and C Rizzo. Large and prolonged food-borne multistate hepatitis A outbreak in Europe associated with consumption of frozen berries, 2013 to 2014. Euro Surveillance: Bulletin Europeen Sur Les Maladies Transmissibles 2015; 20(29), 21192.
NP Nenonen, C Hannoun, L Svensson, K Torén, LM Andersson, J Westin and T Bergström. Norovirus GII.4 detection in environmental samples from patient rooms during nosocomial outbreaks. Journal of Clinical Microbiology 2014; 52(7), 2352-2358.
HM Wu, M Fornek, KJ Schwab, AR Chapin, K Gibson, E Schwab, C Spencer and K Henning. A norovirus outbreak at a long-term-care facility: The role of environmental surface contamination. Infection Control and Hospital Epidemiology 2005; 26(10), 802-810.
B Priyanka, RK Patil and S Dwarakanath. A review on detection methods used for foodborne pathogens. The Indian Journal of Medical Research 2016; 144(3), 327-338.
J Singh, N Birbian, S Sinha and A Goswami. A critical review on PCR, its types and applications. International journal of advanced research in biological sciences 2014; 1(7), 65-80.
C Suther, S Stoufer, Y Zhou and MD Moore. Recent developments in isothermal amplification methods for the detection of foodborne viruses. Frontiers in Microbiology 2022; 13, 841875.
M Tan, C Liao, L Liang, X Yi, Z Zhou and G Wei. Recent advances in recombinase polymerase amplification: Principle, advantages, disadvantages and applications. Frontiers in Cellular and Infection Microbiology 2022; 12, 1019071.
X Feng, Y Liu, Y Zhao, Z Sun, N Xu, C Zhao and W Xia. Recombinase polymerase amplification-based biosensors for rapid zoonoses screening. International Journal of Nanomedicine 2023; 18, 6311-6331.
K Malcı, LE Walls and L Rios-Solis. Rational design of CRISPR/Cas12a-RPA based 1-pot COVID-19 detection with design of experiments. ACS synthetic biology 2022; 11(4), 1555-1567.
X Wu, S Chen, Z Zhang, Y Zhang, P Li, X Chen, M Liu, Q Lu, Z Li, Z Wei and P Xu. Development of recombinase polymerase amplification combined with lateral flow strips for rapid detection of cowpea mild mottle virus. The Plant Pathology Journal 2023; 39(5), 486-493.
A Sun, L Wang, Y Zhang, X Yang, Y Su and X Wu. Development and application of a duplex RT-RPA assay for the simultaneous detection of cymbidium mosaic virus and odontoglossum ringspot virus. Viruses 2024; 16(4), 543.
P Chutoam, S Jinda, S Lethochavalit and U Intamaso. Development of a reverse transcription recombinase polymerase amplification assay for duplex detection of foodborne viruses in oysters. Natural and Life Sciences Communications 2023; 22(1), e2023008.
Z Crannell, A Castellanos-Gonzalez, G Nair, R Mejia and AC White. Multiplexed recombinase polymerase amplification assay to detect intestinal protozoa. Analytical chemistry 2016; 88(3), 1610-1616.
B Ma, J Li, K Chen, X Yu, C Sun and M Zhang. Multiplex recombinase polymerase amplification assay for the simultaneous detection of 3 foodborne pathogens in seafood. Foods 2020; 9(3), 278.
WA Kibbe. OligoCalc: An online oligonucleotide properties calculator. Nucleic Acids Research 2007; 35, W43-W46.
U Intamaso, P Chutoam, S Jinda and S Lethochavalit. Production of encapsidated RNA particles as a working standard in detecting foodborne viruses in oysters. Natural and Life Sciences Communications 2023; 22(2), e2023034.
S Kennedy. There’s no need to be paranoid about RNA purification, Available at: https://bitesizebio.com/262/the-relaxed-approach-to-rna-purification/2016, accessed August 2025
M Johnson. RNA extraction: Materials and methods, Available at: https://www.labome.com/
method/RNA-Extraction.html, accessed August 2025.
L Weidner, S Laner-Plamberger, D Horner, C Pistorius, J Jurkin, M Karbiener, E Schistal, C Kreil and C Jungbauer. Sample buffer containing guanidine-hydrochloride combines biological safety and RNA preservation for SARS-CoV-2 molecular diagnostics. Diagnostics 2022; 12(5), 1186.
J Dong, W Feng, M Lin, S Chen, X Liu, X Wang and Q Chen. Comparative evaluation of PCR-Based, LAMP and RPA-CRISPR/Cas12a assays for the rapid detection of Diaporthe aspalathi. International Journal of Molecular Sciences 2024; 25(11), 5773.
D Das, CW Lin and HS Chuang. LAMP-based point-of-care biosensors for rapid pathogen detection. Biosensors 2022; 12(12), 1068.
Y Wang, R Chen, X Nie, Z Zhong, C Li, K Li, W Huang, X Fu, J Liu and B Nie. Rapid and sensitive detection of potato virus Y by isothermal reverse transcription-recombinase polymerase amplifica¬tion assay in potato. Molecular and Cellular Probes 2020; 50, 101505.
C Zhao, F Sun, X Li, Y Lan, L Du, T Zhou and Y Zhou. Reverse transcription-recombinase polymerase amplification combined with lateral flow strip for detection of rice black-streaked dwarf virus in plants. Journal of Virological Methods 2019; 263, 96-100.
F Hu, Y Zhang, Y Yang, L Peng, S Cui, Q Ma, F Wang and X Wang. A rapid and ultrasensitive RPA-assisted CRISPR-Cas12a/Cas13a nucleic acid diagnostic platform with a smartphone-based portable device. Biosensors & Bioelectronics 2025; 280, 117428.
FM Munguti, DC Kilalo, HK Yegon, I Macharia, SE Seal, AW Mwango’mbe, EN Nyaboga and G Silva. Real-time reverse transcription recombinase polymerase amplification (RT-RPA) assay for detection of cassava brown streak viruses. Scientific Reports 2024; 14(1), 12438.
C Kolm, RL Mach, R Krska and K Brunner. A rapid DNA lateral flow test for the detection of transgenic maize by isothermal amplification of the 35S promoter. Analytical Methods 2014; 7(1), 129-134.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






