Mineralogical and Geochemical Analysis of Mining and Petroleum Rocks in the Khorat Plateau: Implications for CO₂ Mineralization and CCS
DOI:
https://doi.org/10.48048/tis.2026.11560Keywords:
Carbon capture and storage (CCS), CO₂–rock interaction, Mineral trapping, Khorat Plateau, Geochemistry, Mineralogy, X-ray diffraction (XRD), X-ray fluorescence (XRF), Carbon capture and storage (CCS), CO2-rock interaction, Mineral trapping, Khorat Plateau, Geochemistry, Mineralogy, X-ray diffraction (XRD), X-ray fluorescence (XRF)Abstract
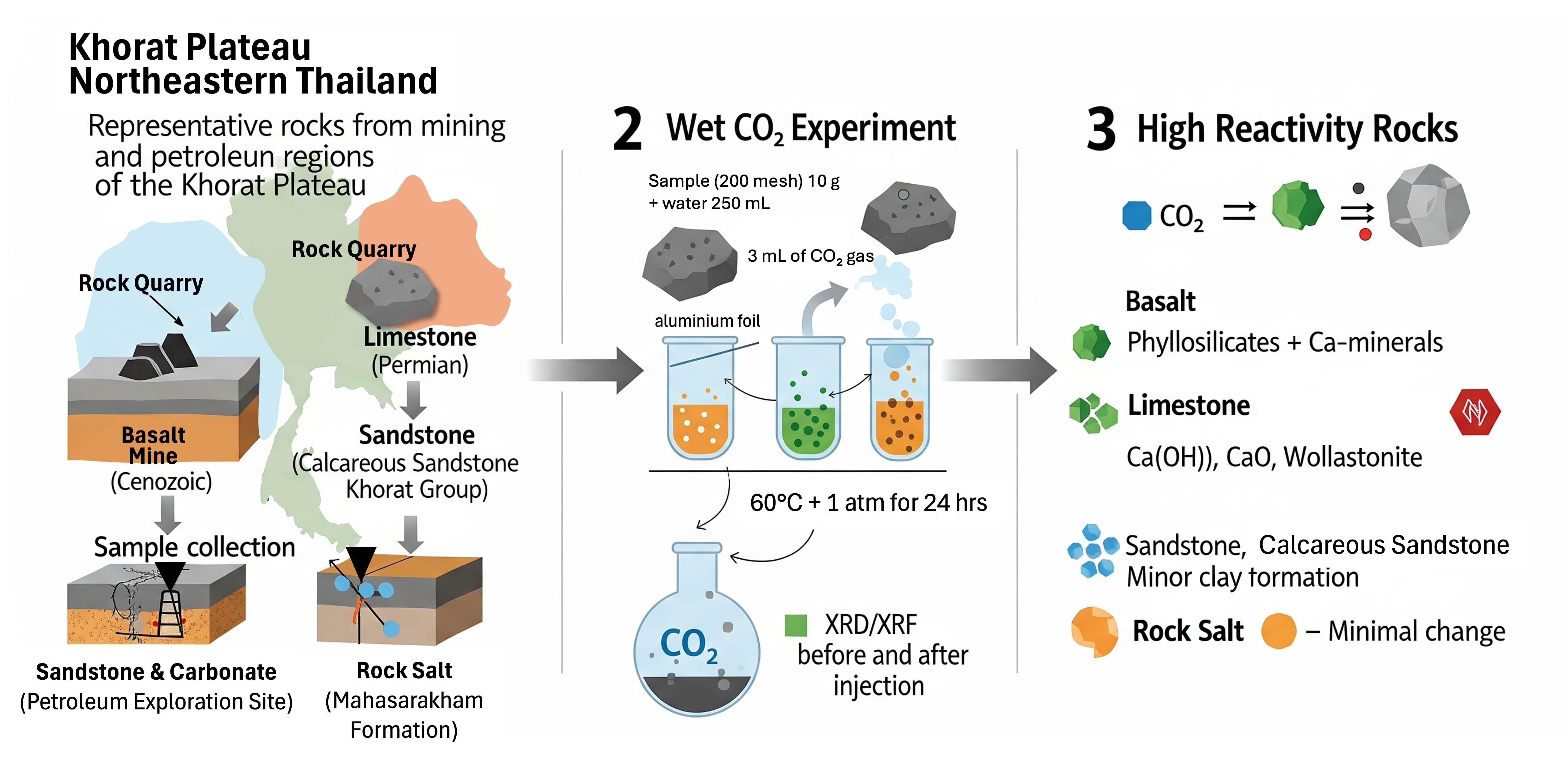
Rising CO2 emissions from energy and industrial sectors underscore the urgent need for effective carbon capture and storage (CCS) strategies, with geological sequestration offering long-term mitigation potential. The Khorat Plateau in northeastern Thailand hosts diverse lithologies - Permian carbonates, Khorat Group sandstones, evaporitic rock salt, and Cenozoic basalts - associated with active mining and petroleum activities. While previous studies have examined CO2 - rock interactions in specific formations such as mudstones of the Triassic Huai Hin Lat Formation, limited data exist on the comparative reactivity of multiple lithologies under wet CO2 conditions. This study evaluates the mineralogical and geochemical responses of representative rock types, including sandstone, calcareous sandstone, limestone, basalt, and rock salt, collected from boreholes and mining sites across the Khorat Plateau. Controlled laboratory experiments involved CO2 exposure in the presence of water, with pre- and post-reaction analyses conducted using X-ray diffraction (XRD) and X-ray fluorescence (XRF). Results show that limestone and basalt exhibit the highest reactivity, forming stable secondary phases such as wollastonite (CaSiO3), portlandite (Ca(OH)2), amorphous CaO, and phyllosilicates, indicating significant potential for mineral trapping. Basalt, in particular, underwent pronounced alteration of primary silicate minerals, while sandstone, calcareous sandstone, and rock salt showed minimal mineralogical changes. These lithology-dependent reactivity patterns highlight basalt and limestone as promising candidates for CCS deployment in the Khorat Plateau, despite earlier reservations regarding basalt suitability. The findings provide the 1st integrated comparison of multiple lithologies from both mining and petroleum contexts in the region, bridging a critical knowledge gap for CCS site selection. By combining experimental mineralogical and geochemical data with regional geological context, this study advances the understanding of CO2 - rock interactions in active industrial and petroleum-bearing settings, offering practical insights for optimizing CCS strategies in Thailand and similar sedimentary basins.
HIGHLIGHTS
- Conducted the 1st mineralogical and geochemical reactivity assessment of Khorat Plateau lithologies under wet CO2
- Basalt-rich formations in the Khorat Plateau exhibit high CO2 reactivity, forming stable carbonates ideal for long-term storage.
- Limestone dissolves rapidly, producing acidic byproducts that may compromise groundwater quality and reservoir stability.
- Feldspathic sandstones provide slower, stable CO2 trapping, whereas calcareous sandstones and halite show limited sequestration potential.
- Lithology, reservoir conditions, and fluid chemistry critically control CCS feasibility in northeastern Thailand.
GRAPHICAL ABSTRACT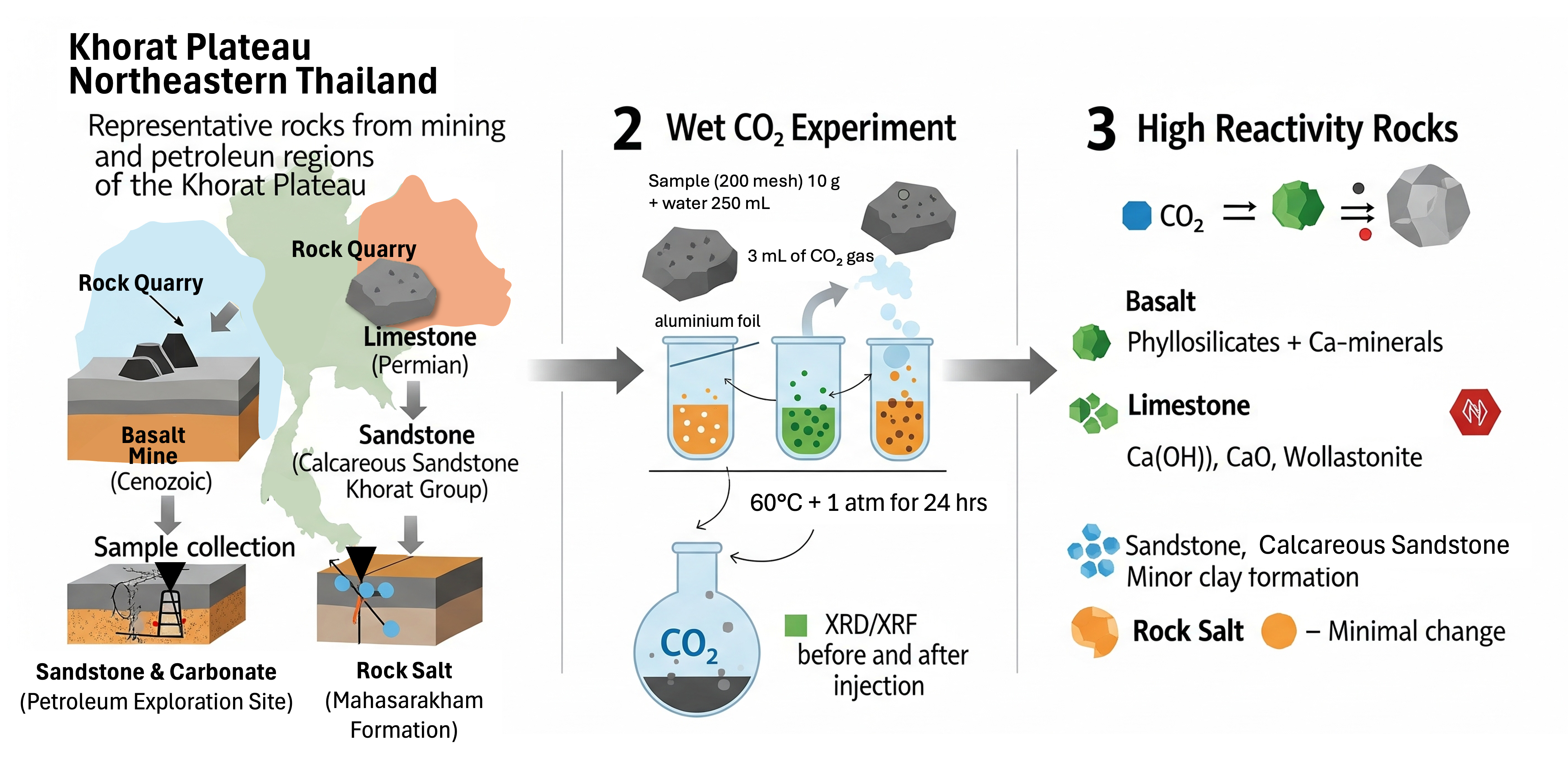
Downloads
References
P Chenrai, S Jitmahantakul, R Bissen and T Assawincharoenkij. A preliminary assessment of geological CO2 storage in the Khorat Plateau, Thailand. Frontiers in Energy Research 2022; 10, 909898.
A Sori and J Moghaddas. A synergistic approach to CO2 sequestration: Evaluating trapping mechanisms in saline aquifers. Chemical Product and Process Modeling 2025; 20(3), 419-443.
V Thanasaksukthawee, T Patthanaporn, N Bangpa, A Suwannathong, N Tippayawong, H Shin and S Tangparitkul. Assessing the geological storage capacity of CO2 in Khorat Sandstone: Geochemistry and fluid flow examinations. International Journal of Greenhouse Gas Control 2025; 141(1), 104322.
N Suwannakarn and PA Salam. Assessment of land-based negative emissions options in Thailand. Carbon Management 2024; 15(1), 2372318.
Department of Primary Industries and Mines (DPIM). Data on the number of operating mining concessions in Northeastern Thailand (in Thai), Available at: https://www1.dpim.go.th, accessed December 2024.
P Chenrai, T Assawincharoenkij, S Jitmahantakul and P Chaiseanwang. Geochemical characteristics of shale gas formation and the potential for carbon storage in Thailand: An example from the Triassic Huai Hin Lat formation. Frontiers in Earth Science 2022; 10, 1085869.
RJ Rosenbauer, B Thomas, JL Bischoff and J Palandri. Carbon sequestration via reaction with basaltic rocks: Geochemical modeling and experimental results. Geochimica et Cosmochimica Acta 2012; 89, 116-133.
SR Gislason and EH Oelkers. Carbon storage in basalt. Science 2014; 344(6182), 373-374.
S O Snaebjörnsdottir, F Wisse, T Fridriksson, H Ármansson, GM Einarsson and SR Gislason. CO2 storage potential of basaltic rocks in Iceland and the oceanic ridges. Energy Procedia 2014; 63(29), 4585-4600.
T Taksavasu, P Arin, T Khatecha and S Kojinok. Microtextural characteristics of ultramafic rock-forming minerals and their effects on carbon sequestration. Minerals 2024; 14(6), 597.
Geological Survey Division. Geological map of Thailand in scale of 1:1,000,000, Available at: https://arcg.is/0Wiv0S, accessed December 2024.
V Singtuen and A Anumart. Characterisation and evaluation of columnar basalt geoheritage in Thailand: Implication for geotourism management in post-quarrying area. Quaestiones Geographicae 2022; 41(1), 37-50.
A Hoffmann and S Siegesmund. The dimension stone potential of Thailand - overview and granite site investigations. Geological Society London Special Publications 2007; 271(1), 43-54.
LJ Shen and N Siritongkham. The characteristics, formation and exploration progress of the potash deposits on the Khorat Plateau, Thailand and Laos, Southeast Asia. China Geology 2020; 3(1), 67-82.
S Jitmahantakul, P Chenrai, T Chaianansutcharit, T Assawincharoenkij, A Tang-on and P Pornkulprasit. Dynamic estimates of pressure and CO2-storage capacity in carbonate reservoirs in a depleted gas field, northeastern Thailand. Case Studies in Chemical and Environmental Engineering 2023; 8(9), 100422.
RL Folk. Practical petrographic classification of limestones. AAPG Bulletin 1959; 43(1), 1-38.
JA Winchester and PA Floyd. Geochemical discrimination of different magma series and their differentiation products using immobile elements. Chemical Geology 1977; 20, 325-343.
M M Herron. Geochemical classification of terrigenous sands and shales from core or log data Journal of Sedimentary Research 1988; 58(5), 820-829.
V Singtuen, S Phajan, A Anumart, B Phajuy, K Srijanta and S Promkotra. Alteration of high alkaline and alkaline basaltic rocks: Parent rocks in the Lava Durian orchard, Sisaket Province, NE Thailand. Heliyon 2022; 7(12), E08619.
V Singtuen and B Phajuy. Geochemistry and alteration of Lampang - Tak Volcanic Rocks, Thailand. Suranaree Journal of Science and Technology 2022; 29(5), 030081-8).
FJ Huertas, S Fiore, F Huertas and J Linares. Experimental study of the hydrothermal formation of kaolinite. Chemical Geology 1999; 156(1-4), 171-190.
N Rodríguez, M Alonso, G Grasa and JC Abanades. Process for capturing CO2 arising from the calcination of the CaCO3 used in cement manufacture. Environmental Science & Technology 2008; 42(18), 6980-6984.
F Amrouche, M Bessaïh and M Mounir. Geochemical reactivity of halite with CO2-rich fluids under simulated subsurface conditions. Geosciences 2023; 13(6), 161.
JM Matter, M Stute, SÓ Snaebjörnsdóttir, EH Oelkers, SR Gislason, ES Aradottir, B Sigfusson, I Gunnarsson, H Sigurdardottir and E Gunnlaugsson. Rapid carbon mineralization for permanent disposal of anthropogenic carbon dioxide emissions. Science 2016; 352(6291), 1312-1314.
BP McGrail, HT Schaef, AM Ho, YJ Chien, JJ Dooley and CL Davidson. Potential for carbon dioxide sequestration in flood basalts. Journal of Geophysical Research 2006; 111(B12), 1-13.
T Taksavasu. Petrographic analysis of mafic and ultramafic rocks in northern Thailand: Implications for CO2 mineralization and enhanced rock weathering approach. Geosciences 2025; 15(3), 89.
J Hartmann, AJ West, P Renforth, P Köhler, CLL Rocha, DA Wolf-Gladrow, HH Dürr and J Scheffran. Enhanced chemical weathering as a geoengineering strategy to reduce atmospheric carbon dioxide, supply nutrients, and mitigate ocean acidification. Reviews of Geophysics 2013; 51(2), 113-149.
A Klemme, T Rixen, M Müller, J Notholt and T Warneke. Destabilization of carbon in tropical peatlands by enhanced weathering. Communications Earth & Environment 2022; 3, 212.
V Vandeginste, C Lim and Y Ji. Exploratory review on environmental aspects of enhanced weathering as a carbon dioxide removal method. Minerals 2024; 14(1), 75.
L Ji. Coupled flow simulation and geomechanical modeling on CO2 storage in a saline aquifer. Earth Sciences 2018; 7(5), 216-226.
OM Ogundipe and EJ Mackay. Modelling halite precipitation during CO2 injection in a heterogeneous reservoir: A North Sea field case. In: Proceedings of the SPE International Conference on Oilfield Chemistry, Galveston, Texas, USA. 2025, p. 15-16.
N Smith, P Boone, A Oguntimehin, G van Essen, R Guo, MA Reynolds, L Friesen, MC Cano and S O’Brien. Quest CCS facility: Halite damage and injectivity remediation in CO2 injection wells. International Journal of Greenhouse Gas Control 2022; 119, 103718.
M Jin, A Ribeiro, E Mackay, L Guimarães and U Bagudu. Geochemical modelling of formation damage risk during CO2 injection in saline aquifers. Journal of Natural Gas Science and Engineering 2016; 35(A), 703-719.
W Xiong, RK Wells, AH Menefee, P Skemer, BR Ellis and DE Giammar. CO2 mineral trapping in fractured basalt. International Journal of Greenhouse Gas Control 2017; 66, 204-217.
D Liu, R Agarwal, Y Li and S Yang. Reactive transport modeling of mineral carbonation in unaltered and altered basalts during CO2 sequestration. International Journal of Greenhouse Gas Control 2019; 85(9), 109-120.
Y Song, S Jun, Y Na, K Kim, Y Jang and J Wang. Geomechanical challenges during geological CO2 storage: A review. Chemical Engineering Journal 2023; 456, 140968.
IPCC. Carbon dioxide capture and storage, Available at: https://www.ipcc.ch/report/carbon-dioxide-capture-and-storage, accessed June 2025.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






