Evaluation of Calcium Oxide (CaO) from Oyster Shell Waste: A Sustainable Heat-Generating and Antimicrobial Agent
DOI:
https://doi.org/10.48048/tis.2026.11055Keywords:
Antimicrobial, Calcination, Calcium oxide, Exothermic, Oyster shellAbstract
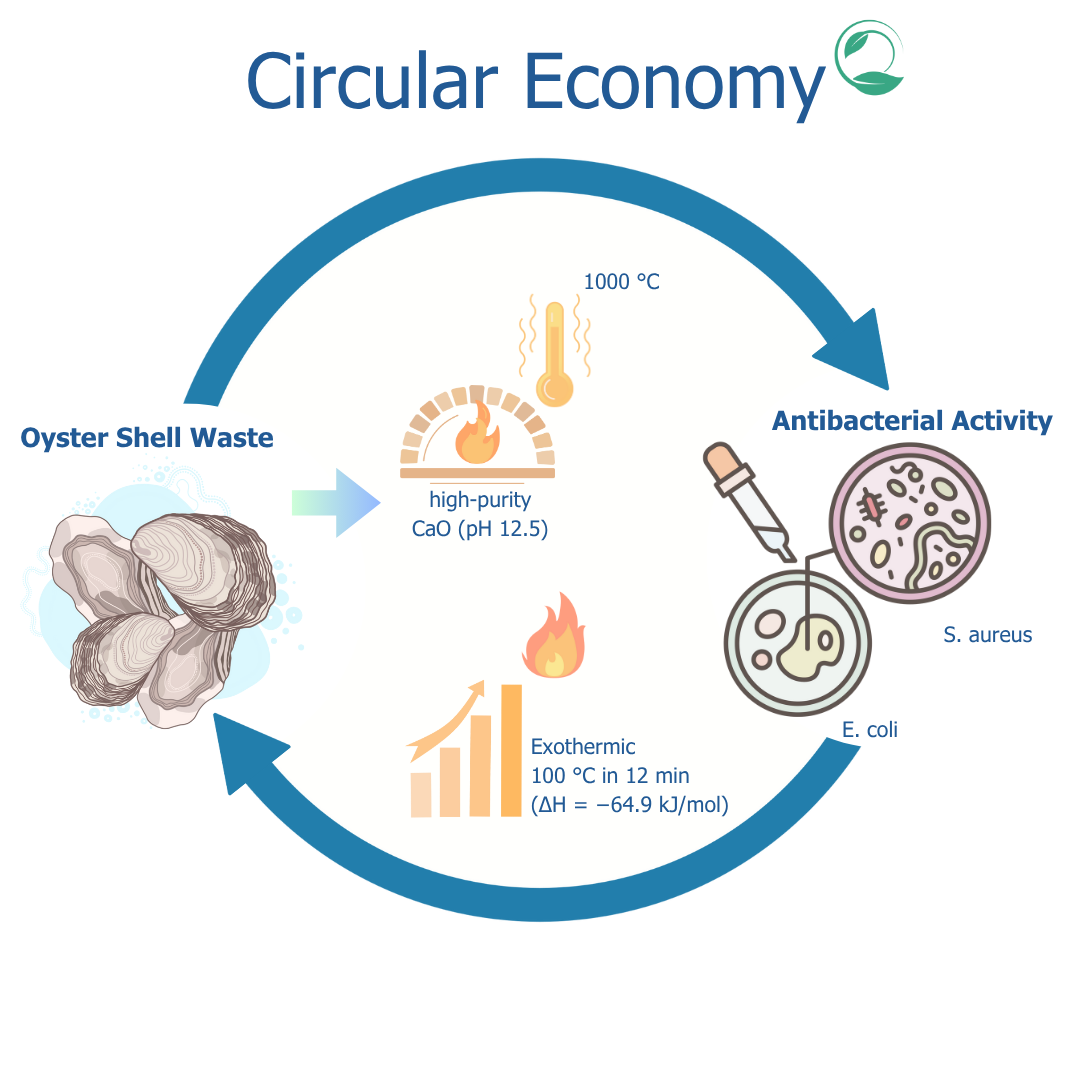
Oyster shell waste is a source of calcium carbonate (CaCO3) that has the potential to be processed into calcium oxide (CaO) through a calcination process. CaO has various applications, including as a heat-generating agent and antimicrobial. However, proper calcination temperature optimization is needed to obtain CaO with optimal physical and chemical properties. This study aims to optimize CaO production from oyster shell waste and to produce its potential in exothermic and antimicrobial applications The method used involves a calcination process at temperatures of 750, 900, and 1000 °C. The CaO produced was characterized at 750, 900, and 1000 °C. CaO characterization was carried out using Scanning Electron Microscopy (SEM) for morphological analysis, X-Ray Diffraction (XRD) for crystallinity, Fourier Transform Independent Spectroscopy (FTIR) for functional groups, and thermogravimetry (TGA) for thermal stability. The pH test was carried out to determine the basicity, while the antibacterial test used the disc diffusion method against Escherichia coli and Staphylococcus aureus. The results showed that calcination at 1000 °C produced CaO with high purity and porous structure that increased the surface area. The pH stability in the range of 12.5 indicated strong basic properties. CaO produced at 1000 °C exhibited significant exothermic reaction when reacting with air, proving its potential as a heat generating agent (T = 100 °C at 12 min with ∆H = −64.9 kJ/mol). It is also revealed great inhibition against E. coli (1.32 mm) and S. aureus (1.36 mm). In conclusion, oyster shell waste-derived CaO is a highly promising sustainable solution for both heat generation and antimicrobial applications, which strongly supports a circular economy approach in biogenic waste management.
HIGHLIGHTS
- Oyster shell waste is successfully utilized as a sustainable raw material to produce high-purity calcium oxide (CaO).
- Optimized calcination at 1000 °C yielded CaO with a porous structure and high basicity (pH 12.5), ideal for industrial applications.
- Produced CaO demonstrated strong exothermic properties (ΔH = −64.9 kJ/mol), reaching 100°C in 12 min upon exposure to air.
- Exhibited significant antibacterial activity against coli (1.32 mm) and S. aureus (1.36 mm).
- Supports circular economy by converting biogenic waste into valuable multifunctional materials.
GRAPHICAL ABSTRACT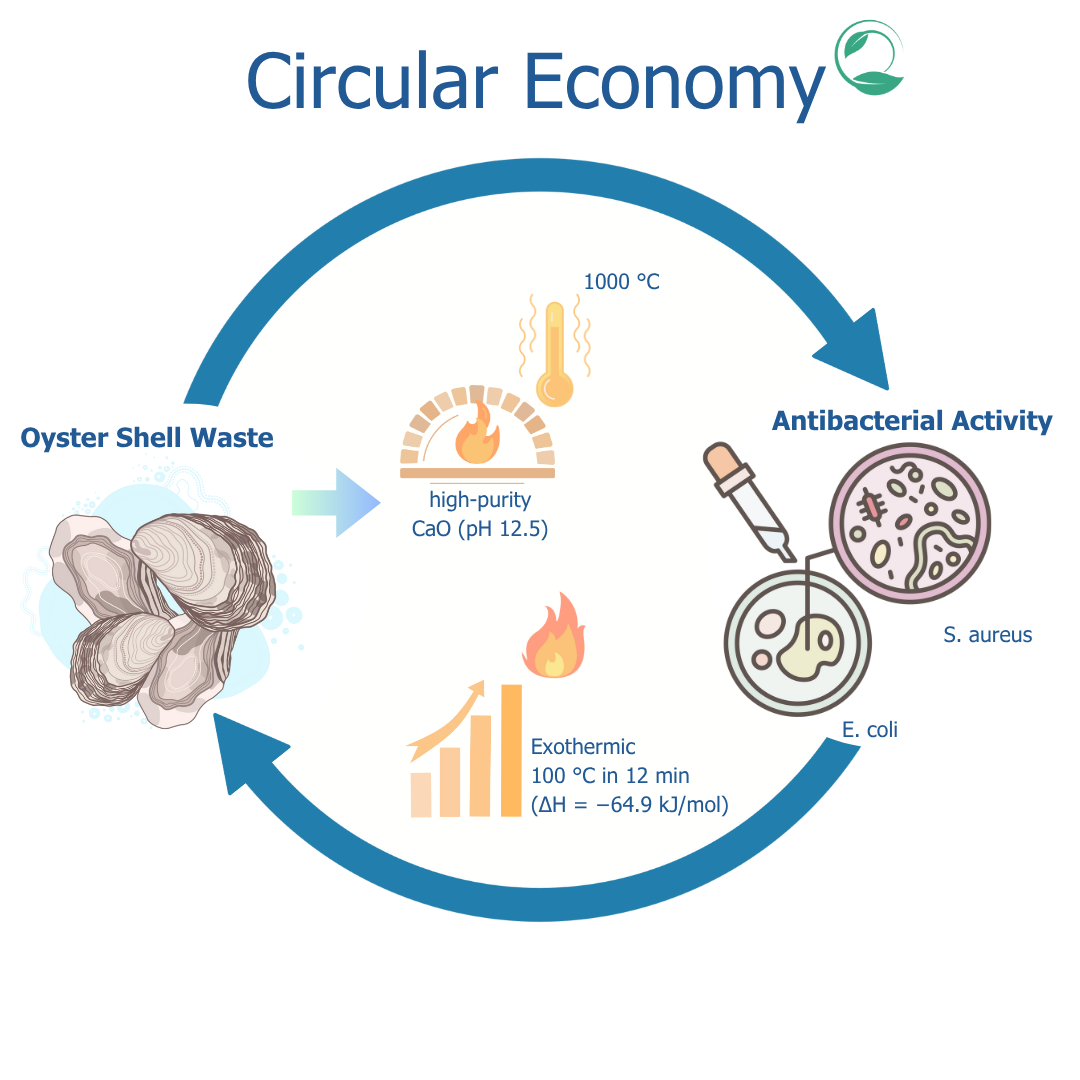
Downloads
References
S Ulagesan, S Krishnan, TJ Nam and YH Choi. A review of bioactive compounds in oyster shell and tissues. Frontiers in Bioengineering and Biotechnology 2022; 10, 913839.
C Xu, R Liu and L Chen. Removal of phosphorus from domestic sewage in rural areas using oyster shell-modified agricultural waste-rice husk biochar. Processes 2023; 11(9), 2577.
S Tongwanichniyom, T Pattamapitoon, N Sangvichien and S Phornphisutthimas. Production of calcium oxide from waste oyster shells for a value-added application of antibacteria. Ecology, Environment and Conservation 2021; 27(2), 539-547.
R Botta, F Asche, JS Borsum and EV Camp. A review of global oyster aquaculture production and consumption. Marine Policy 2020; 117, 103952.
R Shobana, S Vijayalakshmi, B Deepanraj and J Ranjitha. Biodiesel production from Capparis spinosa L seed oil using calcium oxide as a heterogeneous catalyst derived from oyster shell. Materials Today: Proceedings 2023; 80(3), 3216-3220.
L Wang, Y Liu, X Peng, Y Sun, X Liu, H Liu, Q Lin, H Sun, B Yang and X Li. Preparation and characterization of CaO/ZnO core-shell structured nanoparticles. Chemical Research in Chinese Universities 2020; 36, 970-975.
S Kumar, V Sharma, JK Pradhan, SK Sharma, P Singh and JK Sharma. Structural, optical and antibacterial response of CaO nanoparticles synthesized via direct precipitation technique. Nano Biomedicine and Engineering 2021; 13(2), 172-178.
G Marquis, B Ramasamy, S Banwarilal and AP Munusamy. Evaluation of antibacterial activity of plant mediated CaO nanoparticles using Cissus quadrangularis extract. Journal of Photochemistry and Photobiology B 2016; 155, 28-33.
AF Rusdaryanti, U Amalia and S Suharto. Antibacterial activity of CaO from blood cockle shells (Anadara granosa) calcination against Escherichia coli. Biodiversitas 2020; 21(6), 2827-2831.
W Srichanachaichok and D Pissuwan. Micro/nano structural investigation and characterization of mussel shell waste in Thailand as a feasible bioresource of CaO. Materials 2023; 16(2), 805.
K Zheng, F Bider, M Monavari, Z Xu, C Janko, C Alexiou, AM Beltrán and AR Boccaccini. Sol-gel derived B2O3-CaO borate bioactive glasses with hemostatic, antibacterial and pro-angiogenic activities. Regenerative Biomaterials 2024; 11, rbad105.
GN Mediarman, Sumardianto, PH Riyadi, L Rianingsih and L Purnamayati. Potentials of CaO powder result of calcination from green shells (Perna viridis), scallops (Placuna placenta), and blood clams (Anadara granosa) as antibacterial agent. In: Proceedings of the 2nd International Conference on Fisheries and Marine, Ternate, Indonesia. 2021.
X Liang, R Dai, S Chang, Y Wei and B Zhang. Antibacterial mechanism of biogenic calcium oxide and antibacterial activity of calcium oxide/polypropylene composites. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022; 650, 129446.
X Meng, Z Xu, C Wang, J Patitz, AR Boccaccini, A Burkovski and K Zheng. Surface engineering of mesoporous bioactive glass nanoparticles with bacteriophages for enhanced antibacterial activity. Colloids and Surfaces B: Biointerfaces 2024; 234, 113714.
SH Chang, YJ Chen, HJ Tseng, HI Hsiao, HJ Chai, KC Shang, CL Pan and GJ Tsai. Antibacterial activity of chitosan-polylactate fabricated plastic film and its application on the preservation of fish fillet. Polymers 2021; 13(5), 696.
J Widakdo, TM Chen, MC Lin, JH Wu, TL Lin, PJ Yu, WS Hung and KR Lee. Evaluation of the antibacterial activity of eco-friendly hybrid composites on the base of oyster shell powder modified by metal ions and LLDPE. Polymers 2022; 14, 3001.
M Manoj, O Manaf, KM Ismayil and A Sujith. Composites based on poly(ethylene-co-vinyl acetate) and silver-calcined scallop shell powder: Mechanical, thermal, photocatalytic, and antibacterial properties. Journal of Elastomers and Plastics 2021; 53(7), 902-921.
YC Chen, CL Lin, CT Li and DF Hwang. Structural transformation of oyster, hard clam, and sea urchin shells after calcination and their antibacterial activity against foodborne microorganisms. Fisheries Science 2015; 81, 787-794.
SS Karkal, DR Rathod, AS Jamadar, PV Suresh, HNP Kumar and TG Kudre. Fenneropeanus indicus shrimp shell and fishmeal oil: A novel feedstock for biodiesel production and bio derived heterogeneous catalyst development. Catalysis Letters 2024; 154, 1521-1536.
M Mahidin, A Gani, MR Hani, M Syukur, H Hamdani, K Khairil, S Rizal, A Hadi and TMI Mahlia. Use of green mussel shell as a desulfurizer in the blending of low rank coal-biomass briquette combustion. Makara Journal of Technology 2016; 20, 97-102.
SFS Mohamad, S Mohamad and Z Jemaat. Study of calcinations condition on decomposition of calcium carbonate in waste cockle shell to calcium oxide using thermal gravimetric analysis. ARPN Journal of Engineering and Applied Sciences 2016; 11(16), 9917-9921.
YS Liu, KX Tang, YT Liao, RR Huang, Y Zhu, Z Chen, Y Chen and L Lin. Calcined and hydrated shell powder with layered porous structures for food sterilization and pesticide residue removal. In: Proceedings of the Chinese Materials Conference 2022-2023, Shenzhen, China. 2023.
WC Lu, CS Chiu, CW Hsieh, YJ Chan, ZC Liang, CCR Wang, AT Mulio, DHT Le and PH Li. Calcined oyster shell powder as a natural preservative for maintaining quality of white shrimp (Litopenaeus vannamei). Biology 2022; 11, 11020334.
Y Hata, S Hiruma, H Miyazaki and S Nakamura. Sequential approach for water purification using seashell-derived calcium oxide through disinfection and flocculation with polyphosphate for chemical pollutant removal. ACS Omega 2023; 9(11), 12635-12642.
S Dampang, E Purwanti, F Destyorini, SBKB Kurniawan, SRS Abdullah and MF Imron. Analysis of optimum temperature and calcination time in the production of CaO using seashells waste as CaCO3 source. Journal of Ecological Engineering 2021; 22(5), 221-228.
A Bucur, R Banica, MC Pascariu, M Poienar, C Mosoarca, R Bucur, A Negreaa and I Hulka. Eco-valorification of marine shells by hydrothermal conversion in alkaline media. Digest Journal of Nanomaterials and Biostructures 2022; 17(1), 153-160.
SC Wu, HC Hsu, SK Hsu, CP Tseng and WF Ho. Effects of calcination on synthesis of hydroxyapatite derived from oyster shell powders. Journal of the Australian Ceramic Society 2019; 55, 1051-1058.
N Suwannasingha, A Kantavong, S Tunkijjanukij, C Aenglong, HB Liu and W Klaypradit. Effect of calcination temperature on structure and characteristics of calcium oxide powder derived from marine shell waste. Journal of Saudi Chemical Society 2022; 26, 101441.
S Suprihadi, A Thaib, N Nurhayati and L Handayani. The potential of fishery waste as an alternative source of natural calcium: A review. Acta Aquatica: Aquatic Sciences Journal 2023; 10, 163-171.
Q Zhou, B Wu, K Chen, L Ji and Y Wu. Study on thermal decomposition kinetic mechanism and calcination process of phosphorus tailings. Inorganic Chemicals Industry 2023. https://doi.org/10.19964/j.issn.1006-4990.2022-0295
SAC Hockaday, F Dinter and QG Reynolds. The thermal decomposition kinetics of carbonaceous and ferruginous manganese ores in atmospheric conditions. Journal of the Southern African Institute of Mining and Metallurgy 2023; 123(8), 391-398.
Q Yin, H Song, M Xu, H Yan, Y Zhao and X Duan. Thermal decomposition of carbonates coupled with dry reforming of methane to synthesize high-value products: A perspective (in Chinese). Acta Physica Sinica 2023; 39(3), 2210026.
S Seesanong, Y Wongchompoo, B Boonchom, C Sronsri, N Laohavisuti, K Chaiseeda and W Boonwee. Economical and environmentally friendly track of biowaste recycling of scallop shells to calcium lactate. ACS Omega 2022; 7(17), 14756-14764.
M Olszak-Humienik and M Jablonski. Thermal behavior of natural dolomite. Journal of Thermal Analysis and Calorimetry 2015; 119, 2239-2248.
J Zheng, J Huang, L Tao, Z Li and Q Wang. A multifaceted kinetic model for the thermal decomposition of calcium carbonate. Crystals 2020; 10(9), 849.
R Akasaka, A Osawa, R Wada, J Sawai and Y Nakagawa. Antimicrobial activity and transparency of polyvinyl butyral paint containing heated scallop-shell powder. Coatings 2023; 13(2), 364.
N Tangboriboon, R Kunanuruksapong and A Sirivat. Preparation and properties of calcium oxide from eggshells via calcination. Materials Science-Poland 2012; 30, 313-322.
MS Sankar, P Dash, YH Lu, X Hu, AE Mercer, S Wickramarathna, WT Beshah, SL Sanders, Z Arslan, J Dyer and RJ Moorhead. Seasonal changes of trace elements, nutrients, dissolved organic matter, and coastal acidification over the largest oyster reef in the Western Mississippi Sound, USA. Environmental Monitoring and Assessment 2023; 195(1), 175.
S Seesanong, C Seangarun, B Boonchom, N Laohavisuti, W Boonmee, S Thompho and P Rungrojchaipon. Low-cost and eco-friendly calcium oxide prepared via thermal decompositions of calcium carbonate and calcium acetate precursors derived from waste oyster shells. Materials 2024; 17, 3875.
S Seesanong, C Laosinwattana, K Chaiseeda and B Boonchom. A simple and rapid transformation of golden apple snail (Pomacea canaliculata) shells to calcium carbonate, monocalcium and tricalcium phosphates. Asian Journal of Chemistry 2019; 31(11), 2522-2526.
PSCD Silva, WDM Farias, MRBP Gomez, JK Torrecilha, FR Rocha, MA Scapin, RHL Garcia, LRLD Simone and VSD Amaral. Oyster shell element composition as a proxy for environmental studies. Journal of South American Earth Sciences 2024; 134, 104749.
M Sari and Y Yusuf. Synthesis and characterization of hydroxyapatite based on green mussel shells (Perna viridis) with calcination temperature variation using the precipitation method. International Journal of Nanoelectronics and Materials 2018; 11(3), 357-370.
JH Seo, SM Park, BJ Yang and JG Jang. Calcined oyster shell powder as an expansive additive in cement mortar. Materials 2019; 12(8), 1322.
X Yang, K Liu, Y Wen, Y Huang and C Zheng. Application of natural and calcined oyster shell powders to improve latosol and manage nitrogen leaching. International Journal of Environmental Research and Public Health 2023; 20(5), 3919.
MH Azarian and W Sutapun. Biogenic calcium carbonate derived from waste shells for advanced material applications: A review. Frontiers in Materials 2022; 9, 1024977.
B Janković, N Manić, M Jović and I Smičiklas. Kinetic and thermodynamic analysis of thermo-oxidative degradation of seashell powders with different particle size fractions: Compensation effect and iso-equilibrium phenomena. Journal of Thermal Analysis and Calorimetry 2022; 147, 2305-2334.
E Brakemi, K Michael, SP Tan and H Helen. Antimicrobial activity of natural mollusc shells: A review. Process Biochemistry 2024; 137, 122-133.
J Zhang, B Zheng, C Zhang, L Xie and C Fang. Calcined waste shells as a promising, eco-friendly adsorbent, antimicrobial, food preservative, and food packaging material: A mini review. Journal of Food Process Engineering 2023; 46(12), e14477.
S Lin, M Harada, Y Suzuki and H Hatano. CaO hydration rate at high temperature (∼1023 K). Energy and Fuels 2006; 20(3), 903-908.
Y Fang, J Zhao, C Zhang and Y Li. Exothermic performance of the calcined limestone determined by exothermic temperature under fluidization during CaCO3/CaO energy storage cycles. Journal of Thermal Science 2023; 32, 1784-1796.
U Ryde. A fundamental view of enthalpy-entropy compensation. MedChemComm 2014; 5, 1324-1336.
S Kuo, MS Lai and CW Lin. Influence of solution acidity and CaCl2 concentration on the removal of heavy metals from metal-contaminated rice soils. Environmental Pollution 2006; 144(3), 918-925.
K Sadeghi, K Park and J Seo. Oyster shell disposal: Potential as a novel ecofriendly antimicrobial agent for packaging: A mini review. Korean Journal of Packaging Science and Technology 2019; 25(2), 57-62.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






