Optimized CO2 Capture Using Water-Based Absorbent under Circulated Water Conditions for Carbon Capture and Storage (CCS) Applications
DOI:
https://doi.org/10.48048/tis.2026.11148Keywords:
Reaction kinetics, Climate change, Diffusivity, Equilibrium, Absorption, CO2 Capture, EnvironmentAbstract
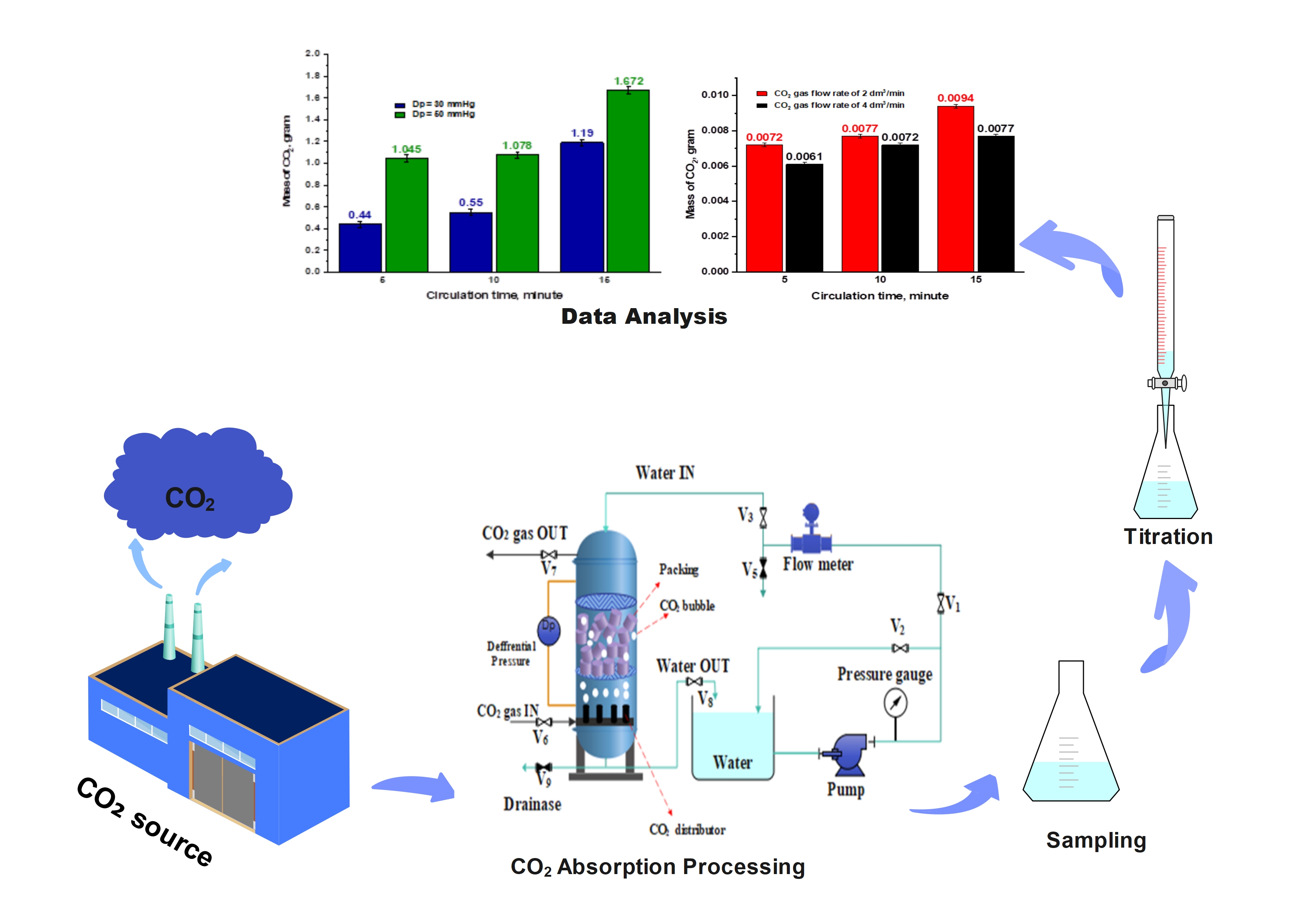
Capturing CO2 emissions using a recirculating water-based system offers a simple and eco-friendly alternative to conventional Carbon Capture and Storage (CCS) methods for reducing emissions and mitigating climate change. To optimize CO2 solubility in water, it is essential to integrate and evaluate the key variables that influence the capture process, enabling the identification of ideal operating conditions. This study aims to analyze the phenomena and examine the effects of variables such as CO2 gas flow rate, water flow rate, water temperature, circulation time, and differential pressure on the effectiveness of the CO2 capture process, as indicated by the mass of CO2 dissolved in water within the system. The experiment was carried out using a transparent absorber column filled with packing material. CO2 gas with 99.9% purity was introduced at flow rates of 2 and 4 dm3/min, while water was recirculated at flow rates of 0.25 and 0.5 dm3/min. The system operated at water temperatures between 24 and 30 °C, with circulation durations of 5, 10, and 15 min, and differential pressures of 30 and 50 mmHg. Samples were collected at the end of each circulation period and CO2 absorption was determined through titration using 0.1 M NaOH and phenolphthalein as the indicator. The results demonstrated that higher water flow rates and increased pressure differentials enhanced the capture performance, as evidenced by greater CO2 absorption. Conversely, elevated water temperatures and higher CO2 gas flow rates led to decreased absorption efficiency. The most favorable conditions were achieved at 24 °C, a water flow rate of 0.5 dm3/min, a circulation time of 15 min, and a differential pressure of 50 mmHg, resulting in 1.672 g of CO2 absorbed. The results of this study indicate that water has the potential to serve as an environmentally friendly alternative technology for CCS applications. However, further research is needed to develop a process that is effective, efficient, and more cost-effective.
HIGHLIGHTS
- Higher water flow rates and pressure differentials significantly improved CO2 capture efficiency.
- Increased water temperature and CO2 gas flow rate reduced absorption performance.
- Optimal conditions yielded 1.672 g CO2 absorbed at 24 °C, 0.5 dm3/min water flow, circulation time of 15 min, and differential pressure (Dp) of 50 mmHg.
GRAPHICAL ABSTRACT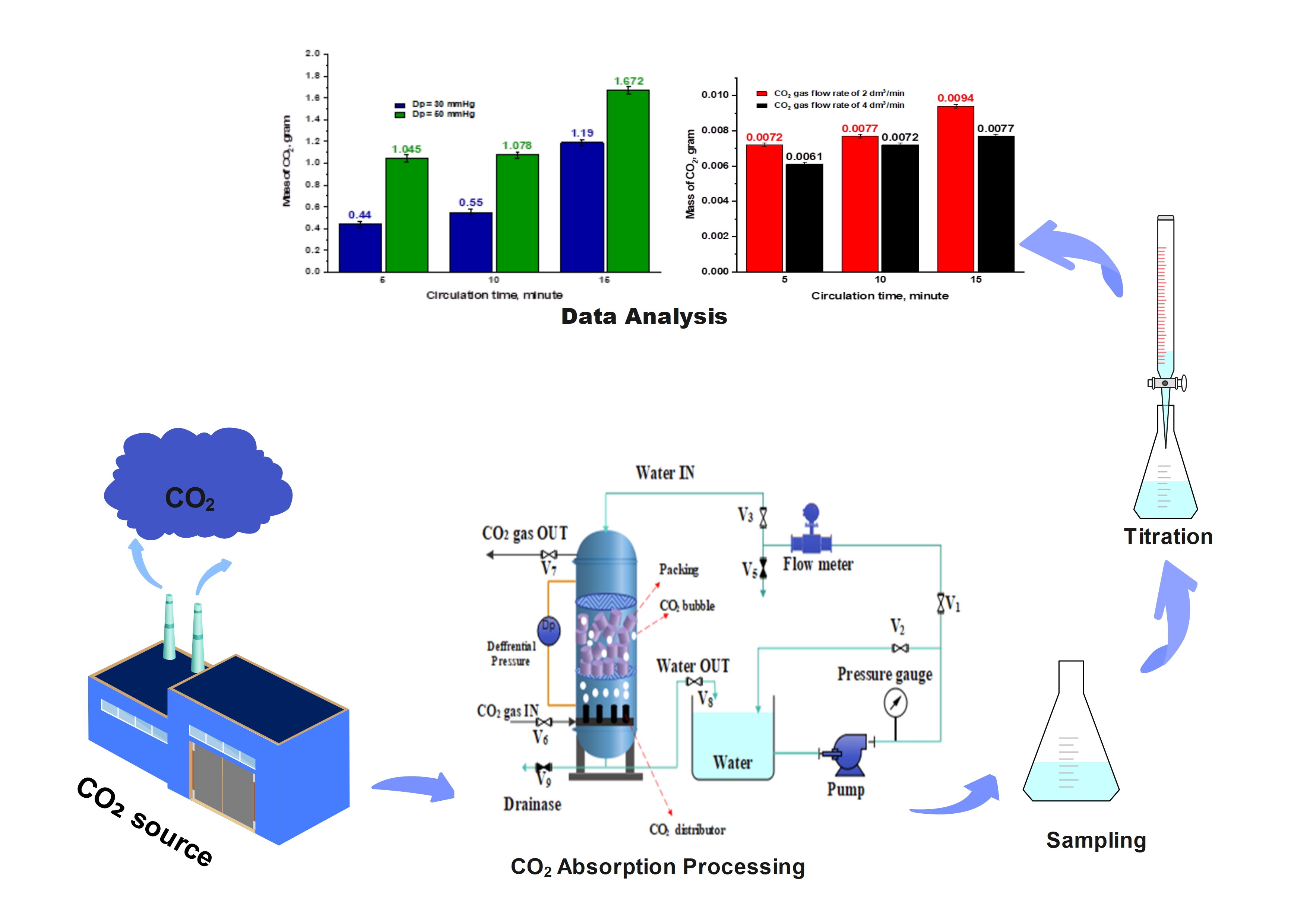
Downloads
References
A Al-lami and A Török. Decomposition of carbon dioxide (CO2) emissions in Hungary: A case study based on the kaya identity and LMDI model. Peri¬odica Polytechnica Transportation Engineering 2025; 53(1), 7-15.
H Jurikova, C Garbelli, R Whiteford, T Reeves, GM Laker, V Liebetrau and L Angiolini. Rapid rise in atmospheric CO2 marked the end of the Late Palaeozoic Ice Age. Nature Geoscience 2025; 18(1), 91-97.
TY Chen and WSW Ho. Effects of pressure and temperature on CO2 facilitation of amino acid salt-containing membranes for post-combustion car¬bon capture. Journal of Membrane Science 2024; 689, 122166.
D Lai, C Luo, Z Wang, Z Shi, T Luo, L Zhang and Z Zhang. Process simulation and techno-economic analysis of CO2 capture by coupling calcium looping with concentrated solar power in coal-fired power plant. Separation and Purification Technology 2025; 358, 130228.
L Yan, Y Liu, K Li, C Geng and B He. Insights into radiation property prediction for numerical simulation of pulverized coal/biomass oxy-fuel combustion. Clean Energy 2025; 9(2), 111-123.
R Krishna and JM van Baten. Non-Idealities in adsorption thermodynamics for CO2 capture from humid natural gas using CALF-20. Separation and Purification Technology 2025; 355, 129553.
F Amirkhani, A Dashti, H Abedsoltan, AH Mohammadi, JL Zhou and A Altaee. Modeling and estimation of CO2 capture by porous liquids through machine learning. Separation and Purification Technology 2025; 359, 130445.
R Gou, KK Wolf, CJ Hoppe, L Wu and G Lohmann. The changing nature of future arctic marine heatwaves and its potential impacts on the ecosystem. Nature Climate Change 2025; 15(2), 162-170.
RY Chan, YZ Zeng, CC Hou, HC Kou and HW Huang. Experimental study of carbon dioxide capture and mineral carbonation using sodium hydroxide solution. Journal of Ecological Engineering 2025; 26(1), 30-45.
JM Monteagudo, A Durán, M Alonso and AI Stoica. Investigation of effectiveness of KOH-activated olive pomace biochar for efficient direct air capture of CO2. Separation and Purification Technology 2025; 352, 127997.
M Ammar, T Dambrauskas, S Parvin, DA Gonzalez‐Casamachin, K Baltakys and J Baltrusaitis. In situ analysis of magnesium, copper, and zinc ammonium carbonate thermal properties. Journal of Thermal Analysis and Calorimetry 2024; 150(3), 1557-1569.
I Raheem, A Tawai, S Amornraksa, M Sriariyanun, A Joshi, M Gupta and SK Maity. A comprehensive review of approaches in carbon capture, and utilization to reduce greenhouse gases. Applied Science and Engineering Progress 2025; 18(2), 7629-7629.
HS Park, JY Kim, HJ Yang, Y Chung, J Na and NH Hur. Utilization of carbon dioxide and nitrate to produce sodium bicarbonate through a nitrate hydrogenation method. Journal of CO2 Utilization 2025; 94, 103060.
N Nuryoto, MH Alfarizi, MAS Kelana and RNT Bagaskara. Alternative technology towards clean and sustainable industry: Conversion of carbon dioxide gas into potassium carbonate. Advances in Science and Technology Research Journal 2025; 19(4), 183-197.
TM Thiedemann and M Wark. A compact review of current technologies for carbon capture as well as storing and utilizing the captured CO2. Processes 2025; 13(1), 283.
CH Yu, CH Huang and CS Tan. A review of CO2 capture by absorption and adsorption. Aerosol and Air Quality Research 2012; 12(5), 745-769.
S Kontsevoi. Decomposition mechanism and dissociation constants of bicarbonate ions. Water and Water Purification Technologies. Scientific and Technical News 2023; 35(1), 3-8.
RM Rivera and T van Gerven. Production of calcium carbonate with different morphology by simultaneous CO2 capture and mineralisation. Journal of CO2 Utilization 2020; 41, 101241.
AAH Mourad, AF Mohammad, M Altarawneh, AH Al‐Marzouqi, MH El‐Naas and MH Al‐Marzouqi. Effects of potassium hydroxide and aluminum oxide on the performance of a modified solvay process for CO2 capture: A comparative study. International Journal of Energy Research 2021; 45(9), 13952-13964.
O Gutierrez-Sanchez, B De Mot, N Daems, M Bulut, J Vaes, D Pant and T Breugelmans. Electrochemical conversion of CO2 from direct air capture solutions. Energy & Fuels 2022; 36(21), 13115-13123.
S Ghaffari, MF Gutierrez, A Seidel-Morgenstern, H Lorenz and P Schulze. Sodium hydroxide-based CO2 direct air capture for soda ash production - fundamentals for process engineering. Industrial & Engineering Chemistry Research 2023; 62(19), 7566-7579.
S Khan, L Navalgund, K Joshi and A Kumar. Design and fabrication of a system to capture ambient CO2. Journal of Air Pollution and Health 2023; 8(4), 399-410.
L Li, H Yu, L Ji, S Zhou, V Dao, P Feron and E Benhelal. Integrated CO2 capture and mineralization approach based on KOH and cement-based wastes. Journal of Environmental Chemical Engineering 2024; 12(5), 113382.
F Tzirakis, LA Diaz, I Chararas, DMM de Oca, Z Zhao, P Seferlis and AI Papadopoulos. Selection of solvents for integrated CO2 absorption and electrochemical reduction systems. AIChE Journal 2025; 71(5), e18734.
X Yu, H Wu, W Li and H Yang. Emission characteristics of ethanolamine and ammonia in CO2 capture process by chemical absorption based on ethanolamine solution. Journal of Chemical Technology & Biotechnology 2025; 100(4), 688-696.
H Asgharian, F Iov, MP Nielsen, V Liso, S Burt and L Baxter. Analysis of cryogenic CO2 capture technology integrated with water-ammonia absorption refrigeration cycle in cement plants. Separation and Purification Technology 2025; 353, 128419.
AA Ujile. Mass transfer, absorption. In: AJ Kehinde (Ed.). Chemical engineering unit operation, synthesis and basic design calculation. 1st ed. Bomn Prints, Ibadan, Nigeria, 2014.
O Levenspiel. Chemical reaction engineering. 3rd ed. John Wiley & Sons, New York, 1998.
JM Smith, HC Van Ness, MM Abbott and MT Swihart. Introduction to chemical engineering thermodynamics. 8th ed. McGraw-Hill Education, Ohio, United States, 2018.
N Nuryoto, N Mas’ulunniah, AS Choerunnisa and S Suripno. Pemanfaatan karbon dioksida untuk sintesis precipitated calcium carbonate (PCC) dengan metode karbonasi (in Indonesian). Jurnal Integrasi Proses 2021; 10(2), 90-95.
Z Rastegar and A Ghaemi. CO2 absorption into potassium hydroxide aqueous solution: Experimental and modeling. Heat and Mass Transfer 2022; 58(3), 365-381.
TJ Kang, JH Lee, DH Lee, HS Kim and SH Kang. Effect of high temperature on CO2 gasification kinetics of sub-bituminous coal fly ash. Sustainability 2025; 17(4), 1519.
A Elshani, K Pehlivani, B Kelmendi and I Cacaj. Possibility and determination of the use of CO2 produced by the production of beers. Journal of Pharmaceutical Science and Reearch 2018; 10(5), 1229-30.
A Sumayli, Z Ahmed, V Jain, R Roopashree, A Kumar, A Kashyap and MK Abosaoda. Computational evaluation of micropores wetting effect on CO2 removal through membrane contactor. Scientific Reports 2025; 15(1), 780.
LL Wang. Study on effect of gas-liquid 2 phase physical feature on slug flow in microchannels. Frontiers in Physics 2023; 11, 1125220.
MI Alhamid and MB Perdana. Effect of methane gas flow rate on adsorption capacity and temperature distribution of activated carbon. International Journal of Technology 2015; 6(4), 584-593.
M Bagi, MV Razlighi, M Shanbedi and A Karim. Parametric study on CO2 removal from natural gas by hollow fiber membrane contactor: A CFD approach. Chemical Engineering & Technology 2024; 47(4), 732-738.
AC Bozonc, AM Cormos, S Dragan, C Dinca and CC Cormos. Dynamic modeling of CO2 absorption process using hollow-fiber membrane contactor in MEA solution. Energies 2022; 15(19), 7241.
HS Rambe, I Irvan, B Trisakti, T bin Nur, MS Cahyono and MN Aridito. CO2 absorption using aqueous potassium carbonate (K2CO3) promoted by piperazine (PZ) in packed column reactor. IOP Conference Series: Earth and Environmental Science 2024; 1352(1), 012024.
AM Abou-Elanwar, S Lee, I Jang, S Lee, S Hong, S Kim and Y Kim. Fertilizer-drawn forward osmosis and membrane contactor system for CO2 capture using ammonia-rich wastewater. Journal of Membrane Science 2024; 700, 122654.
NT Abdullah and LK Shakir. Efficient carbon dioxide capture in packed columns by solvents blend promoted by chemical additives. Journal of Ecological Engineering 2024; 25(10), 1-15.
YA Cengel, MA Boles and M Konoglu. Thermodynamics: An engineering approach. 9th ed. McGraw-Hill Education, Ohio, United States, 2019.
H Yang, X Hou, L Jin, H Yu, W Li, S Kong and L Yang. Energy-efficient CO2 capture using low-viscosity water-lean aromatic amine solvent: Performance, mechanism and thermodynamics. Separation and Purification Technology 2025; 359, 130391.
XYD Soo, JJC Lee, WY Wu, L Tao, C Wang, Q Zhu and J Bu. Advancements in CO2 capture by absorption and adsorption: A comprehensive review. Journal of CO2 Utilization 2024; 81, 102727.
J Kotowicz, K Niesporek and O Baszczeńska. Advancements and challenges in direct air capture technologies: Energy intensity, novel methods, economics, and location strategies. Energies 2025; 18(3), 496.
V Barahimi, M Ho and E Croiset. From lab to fab: Development and deployment of direct air capture of CO2. Energies 2023; 16(17), 6385.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






