Temperature and Light-Dependent Quercetin Glycoside Transformation in Shallot Bulbs: Integrated Assessment of Bioactive Dynamics and Storage Quality
DOI:
https://doi.org/10.48048/tis.2026.11293Keywords:
Allium ascalonicum L, Temperature, Light exposure, Quercetin glycoside, Bioactive retention, Postharvest storage, Allium ascalonicum L., Temperature, Light exposure, Quercetin glycoside, Bioactive retention, Postharvest storageAbstract
Shallots (Allium ascalonicum L.) contain valuable quercetin glycoside bioactive compounds that undergo complex transformations during postharvest storage. This study aimed to elucidate the transformation kinetics of individual quercetin compounds under different temperature-light combinations to optimize storage protocols for improved bioactive retention. Fresh shallot bulbs were stored using factorial combinations of temperature (15 vs 30 °C) and light conditions (darkness vs 18,000 lux). Individual quercetin compounds (quercetin 3,4’-diglucoside, quercetin 4’-glucoside, free quercetin, and quercetin 3-glucoside), reducing sugar content, and weight loss were monitored during storage. Temperature and light exhibited interactive effects on the kinetics of quercetin transformation. The combination of 15 °C with 18,000 lux illumination produced maximum free quercetin accumulation (27.00 g/kg dw) at day 12, substantially enhancing bioactive compound concentrations. Higher temperature (30 °C) accelerated conversion rates but reduced overall compound stability, with rapid degradation observed in later storage phases. Additionally, storage at 15 °C in darkness best maintains reducing sugar stability (64.51 mg/100 g dw), whereas storage at 30 °C with light exposure shows a steep decline to 32.89 mg/100 g dw, indicating metabolic instability. These findings provide insight into optimizing postharvest storage of Allium crops to retain functional compounds in tropical environments.
HIGHLIGHTS
- Temperature-light interactions control quercetin transformation
- 15 °C with light maximizes free quercetin
- Cool, dark storage best preserves reducing sugar stability
- High temperature (30 °C) increases bioactive conversion but reduces compound stability
- Temperature-light management protocols enhance bioactive retention
GRAPHICAL ABSTRACT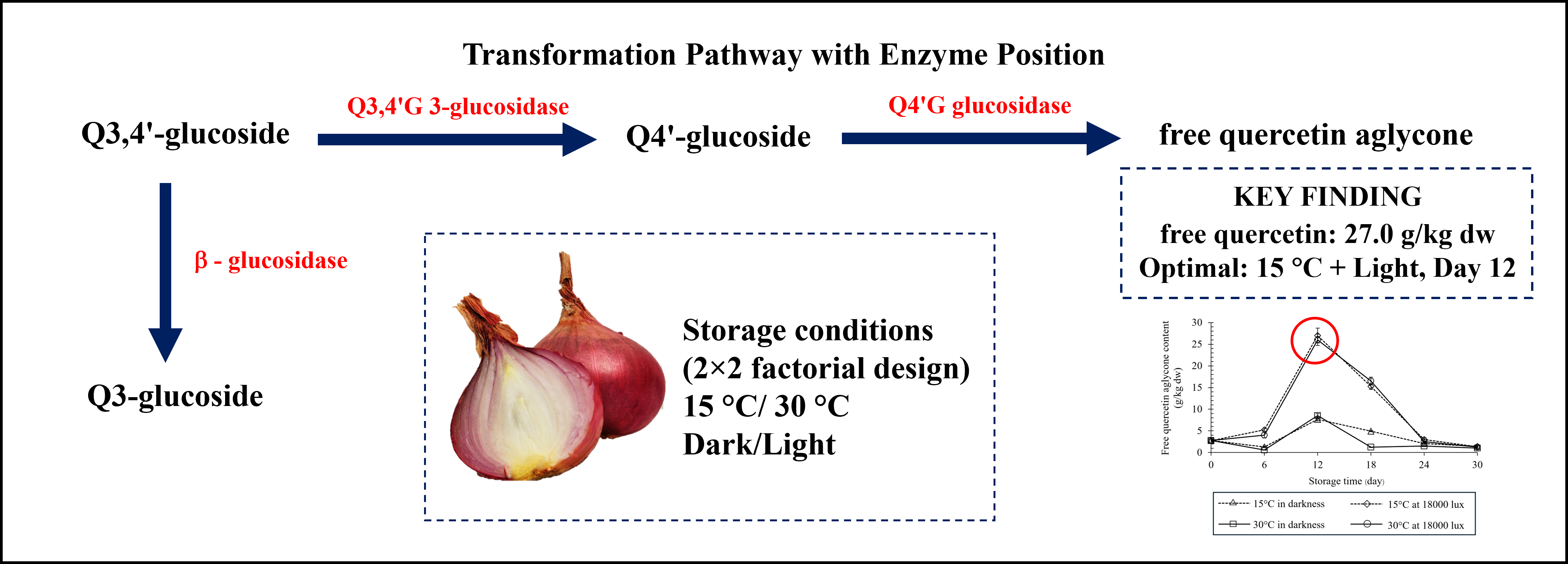
Downloads
References
C Moldovan, O Frumuzachi, M Babota, L Barros, A Mocan, S Carradori and G Crisan. Therapeutic uses and pharmacological properties of shallot (Allium ascalonicum): A systematic review. Frontiers in Nutrition 2022; 9, 903686.
J Yang, KJ Meyers, J van der Heide and RH Liu. Varietal differences in phenolic content and antioxidant and antiproliferative activities of onions. Journal of Agricultural and Food Chemistry 2004; 52(22), 6787-6793.
W Wiczkowski, J Romaszko, A Bucinski, D Szawara-Nowak, J Honke, H Zielinski and MK Piskula. Quercetin from shallots (Allium cepa L. var. aggregatum) is more bioavailable than its glucosides. The Journal of Nutrition 2008; 138(5), 885-888.
F Aghababaei and M Hadidi. Recent advances in potential health benefits of quercetin. Pharmaceuticals 2023; 16, 1020.
R Tanomrat, W Payuhakrit, C Naktubtim, P Aimvijarn, T Khorporn, N Yongpraphat and P Suwannalert. Pharmacological and in vitro studies on quercetin-rich onion peel extract (Allium cepa) and n-acetylcysteine enhancing apoptosis in colorectal cancer. Trends in Sciences 2025; 22(3), 9111.
M Lesjak, I Beara, N Simin, D Pintać, T Majkić, K Bekvalac, D Orčić and N Mimica-Dukić. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. Journal of Functional Foods 2018; 40, 68-75.
HV Beretta, F Bannoud, M Insani, F Berli, P Hirschegger, CR Galmarini and PF Cavagnaro. Relationships between bioactive compound content and the antiplatelet and antioxidant activities of 6 Allium vegetable species. Food Technology and Biotechnology 2017; 55(2), 266-275.
W Wang, C Sun, L Mao, P Ma, F Liu, J Yang and Y Gao. The biological activities, chemical stability, metabolism and delivery systems of quercetin: A review. Trends in Food Science & Technology 2016; 56, 21-38.
GB Calica and MMN Dulay. Assessment of the postharvest systems and losses of shallots in Ilocos, Philippines. Asian Journal of Postharvest and Mechanization 2018; 1(1), 81-92.
S Rojanatamornnat, T Thongsook and U Suttisansanee. Effect of different exposed lights on quercetin and quercetin glucoside content in onion (Allium cepa L.). International Food Research Journal 2015; 22(4), 1424-1432.
F Flamminii, G D’Alessio, M Chiarini, A Di Michele, A De Bruno, D Mastrocola and CD Di Mattia. Valorization of onion by-products bioactive compounds by spray drying encapsulation technique. Foods 2025; 14(3), 425.
JM Moreno-Rojas, A Moreno-Ortega, JL Ordóñez, R Moreno-Rojas, J Pérez-Aparicio and G Pereira-Caro. Development and validation of UHPLC-HRMS methodology for the determination of flavonoids, amino acids and organosulfur compounds in black onion, a novel derived product from fresh shallot onions (Allium cepa var. aggregatum). LWT-Food Science and Technology 2018; 97, 376-383.
XX Zhao, FJ Lin, H Li, HB Li, DT Wu, F Geng, W Ma, Y Wang, BH Miao and RY Gan. Recent advances in bioactive compounds, health functions, and safety concerns of onion (Allium cepa L.). Frontiers in Nutrition 2021; 8, 669805.
K Sharma and YR Lee. Effect of different storage temperature on chemical composition of onion (Allium cepa L.) and its enzymes. Journal of Food Science and Technology 2016; 53(7), 2992-2997.
EY Ko, SH Nile, K Sharma, GH Li and SW Park. Effect of different exposed lights on quercetin and quercetin glucoside content in onion (Allium cepa L.). Saudi Journal of Biological Sciences 2015; 22(4), 398-403.
AK Singh, S Dhanapal and BS Yadav. The dynamic responses of plant physiology and metabolism during environmental stress progression. Molecular Biology Reports 2020; 47, 1459-1470.
GL Miller. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry 1959; 31(3), 426-428.
MN Islam, A Wang, JS Pedersen, JN Sørensen, O Korner and M Edelenbos. Online measurement of temperature and relative humidity as marker tools for quality changes in onion bulbs during storage. PLoS One 2019; 14(1), e0210577.
O Galsurker, A Doron-Faigenboim, P Teper-Bamnolker, A Daus, A Lers and D Eshel. Differential response to heat stress in outer and inner onion bulb scales. Journal of Experimental Botany 2018; 69(16), 4047-4064.
I Lara, A Heredia and E Domínguez. Shelf life potential and the fruit cuticle: The unexpected player. Frontiers Plant Science 2019; 10, 770.
S Gao, X Liu, Y Liu, B Cao, Z Chen and K Xu. Photosynthetic characteristics and chloroplast ultrastructure of welsh onion (Allium fistulosum L.) grown under different LED wavelengths. BMC Plant Biology 2020; 20, 78.
L Riglet, S Gatti and E Moyroud. Sculpting the surface: Structural patterning of plant epidermis. iScience 2021; 24(11), 103346.
U Takahama. Oxidation of vacuolar and apoplastic phenolic substrates by peroxidase: Physiological significance of the oxidation reactions. Phytochemistry Reviews 2004; 3, 207-219.
KS Yoo, EJ Lee and BS Patil. Changes in quercetin glucoside concentrations of onion bulbs by scales, during storage, and in sprouting leaves exposed to UV. Postharvest Biology and Technology 2013; 83, 65-71.
A Zhou and OA Sadik. Comparative analysis of quercetin oxidation by electrochemical, enzymatic, autoxidation, and free radical generation techniques: A mechanistic study. Journal of Agricultural and Food Chemistry 2008; 56, 12081-12091.
L Xie, Z Deng, J Zhang, H Dong, W Wang, B Xing and X Liu. Comparison of flavonoid O-glycoside, C-glycoside and their aglycones on antioxidant capacity and metabolism during in vitro digestion and in vivo. Foods 2022; 11(6), 882.
SA Petropoulos, G Ntatsi and ICFR Ferreira. Long-term storage of onion and the factors that affect its quality: A critical review. Food Reviews International 2016; 33(1), 62-83.
S Raines, C Henson and MJ Havey. Genetic analyses of soluble carbohydrate concentrations in onion bulbs. Journal of the American Society for Horticultural Science 2009; 134(6), 618-623.
GA Chope, K Cools, JP Hammond, AJ Thompson and LA Terry. Physiological, biochemical and transcriptional analysis of onion bulbs during storage. Annals of Botany 2012; 109(4), 819-831.
Shivakumar and SY Chandrashekar. Physio-chemical changes during post harvest handing of onion (Allium cepa L.) - a review. Agricultural Research Communication Centre 2014; 35(3), 225-232.
A Martínez-Sánchez, JA Tudela, C Luna, A Allende and MI Gil. Low oxygen levels and light exposure affect quality of fresh-cut Romaine lettuce. Postharvest Biology and Technology 2011; 59, 34-42.
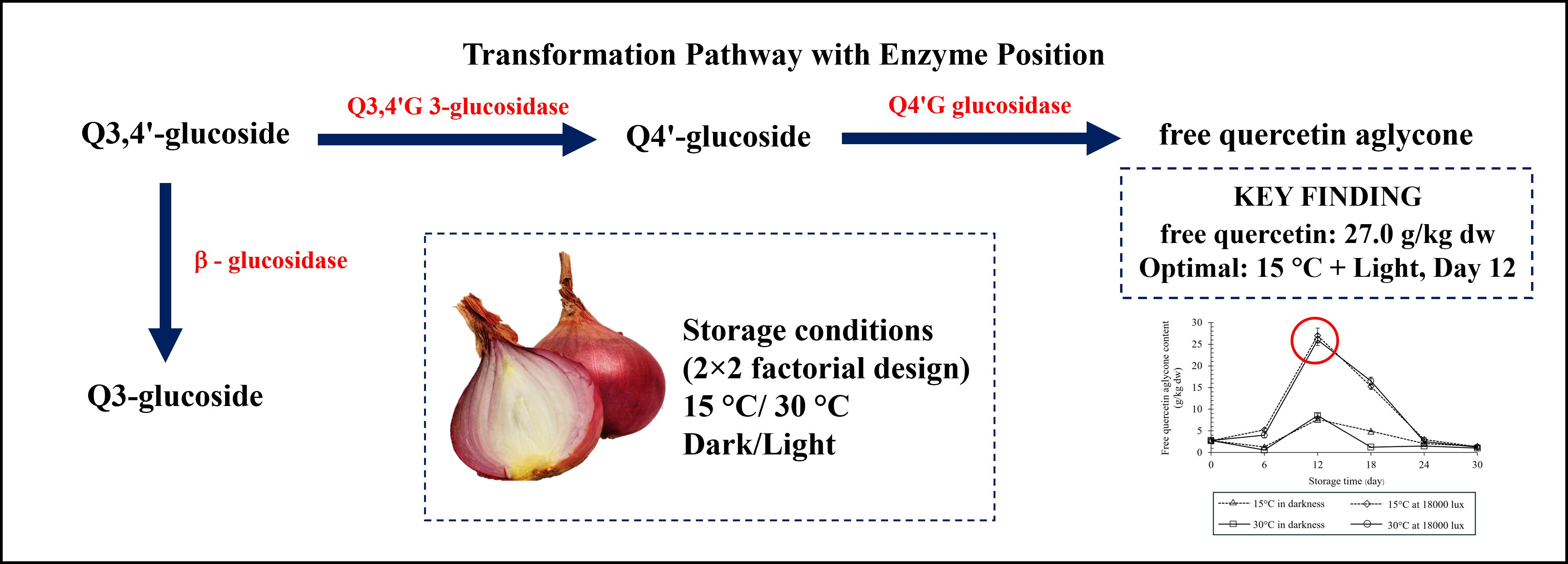
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






