Integrated Evaluation of NADES for Green Extraction of Bioactive Compounds from Strawberries: A Study on Anthocyanin, Antioxidant Activity, Chemometric FTIR Profiling
DOI:
https://doi.org/10.48048/tis.2026.11388Keywords:
Anthocyanins, Chemometric, FTIR, NADES, PCA, Anthocyanins, Chemometric, FTIR, NADES, PCAAbstract
Strawberries (Fragaria x ananassa) are a rich source of anthocyanins and phenolic compounds with strong antioxidant properties. This study introduces an integrated approach combining quantitative analysis, including total anthocyanin content, total phenolic content, and antioxidant activity via DPPH assay, along with qualitative profiling using Fourier Transform Infrared (FTIR) spectroscopy and Principal Component Analysis (PCA). The aim was to thoroughly assess the extraction efficiency of 5 Natural Deep Eutectic Solvents (NADES), synthesized from choline chloride and various organic acids (citric, oxalic, malic, tartaric and lactic), in comparison to ethanol. Among these, NADES E (choline chloride:lactic acid) showed the highest extraction performance, yielding 125.57 ± 12.22 mg/100 g anthocyanins, 11,032.02 ± 785.13 mg GAE/g phenolics, and 89.15 ± 6.1% antioxidant activity. Interestingly, NADES A (choline chloride:citric acid) produced similar bioactive yields but exhibited the most intense FTIR absorbance in regions associated with phenolic functional groups (-OH, C=O and C=C), indicating a different compound affinity or preservation capacity. This suggests that while NADES E is superior for quantitative yield, NADES A might better retain specific chemical fingerprints. Moreover, PCA demonstrated clear separation of NADES A, C, and E from ethanol, highlighting the superior compound selectivity and profile retention of NADES. These findings highlight the importance of integrating multiple analytical techniques to evaluate both the efficiency and selectivity of green solvent systems for extracting bioactive compounds from plant matrices.
HIGHLIGHTS
- Natural Deep Eutectic Solvents (NADES) demonstrated superior extraction performance compared to ethanol for isolating anthocyanins and phenolic compounds from strawberries, supporting their potential as green, sustainable alternatives in food and phytochemical industries.
- Principal Component Analysis (PCA) based on FTIR spectra (1,400 - 1,700 cm−1) revealed clear separation between NADES-based extracts and ethanol extracts, with NADES A, C, and E forming distinct clusters, indicating higher chemical selectivity and unique extract profiles associated with different NADES compositions.
- This study integrates FTIR spectroscopy, PCA chemometric analysis, and quantitative bioactive profiling, offering a comprehensive understanding of both extraction efficiency and compound specificity, and highlighting the importance of solvent design in optimizing green extraction strategies.
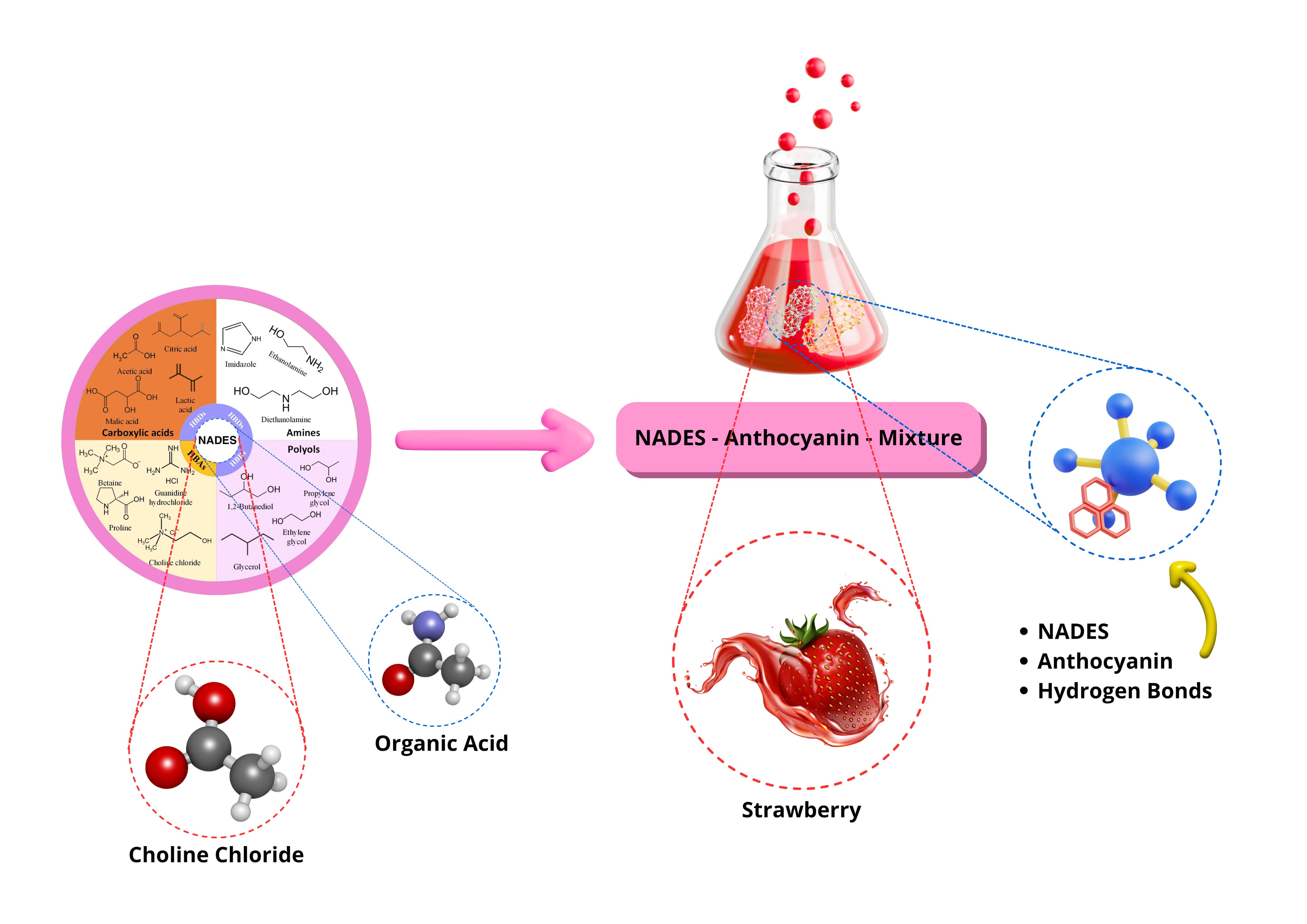
GRAPHICAL ABSTRACT
Downloads
References
P Padmanabhan, A Mizran, JA Sullivan and G Paliyath. Strawberries. In: B Caballero, PM Finglas and F Toldrá (Eds.). Encyclopedia of food and health. Academic Press, Oxford, England, 2016, p. 193-198.
AM Mustafa, S Angeloni, D Abouelenein, L Acquaticci, J Xiao, G Sagratini, F Maggi, S Vittori and G Caprioli. A new HPLC-MS/MS method for the simultaneous determination of 36 polyphenols in blueberry, strawberry and their commercial products and determination of antioxidant activity. Food Chemistry 2022; 367, 130743.
M Huang, Y Han, L Li, K Rakariyatham, X Wu, Z Gao and H Xiao. Protective effects of non-extractable phenolics from strawberry against inflammation and colon cancer in vitro. Food Chemistry 2022; 374, 131759.
X Sui, Y Zhang, L Jiang and W Zhou. Anthocyanins in food. In: L Melton, F Shahidi and P Varelis (Eds.). Encyclopedia of food chemistry. Academic Press, Oxford, England, 2019, p. 10-17.
D Li, X Zhang, L Li, MS Aghdam, X Wei, J Liu, Y Xu and Z Luo. Elevated CO2 delayed the chlorophyll degradation and anthocyanin accumulation in postharvest strawberry fruit. Food Chemistry 2019; 285, 163-170.
J Hellström, S Karhu, J Karhu, E Järvenpää and AL Välimaa. Phenolic profiles differentiate wild bilberry and cultivated blueberry fruit. LWT 2024; 199, 116080.
Y Yuan, Y Tian, S Gao, X Zhang, X Gao and J He. Effects of environmental factors and fermentation on red raspberry anthocyanins stability. LWT 2023; 173, 114252.
GAB Canuto, DR Oliveira, LSM da Conceição, JPS Farah and MFM Tavares. Development and validation of a liquid chromatography method for anthocyanins in strawberry (Fragaria spp.) and complementary studies on stability, kinetics and antioxidant power. Food Chemistry 2016; 192, 566-574.
MU Kousar, A Jabeen, T Fatima, SZ Hussain, IA Zargar, T Amin and M Yaseen. Optimization of ultrasonic-assisted extraction of eugenol-rich fraction from basil leaves: Characterization of extract for phenols, flavonoids and antioxidant activity. Food Chemistry Advances 2023; 3, 100374.
Y Wang, Y Ye, L Wang, W Yin and J Liang. Antioxidant activity and subcritical water extraction of anthocyanin from raspberry process optimization by response surface methodology. Food Bioscience 2021; 44, 101394.
F Pena-Pereira and IDL Calle. Solvents and eutectic solvents. In: P Worsfold, C Poole, A Townshend and M Miró (Eds.). Encyclopedia of analytical science. Academic Press, Oxford, England, 2019, p. 184-190.
DTD Silva, R Pauletto, SDS Cavalheiro, VC Bochi, E Rodrigues, J Weber, CDBD Silva, FDP Morisso, MT Barcia and T Emanuelli. Natural deep eutectic solvents as a biocompatible tool for the extraction of blueberry anthocyanins. Journal of Food Composition and Analysis 2020; 89, 103470.
BH Stuart. Organic molecules. In: J Wiley and L Sons (Eds.). Infrared spectroscopy: Fundamentals and applications. Wiley, Queensland, Australia, 2004.
IT Jolliffe and J Cadima. Principal component analysis: A review and recent developments. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 2016; 374(2065), 20150202.
MS Jovanović, N Krgović, J Živković, T Stević, G Zdunić, D Bigović and K Šavikin. Ultrasound-assisted natural deep eutectic solvents extraction of bilberry anthocyanins: Optimization, bioactivities, and storage stability. Plants 2022; 11(20), 2680.
O Zannou and I Koca. Greener extraction of anthocyanins and antioxidant activity from blackberry (Rubus spp) using natural deep eutectic solvents. LWT 2022; 158, 113184.
AR Pereira, VC Fernandes, C Delerue-Matos, VD Freitas, N Mateus and J Oliveira. Exploring acylated anthocyanin-based extracts as a natural alternative to synthetic food dyes: Stability and application insights. Food Chemistry 2024; 461, 140945.
T Taghavi, H Patel and R Rafie. Anthocyanin extraction method and sample preparation affect anthocyanin yield of strawberries. Natural Product Communications 2022; 17(5), 1934578X2210999.
T Dzhanfezova, G Barba-Espín, R Mülle, B Joernsgaard, JN Hegelund, B Madsen, DH Larsen, MM Vega and TB Toldam-Andersen. Anthocyanin profile, antioxidant activity and total phenolic content of a strawberry (Fragaria × ananassa Duch) genetic resource collection. Food Bioscience 2020; 36, 100620.
MZ Alam, MSR Alhebsi, S Ghnimi and A Kamal-Eldin. Inability of total antioxidant activity assays to accurately assess the phenolic compounds of date palm fruit (Phoenix dactylifera L.). NFS Journal 2021; 22, 32-40.
B Enaru, G Drețcanu, TD Pop, A Stǎnilǎ and Z Diaconeasa. Anthocyanins: Factors affecting their stability and degradation. Antioxidants 2021; 10(12), 1967.
S Zhang, S Lin, J Zhang and W Liu. Ultrasound-assisted natural deep eutectic solvent extraction of anthocyanin from Vitis davidii Foex. pomace: Optimization, identification, antioxidant activity and stability. Heliyon 2024; 10(12), e33066.
P Velásquez, D Bustos, G Montenegro and A Giordano. Ultrasound-assisted extraction of anthocyanins using natural deep eutectic solvents and their incorporation in edible films. Molecules 2021; 26(4), 984.
AN Nunes, A Borges, AA Matias, MR Bronze and J Oliveira. Alternative extraction and downstream purification processes for anthocyanins. Molecules 2022; 27(2), 368.
ME Alañón, M Ivanović, S Pimentel-Mora, I Borrás-Linares, D Arráez-Román and A Segura-Carretero. A novel sustainable approach for the extraction of value-added compounds from Hibiscus sabdariffa L. calyces by natural deep eutectic solvents. Food Research International 2020; 137, 109646.
Y Dai, E Rozema, R Verpoorte and YH Choi. Application of natural deep eutectic solvents to the extraction of anthocyanins from Catharanthus roseus with high extractability and stability replacing conventional organic solvents. Journal of Chromatography A 2016; 1434, 50-56.
Y Bi, X Chi, R Zhang, Y Lu, Z Wang, Q Dong, C Ding, R Yang and L Jiang. Highly efficient extraction of mulberry anthocyanins in deep eutectic solvents: Insights of degradation kinetics and stability evaluation. Innovative Food Science & Emerging Technologies 2020; 66, 102512.
S Prakash, A Goswami, R Patil, A Mitra and NN Kutty. An eco-friendly approach to extract anthocyanins from rose flowers using natural deep eutectic solvents. Industrial Crops and Products 2024; 210, 118059.
M Lakshmikanthan, S Muthu, K Krishnan, AB Altemimi, NN Haider, L Govindan, J Selvakumari, ZT Alkanan, F Cacciola and YM Francis. A comprehensive review on anthocyanin-rich foods: Insights into extraction, medicinal potential, and sustainable applications. Journal of Agriculture and Food Research 2024; 17, 101245.
NR Hernández-Martínez, C Blanchard, D Wells and MR Salazar-Gutiérrez. Current state and future perspectives of commercial strawberry production: A review. Scientia Horticulturae 2023; 312, 111893.
I Sadowska-Bartosz and G Bartosz. Antioxidant activity of anthocyanins and anthocyanidins: A critical review. International Journal of Molecular Sciences 2024; 25(22), 12001.
QQ Koh, ZL Chew, Y Zhao, YL Kua, S Gan, KW Tan, TZE Lee and HLN Lau. Formulation and characterization of natural deep eutectic solvents (NADES) for simultaneous phenolics and carotenes extraction from fresh oil palm leaf. Food and Bioproducts Processing 2024; 147, 459-473.
A Paiva, R Craveiro, I Aroso, M Martins, RL Reis and ARC Duarte. Natural deep eutectic solvents - solvents for the 21st century. ACS Sustainable Chemistry & Engineering 2014; 2(5), 1063-1071.
NPE Hikmawanti, D Ramadon, I Jantan and A Mun’Im. Natural Deep Eutectic Solvents (NADES): Phytochemical extraction performance enhancer for pharmaceutical and nutraceutical product development. Plants 2021; 10(10), 2091.
K Radošević, N Ćurko, VG Srček, MC Bubalo, M Tomašević, KK Ganić and IR Redovniković. Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT 2016; 73, 45-51.
S Milošević, AB Markovinović, N Teslić, A Mišan, M Pojić, IB Karačonji, K Jurica, D Lasić, P Putnik, DB Kovačević and B Pavlić. Use of natural deep eutectic solvent (NADES) as a green extraction of antioxidant polyphenols from strawberry tree fruit (Arbutus unedo L.): An optimization study. Microchemical Journal 2024; 200, 110284.
H Du, W Wu, Y Wang, P Tang, Y Wu, J Qiu, C Xu, L Li and M Yang. Natural deep eutectic solvent-ultrasound for the extraction of flavonoids from Fructus aurantii: Theoretical screening, experimental and mechanism. Arabian Journal of Chemistry 2024; 17(9), 105886.
K Pusty, KK Dash, S Giri, GVSB Raj, A Tiwari, AM Shaikh and K Béla. Ultrasound assisted phytochemical extraction of red cabbage by using deep eutectic solvent: Modelling using ANFIS and optimization by genetic algorithms. Ultrasonics Sonochemistry 2024; 102, 106762.
X Fu, D Wang, T Belwal, J Xie, Y Xu, L Li, L Zou, L Zhang and Z Luo. Natural deep eutectic solvent enhanced pulse-ultrasonication assisted extraction as a multi-stability protective and efficient green strategy to extract anthocyanin from blueberry pomace. LWT 2021; 144, 111220.
R Thakur, V Gupta, T Ghosh and AB Das. Effect of anthocyanin-natural deep eutectic solvent (lactic acid/fructose) on mechanical, thermal, barrier, and pH-sensitive properties of polyvinyl alcohol based edible films. Food Packaging and Shelf Life 2022; 33, 100914.
A Palos-Hernandez, MYG Fernández, JE Burrieza, JL Pérez-Iglesias and AM González-Paramás. Obtaining green extracts rich in phenolic compounds from underexploited food by-products using natural deep eutectic solvents. Opportunities and challenges. Sustainable Chemistry and Pharmacy 2022; 29, 100773.
O Kapitan, D Kusumawardhani, F Nitti and G Kedjo. Profil senyawa polar dan semi polar daun tembelekan (Lantana camara) asal pulau timor (in Indonesian). Indigenous Biologi: Jurnal Pendidikan Dan Sains Biologi 2024; 7(3), 111-121.
IL Tarigan, JAN Sinurat, II Ramadhan, DE Wijaya, RD Puspitasari and MS Sowe. Green extraction of bioactive compounds from Peronema canescens Jack. using sodium acetate/glycerol-based natural deep eutectic solvents as a source of natural antioxidants. Jurnal Kartika Kimia 2025; 8(1), 60-70.
R Mogale, YW Abraha, M Schutte-Smith, HG Visser and E Erasmus. Natural deep eutectic solvents (NADES) derived from choline chloride and indole-3-butyric acid for optimized CO2 utilization through cycloaddition with epichlorohydrin. Journal of Ionic Liquids 2025; 5, 100142.
M El Mouftari, I Essafi, A Khalidi, F Kzaiber, GAM Ali, FZ Mahjoubi and A Oussama. Applications of FTIR and chemometrics methods in authenticity analysis of walnut oil. Emergent Materials 2022; 5(1), 167-174.
D Granato, JS Santos, GB Escher, BL Ferreira and RM Maggio. Use of principal component analysis (PCA) and hierarchical cluster analysis (HCA) for multivariate association between bioactive compounds and functional properties in foods: A critical perspective. Trends in Food Science & Technology 2018; 72, 83-90.
ABD Nandiyanto, R Oktiani and R Ragadhita. How to read and interpret FTIR spectroscope of organic material. Indonesian Journal of Science and Technology 2019; 4(1), 97-118.
JO Airouyuwa, H Mostafa, A Riaz and S Maqsood. Utilization of natural deep eutectic solvents and ultrasound-assisted extraction as green extraction technique for the recovery of bioactive compounds from date palm (Phoenix dactylifera L.) seeds: An investigation into optimization of process parameters. Ultrasonics Sonochemistry 2022; 91, 106233.
T Jeliński, M Przybyłek and P Cysewski. Natural deep eutectic solvents as agents for improving solubility, stability and delivery of curcumin. Pharmaceutical Research 2019; 36(8), 116.
M Mayonu, S Babaee, L Jiang, D Wilson and B Wang. Evaluation of a new approach for principal component analysis application in metabolomics studies. Analytical Letters 2025. https://doi.org/10.1080/00032719.2025.
R Qin, H Chen, R Wen, G Li and Z Meng. Effect of boric acid on the ionization equilibrium of α-hydroxy carboxylic acids and the study of its applications. Molecules 2023; 28(12), 4723.
P Kalhor and K Ghandi. Deep eutectic solvents for pretreatment, extraction, and catalysis of biomass and food waste. Molecules 2019; 24, E4012.
C Florindo, LC Branco and IM Marrucho. Quest for green-solvent design: From hydrophilic to hydrophobic (deep) eutectic solvents. ChemSusChem 2019; 12(8), 1549-1559.
ML Picchio, D Minudri, D Mantione, M Criado-Gonzalez, G Guzmán-González, R Schmarsow, AJ Müller, LC Tomé, RJ Minari and D Mecerreyes. Natural deep eutectic solvents based on choline chloride and phenolic compounds as efficient bioadhesives and corrosion protectors. ACS Sustainable Chemistry & Engineering 2022; 10(25), 8135-8142.
ON Pozharitskaya, ED Obluchinskaya, VA Shikova, EV Flisyuk, EV Vishnyakov, EV Makarevich and AN Shikov. Physicochemical and antimicrobial properties of lactic acid-based natural deep eutectic solvents as a function of water content. Applied Sciences 2024; 14(22), 10409.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






