Extraction Optimization of Phenolics from Beta Vulgaris Stems by High-Intensity Ultrasound with Response Surface Methodology Approach
DOI:
https://doi.org/10.48048/tis.2026.11399Keywords:
Beetroot stalk, Box-Behnken design, Soluble phenols, Flavonoids, Anthocyanins, Antioxidant activity, Green extractionAbstract
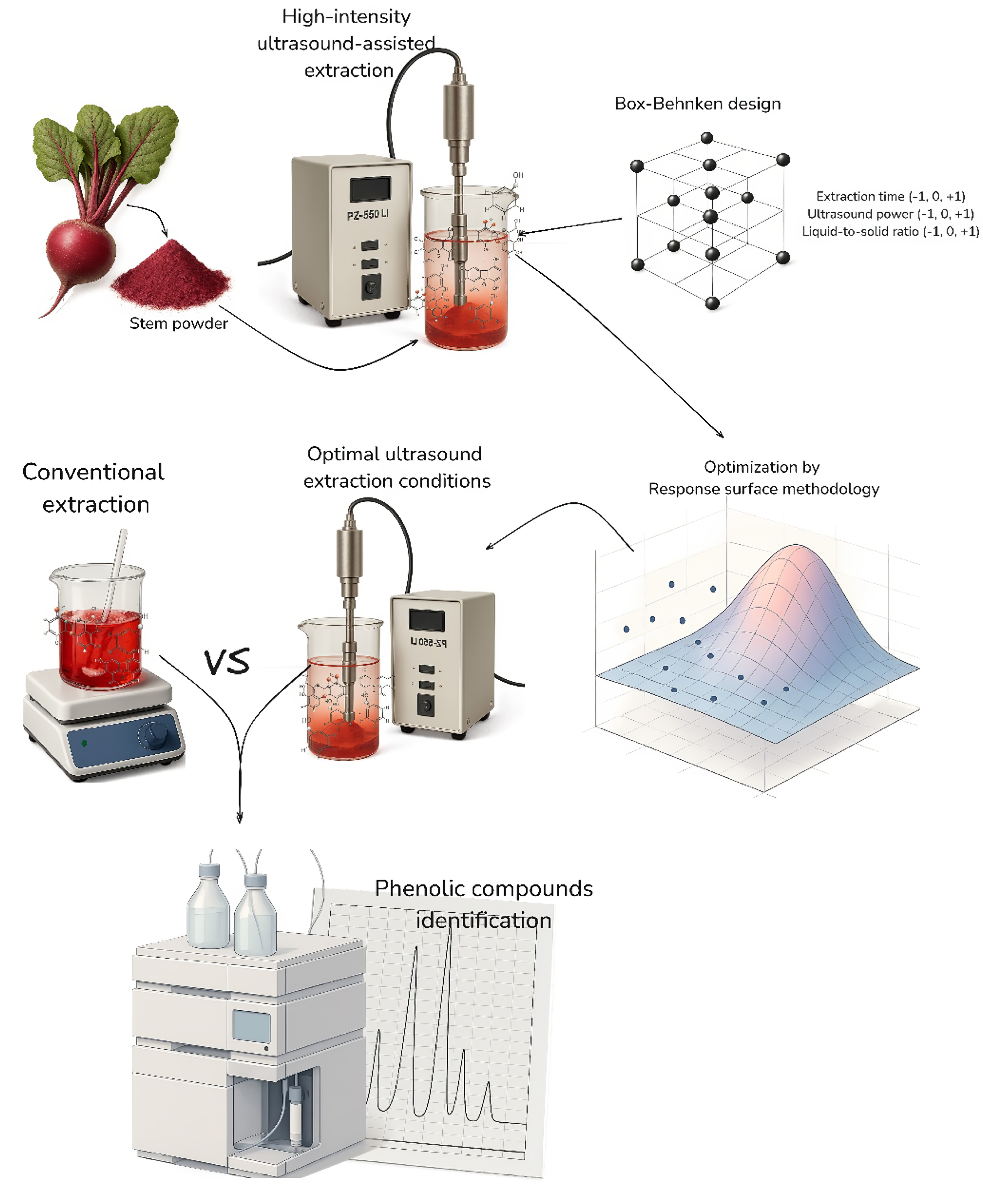
High-intensity ultrasound-assisted extraction was used to extract total soluble phenols, flavonoids, and anthocyanins from Beta vulgaris stems, and to evaluate their antioxidant activities (DPPH, ABTS, and FRAP). The effect of extraction time (X1: 2, 4, and 6 min), ultrasound power (X2: 80%, 90% and 100%), and liquid-to-solid ratio (X3: 10:1, 15:1, and 20:1 mL/g) was investigated using response surface methodology. The high-intensity ultrasound-assisted extraction models for all responses were adjusted to a 2nd-order polynomial equation (R2 = 0.94 - 0.99, lack of fit > 0.05). Optimal high-intensity ultrasound-assisted extraction conditions differ for each response, X1: 3.08 min, X2: 100%, and X3: 17.32:1 mL/g for total soluble phenols, X1: 5.99 min, X2: 95.04% and X3: 17.28:1 mL/g for total flavonoids, and X1: 6 min, X2: 92.24%, and X3: 12.97:1 mL/g for total anthocyanins. Moreover, all evaluated conditions exhibited antioxidant properties by DPPH, ABTS, and FRAP. Furthermore, the validated high-intensity ultrasound-assisted extraction conditions (3.08 min for extraction time, 100% power ultrasound, and 17.32 mL/g of liquid-to-solid ratio) yielded 2.35 times higher soluble phenols content than conventional extraction method (magnetic stirring at 400 rpm for 60 min), with higher (p ˂ 0.05) flavonoids, ABTS, and FRAP values, and similar values (p > 0.05) for anthocyanins and DPPH. Furthermore, shikimic, protocatechuic, 4-hydroxybenzoic, gallic, chlorogenic, neochlorogenic, and trans-ferulic acids were higher under high-intensity ultrasound-assisted extraction than conventional extraction according to the HPLC analysis. It demonstrated that high-intensity ultrasound-assisted extraction is an efficacious technology for extracting bioactive molecules. In addition, future research could focus on isolating and purifying the phenolic compounds extracted from B. vulgaris stem powder, which have potential applications in food and non-food industries.
HIGHLIGHTS
- Beta vulgaris stems are a source of phytochemicals
- Ultrasound is a viable technology for extracting bioactive compounds
- Beta vulgaris extracts exhibited antioxidant properties
GRAPHICAL ABSTRACT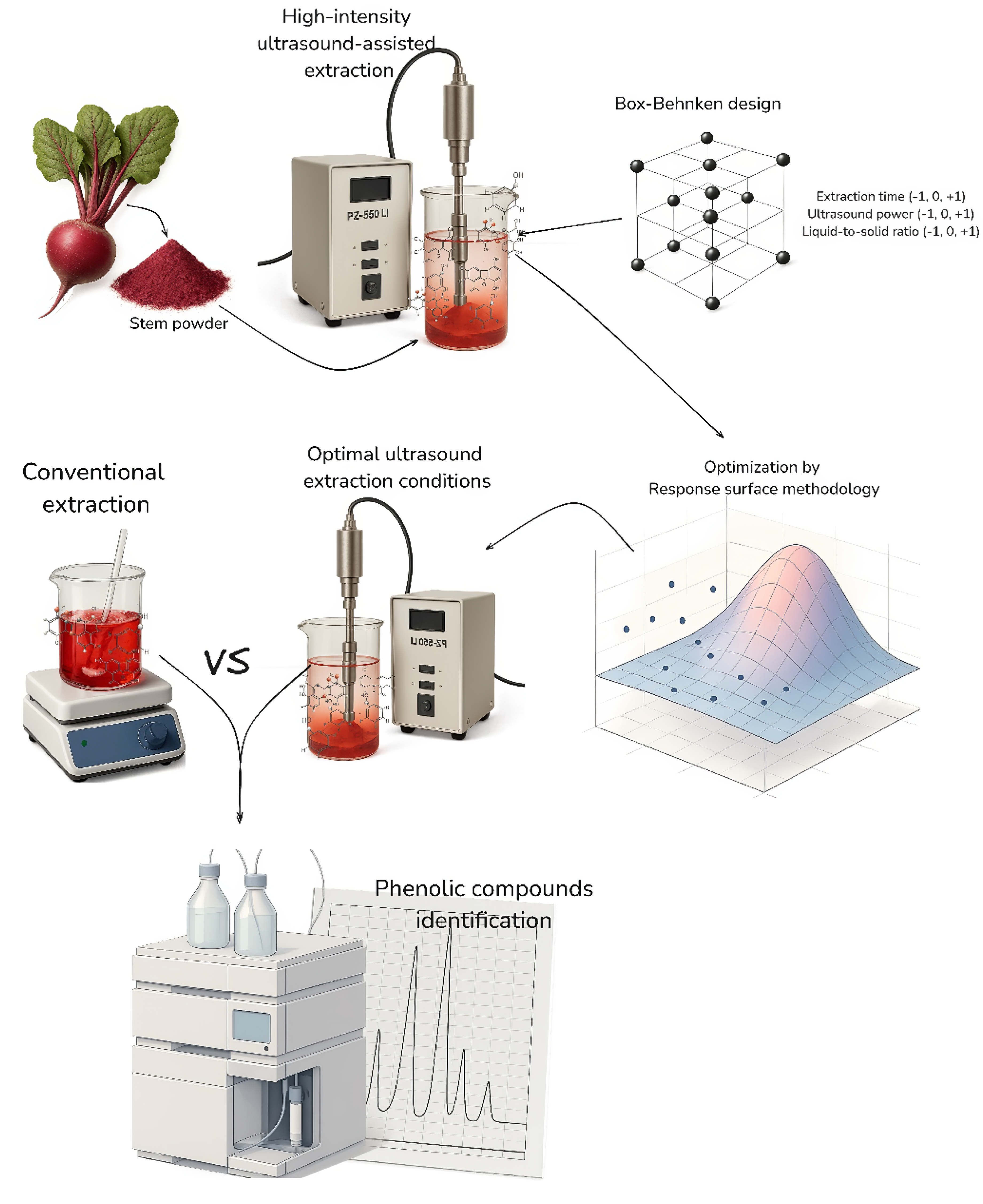
Downloads
References
FAO. Seeking end to loss and waste of food along production chain. 2025, Available at: https://www.fao.org/in-action/seeking-end-to-loss-and-waste-of-food-along-production-chain/en, accessed January 2025.
United Nations. Department of Economic and Social Affairs. Sustainable Development. Department of Economic and Social Affairs. Sustainable Development. 2023, Available at: https://sdgs.un.org/gsdr/gsdr2023, accessed December 2024.
L Panzella, F Moccia, R Nasti, S Marzorati, L Verotta and A Napolitano. Bioactive phenolic compounds from agri-food wastes: An update on green and sustainable extraction methodologies. Frontiers in Nutrition 2020; 7, 60.
J Dukić, M Hunić, M Nutrizio and AR Jambrak. Influence of high-power ultrasound on yield of proteins and specialized plant metabolites from sugar beet leaves (Beta vulgaris subsp. vulgaris var. altissima). Applied Sciences 2022; 12(18), 8949.
R Bashir, S Tabassum, A Adnan, A Rashid and A Adnan. Bioactive profile, pharmacological attributes and potential application of Beta vulgaris. Journal of Food Measurement and Characterization 2024; 18(5), 3732-3743.
FAO. Crops and livestock products; Yield and production quantity of crops. 2023, Available at: https://www.fao.org/faostat/en/#home, accessed December 2024.
N Chhikara, K Kushwaha, P Sharma, Y Gat and A Panghal. Bioactive compounds of beetroot and utilization in food processing industry: A critical review. Food Chemistry 2019; 272, 192-200.
E Abdo, S El-Sohaimy, O Shaltout, A Abdalla and A Zeitoun. Nutritional evaluation of beetroots (Beta vulgaris L.) and its potential application in a functional beverage. Plants 2020; 9(12), 1752.
C Mella, N Rojas, H Calderon-Bravo and LA Muñoz. Evaluating biocompounds in discarded beetroot (Beta vulgaris) leaves and stems for sustainable food processing solutions. Foods 2024; 13(16), 2603.
E Abdo, HMM Mansour, AM Galal, SA El-Sohaimy, MAE Gomaa, OE Shaltout and MG Allam. Beetroot stalk extract as a functional colorant for stirred yogurt beverages: Effect on nutritional value and stability during storage. Fermentation 2023; 9(10), 878.
S Singh, PK Omre, K Chand, A Kumar and P Awasthi. Process optimization of ultrasonic assisted extraction of betalains from red beet, Beta vulgaris L. waste stalks. Indian Journal of Experimental Biology 2021; 59(12), 858-866.
EM Abdo, OES Shaltout, S Ali and HMM Mansour. Functional orange juice fortified with beetroot by-products attenuates hyperlipidemia and obesity induced by a high-fat diet. Antioxidants 2022; 11(3), 457.
CD Dos Santos, RK Scherer, AS Cassini, LD Ferreira and IC Tessaro. Clarification of red beet stalks extract by microfiltration combined with ultrafiltration. Journal of Food Engineering 2016; 185, 35-41.
HBH Koubaier, A Snoussi, I Essaidi, MM Chaabouni, P Thonart and N Bouzouita. Betalain and phenolic compositions, antioxidant activity of tunisian red beet (Beta vulgaris L. conditiva) roots and stems extracts. International Journal of Food Properties 2014; 17(9), 1934-1945.
FM Abdel-Aziz, RM Aboelhaggag and EM Abo-Zaid. Healthy sorbet with eggplant peel and beet stalk-leaves extracts as natural colorants. Egyptian Journal of Chemistry 2023; 66(9), 145-157.
HF Battisella-Lasta, L Lentz, N Mezzomo and SR Salvador-Ferreira. Supercritical CO2 to recover extracts enriched in antioxidant compounds from beetroot aerial parts. Biocatalysis and Agricultural Biotechnology 2019; 19(1), 101169.
P Tutunchi, L Roufegarinejad, H Hamishehkar and A Alizadeh. Extraction of red beet extract with β-cyclodextrin-enhanced ultrasound assisted extraction: A strategy for enhancing the extraction efficacy of bioactive compounds and their stability in food models. Food Chemistry 2019; 297, 124994.
HF Battistella-Lasta, L Lentz, LG Gonçalves-Rodrigues, N Mezzomo, L Vitali and SR Salvador-Ferreira. Pressurized liquid extraction applied for the recovery of phenolic compounds from beetroot waste. Biocatalysis and Agricultural Biotechnology 2019; 21, 101353.
NF Ganwarige-Sumali, K Wood, EH Papaioannou, LJ Marshall, NN Sergeeva and C Boesch. Application of an ultrasound-assisted extraction method to recover betalains and polyphenols from red beetroot waste. ACS Sustainable Chemistry & Engineering 2021; 26(9), 8736-8747.
JP Prenhaca-Silva, B Cervejeira-Bolanho, N Stevanato, T Bovo-Masa and CD Silva. Ultrasound-assisted extraction of red beet pigments (Beta vulgaris L.): Influence of operational parameters and kinetic modeling. Journal of Food Processing and Preservation. 2020; 44(22).
P Ebrahimi, D Mihaylova, CM Marangon, L Grigoletto and A Lante. Impact of sample pretreatment and extraction methods on the bioactive compounds of sugar beet (Beta vulgaris L.) leaves. Molecules 2022; 27(22), 8110.
J Nutter, MV Fernandez, RJ Jagus and MV Agüero. Development of an aqueous ultrasound-assisted extraction process of bioactive compounds from beet leaves: A proposal for reducing losses and increasing biomass utilization. Journal of the Science of Food and Agriculture 2021; 101(5), 1989-1997.
A Lante, P Ebrahimi and D Mihaylova. Comparison of green technologies for valorizing sugar beet (Beta vulgaris L.) leaves. Food Science and Technology 2022; 5(2), 119-130.
A Demuner, A Dias, D Blank, C Cerceau, R Sousa, C Reis, M Santos and P Stringheta. Ultrasound-assisted extraction of active compounds from Beta vulgaris using deep eutectic solvents. Journal of Food Science and Technology 2023; 43(4), e107022.
A Ilghami, S Ghanbarzadeh and H Hamishehkar. Optimization of the ultrasonic-assisted extraction of phenolic compounds, ferric reducing activity and antioxidant activity of the Beta vulgaris using response surface methodology. Pharmaceutical Sciences. 2015; 21(1), 46-50.
LM Anaya-Esparza, EF Aurora-Vigo, Z Villagrán, E Rodríguez-Lafitte, JM Ruvalcaba-Gómez, MA Solano-Cornejo, V Zamora-Gasga, E Montalvo-González, H Gómez-Rodríguez, C Aceves-Aldrete and N González-Silva. Design of experiments for optimizing ultrasound-assisted extraction of bioactive compounds from plant-based sources. Molecules 2023; 28(23), 7752.
S Hernández-Estrada, LM Anaya-Esparza, S González-Torres, LA Hernández-Villaseñor, VM Gómez-Rodríguez, H Ramírez-Vega, Z Villagrán, JM Ruvalcaba-Gómez, N Rodríguez-Barajas and E Montalvo-González. Extraction of soluble phenols and flavonoids from native Mexican pigmented corn kernel powder by ultrasound: Optimization process using response surface methodology. Applied Sciences 2024; 14(17), 7869.
J Pérez-Jiménez, S Arranz, M Tabernero, M Díaz-Rubio, J Serrano, I Goñi and F Saura-Calixto. Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: Extraction, measurement and expression of results. Food Research International 2008; 41(3), 274-285.
FR Montreau. Sur le dosage des composés phénoliques totaux dans les vins par la méthode folin-ciocalteu. OENO One 1972; 24(4), 397-404
AK Esmaeili, RM Taha, S Mohajer and B Banisalam. Antioxidant activity and total phenolic and flavonoid content of various solvent extracts from in vivo and in vitro grown Trifolium pratense L. (red clover). BioMed Research International 2015; 2015, 643285.
FJ Barba, HN Rajha, E Debs, AM Abi-Khattar, S Khabbaz, BN Dar, MJ Simirgiotis, JM Castagnini, RG Maroun and N Louka. Optimization of polyphenols’ recovery from purple corn cobs assisted by infrared technology and use of extracted anthocyanins as a natural colorant in pickled turnip. Molecules 2022; 27(16), 5222.
RL Prior, X Wu and K Schaich. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. Journal of Agricultural and Food Chemistry 2005; 53(10), 4290-4302.
R Re, N Pellegrini, A Proteggente, A Pannala, M Yang and C Rice-Evans. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine 1999; 26(9-10), 1231-1237.
IFF Benzie and JJ Strain. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry 1996; 239(1), 70-76.
N González-Silva, Y Nolasco-González, G Aguilar-Hernández, Z Villagrán, J Acosta, E Montalvo-González and LM Anaya-Espaza. Ultrasound-assisted extraction of phenolic compounds from Psidium cattleianum leaves: Optimization using the response surface methodology. Molecules 2022; 27(11), 3557.
G Aguilar-Hernández, ML García-Magaña, M Vivar-Vera, SG Sáyago-Ayerdi, JA Sánchez-Burgos, J Morales-Castro, LM Anaya-Esparza and EM González. Optimization of ultrasound-assisted extraction of phenolic compounds from Annona muricata by-products and pulp. Molecules 2019; 24(5), 904.
H Myo and N Khat-udomkiri. Optimization of ultrasound-assisted extraction of bioactive compounds from coffee pulp using propylene glycol as a solvent and their antioxidant activities. Ultrasonics Sonochemistry 2022; 44(4), 106127.
M Iftikhar, H Zhang, A Iftikhar, A Raza, N Begum, A Tahamina, H Syed, M Khan and J Wang. Study on optimization of ultrasonic assisted extraction of phenolic compounds from rye bran. LWT 2020; 134(4), 110243.
HM Ali, W Almagribi and MN Al-Rashidi. Antiradical and reductant activities of anthocyanidins and anthocyanins, structure–activity relationship and synthesis. Food Chemistry 2016; 194, 1275-1282.
VG Nikolić, DZ Troter, IM Savić, IM Savić-Gajić, JB Zvezdanović, IB Konstantinović and SS Konstantinović. Design and optimization of “greener” and sustainable ultrasound-assisted extraction of valuable bioactive compounds from common centaury (Centaurium erythraea Rafn) aerial parts: A comparative study using aqueous propylene glycol and ethanol. Industrial Crops and Products 2023; 192, 116070.
G Fernández-Barbero, C Pinedo, E Espada-Bellido, M Ferreiro-González, C Carrera, M Palma and C García-Barroso. Optimization of ultrasound-assisted extraction of bioactive compounds from jabuticaba (Myrciaria cauliflora) fruit through a Box-Behnken experimental design. Food Science and Technology 2019; 39(3), 1-12.
CB Aware, RR Patil, GD Vyavahare, ST Gurme and JP Jadhav. Ultrasound-assisted aqueous extraction of phenolic, flavonoid compounds and antioxidant activity of Mucuna macrocarpa beans: Response surface methodology optimization. Journal of the American College of Nutrition 2019; 38(4), 364-372.
T Maher, NA Kabbashi, MES Mirghani, MZ Alam, D Daddiouaissa, F Abdulhafiz, MF Hanif-Reduan, JI Omran, MKAA Razab and A Mohammed. Optimization of ultrasound-assisted extraction of bioactive compounds from Acacia seyal gum using response surface methodology and their chemical content identification by Raman, FTIR, and GC-TOFMS. Antioxidants 2021; 10(10), 1612.
T Alves, CC Triques, PA Palsikowski, CD Silva, ML Fiorese, EAD Silva and MR Fagundes-Klen. Improved extraction of bioactive compounds from Monteverdia aquifolia leaves by pressurized-liquid and ultrasound-assisted extraction: Yield and chemical composition. The Journal of Supercritical Fluids 2022; 181, 105468.
A Sanou, K Konaté, K Kabakdé, R Dakuyo, D Bazié, S Hemayoro and M Dicko. Modelling and optimisation of ultrasound-assisted extraction of roselle phenolic compounds using the surface response method. Scientific Reports 2023; 13(1), 358.
L Yang, JG Jiang, WF Li, J Chen, DY Wang and L Zhu. Optimum extraction process of polyphenols from the bark of Phyllanthus emblica L. based on the response surface methodology. Journal of Separation Science 2009; 32(9), 1437-1444.
X Chen, J Ding, D Ji, S He and H Ma. Optimization of ultrasonic-assisted extraction conditions for bioactive components from coffee leaves using the Taguchi design and response surface methodology. Journal of Food Science 2020; 85(6), 1742-1751.
T Ahmed, M Rana, M Hossain, S Ullah and M Suzauddula. Optimization of ultrasound‑assisted extraction using response surface methodology for total anthocyanin content, total phenolic content, and antioxidant activities of roselle (Hibiscus sabdariffa L.) calyces and comparison with conventional Soxhlet extraction. Biomass Conversion and Biorefinery 2024; 14, 28985-28999.
H Ozcan and I Damar. Valorization of spinach roots for recovery of phenolic compounds by ultrasound-assisted extraction: Characterization, optimization, and bioaccessibility. European Food Research and Technology 2023; 249, 1899-1913.
B Martín-García, MJ Aznar-Ramos, V Verardo and AM Gómez-Caravaca. The establishment of ultrasonic-assisted extraction for the recovery of phenolic compounds and evaluation of their antioxidant activity from Morus alba leaves. Foods 2022; 11(3), 314.
S Wu, W Chen, S Lu, H Zhang and L Yin. Metabolic engineering of shikimic acid biosynthesis pathway for the production of shikimic acid and its branched products in microorganisms: Advances and prospects. Molecules 2022; 27(15), 4779.
DV Bochkov, SV Sysolyatin, AI Kalashnikov and IA Surmacheva. Shikimic acid: Review of its analytical, isolation, and purification techniques from plant and microbial sources. The Journal of Biological Chemistry 2011; 5(1), 5-17.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






