Optimization of Solvent Systems and Extraction Techniques for Enhanced Multifunctional Bioactivities of Vernonia Cinerea (L.) Less.: A Comprehensive Study on Antioxidant, Tyrosinase Inhibitory, Antibacterial, and Antidiabetic Enzyme Activities
DOI:
https://doi.org/10.48048/tis.2026.11007Keywords:
Vernonia cinerea (L.) Less., Total Phenolic Content, Antioxidant, Tyrosinase Inhibitory, Antibacterial, Anti-α-amylase Activity, Anti-α-glucosidase Activity, Vernonia cinerea (L.) Less., Total phenolic content, Antioxidant, Tyrosinase inhibitory, Antibacterial, Anti-α-amylase Activity, Anti-α-glucosidase ActivityAbstract
The objective of the study was to identify the suitable condition of solvent and an effective extraction technique for improving the biological activity of the extracts of Vernonia cinerea plant, focusing on free radical scavenging, tyrosinase and inhibition, bacterium inhibition, α-amylase and α-glucosidase inhibition activity. The sample from leaves, flowers, and stems of the plants was extracted with 5 types of solvent in the present study. The efficacy of biological activity was evaluated in terms of total phenolic and flavonoid content, DPPH and FRAP scavenging activity, tyrosinase inhibition activity, bacterium inhibition, agar well diffusion assay, and inhibition of α-amylase and α-glucosidase. The results showed that the MAE method with methanol solvent was the most effective in all tests. The extracts from the leaf exhibited maximum biological activity with the highest TPC and TFC of 195.48 ± 4.18 mg GAE/g extract and 98.07 ± 0.33 mg QE/g extract, respectively, IC50 values for DPPH and FRAP was 45.02 ± 0.13 µg/mL and 169.91 ± 0.51 mg FeSO4/g extract, respectively, IC50 value for tyrosinase inhibition was 18.48 ± 0.19 mg/mL, which was approximate inhibition as kojic acid, bacterial inhibition for Staphylococcus aureus, Bacillus cereus, and Salmonella typhimurium was maximum in 12.19 ± 0.01 mm, 13.03 ± 0.01 mm and 12.03 ± 0.02 mm, against α-amylase and α-glucosidase inhibition was 73.76% and 63.61% which was approximately 96.5% and 93.5% of market acarbose, respectively. The study evidenced that the MAE with methanol is an effective method for extracting multitasking active ingredient from the V. cinerea at once in the future when plants have the highest potential.
HIGHLIGHTS
- Microwave-assisted extraction (MAE) with methanol yielded superior efficiency compared to fermentation and ultrasonic extraction.
- Vernonia cinerea leaf extract exhibited higher concentrations of total phenolic compounds and total flavonoids than flower and stem extracts.
- Vernonia cinerea leaf extract exhibited biological activities including free radical scavenging, tyrosinase inhibition, α-amylase inhibition, and α-glucosidase inhibition.
GRAPHICAL ABSTRACT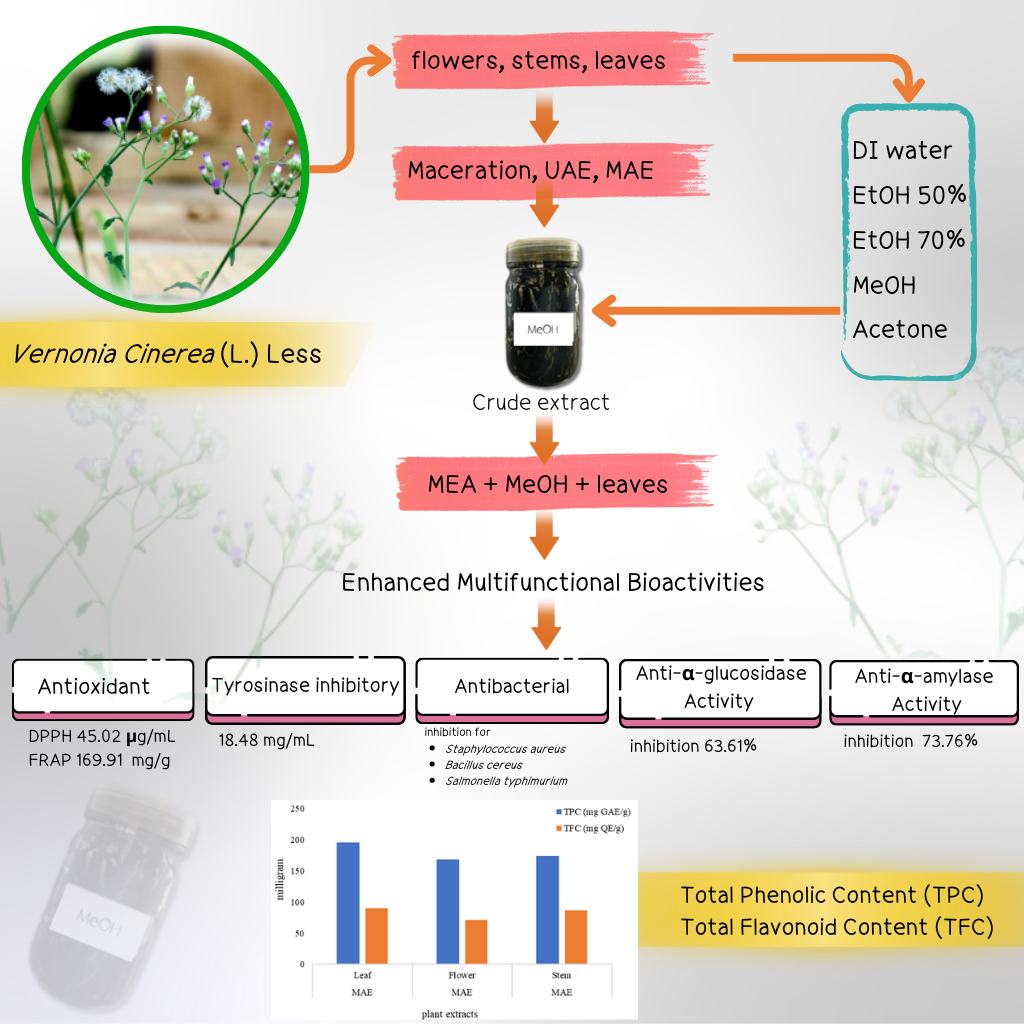
Downloads
References
V Lobo, A Patil, A Phatak and N Chandra. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacognosy Reviews 2010; 4(8), 118-126.
L Panzella and A Napolitano. Natural and bioinspired phenolic compounds as tyrosinase inhibitors for the treatment of skin hyperpigmentation: Recent advances. Cosmetics 2019; 6(4), 57.
Department of Thai Traditional and Alternative Medicine. Standard herbal list: Thai herbal pharmacopoeia. Ministry of Public Health, Bangkok, Thailand, 2014.
B Srichaikul, H Theerasin and N Ruangrungsi. Phenolic contents and antioxidant capacity of different extracts of Vernonia cinerea (L.) less. Thai Journal of Pharmaceutical Sciences 2019; 43(1), 8-14.
L Pisano, M Turco and CT Supuran. Biomedical applications of tyrosinases and tyrosinase inhibitors. The Enzymes 2024; 56, 261-280.
MA Sonibare, OT Aremu and PN Okorie. Antioxidant and antimicrobial activities of solvent fractions of Vernonia cinerea (L.) less leaf extract. African Health Sciences 2016; 16(2), 629-639.
U Etxeberria, ALDL Garza, J Campión, JA Martinez and FI Milagro. Antidiabetic effects of natural plant extracts via inhibition of carbohydrate hydrolysis enzymes with emphasis on pancreatic alpha amylase. Current Pharmaceutical Design 2012; 16(3), 269-297.
OR Alara, A Nour, CI Ukaegbu and AH Nour. Vernonia cinerea leaves as the source of phenolic compounds, antioxidants, and anti-diabetic activity using microwave-assisted extraction technique. Industrial Crops and Products 2018; 122, 533-544.
NN Azwanida. A review on the extraction methods use in medicinal plants, principle, strength and limitation. Medicinal & Aromatic Plants 2015; 4(3), 1000196.
F Chemat, M Abert Vian and G Cravotto. Green extraction of natural products: Concept and principles. International Journal of Molecular Sciences 2012; 13(7), 8615-8627.
H López-Salazar, BH Camacho-Díaz, MLA Ocampo and AR Jiménez-Aparicio. Microwave-assisted extraction of functional compounds from plants: A review. Bioresources 2023; 18(3), 6614-6638.
M Vinatoru, TJ Mason and I Calinescu. Ultrasound-assisted extraction (UAE) and microwave-assisted extraction (MAE) of functional compounds from plant materials. TrAC Trends in Analytical Chemistry 2017; 97, 159-178.
V Mandal, Y Mohan and S Hemalatha. Microwave assisted extraction - An innovative and promising extraction tool for medicinal plant research. Pharmacognosy Reviews 2007; 1(1), 7-18.
AV Le, SE Parks, MH Nguyen and P Roach. Optimisation of the microwave-assisted ethanol extraction of saponins from Gac (Momordica cochinchinensis Spreng.) seeds. Medicines 2018; 5(3), 70.
VL Singleton and JA Rossi. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture 1965; 16(3), 144-158.
CC Chang, MH Yang, HM Wen and JC Chern. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. Journal of Food and Drug Analysis 2002; 10(3), 178-182.
W Brand-Williams, ME Cuvelier and C Berset. Use of a free radical method to evaluate antioxidant activity. LWT - Food Science and Technology 1995; 28(1), 25-30.
K Thaipong, U Boonprakob, K Crosby, L Cisneros-Zevallos and DH Byrne. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. Journal of Food Composition and Analysis 2006; 19(6-7), 669-675.
Y Masamoto, H Ando, Y Murata, Y Shimoishi, M Tada and K Takahata. Mushroom tyrosinase inhibitory activity of esculetin isolated from seeds of Euphorbia lathyris L. Bioscience, Biotechnology, and Biochemistry 2003; 67(3), 631-634.
M Balouiri, M Sadiki and SK Ibnsouda. Methods for in vitro evaluating antimicrobial activity: A review. Journal of Pharmaceutical Analysis 2017; 6(2), 71-79.
JH Jorgensen and MJ Ferraro. Antimicrobial susceptibility testing: A review of general principles and contemporary practices. Clinical Infectious Diseases 2009; 49(11), 1749-1755.
FJ Gella, G Gubern, R Vidal and F Canalias. Determination of total and pancreatic α-amylase in human serum with 2-chloro-4-nitrophenyl-α-D-maltotrioside as substrate. Clinica Chimica Acta 1997; 259(1-2), 147-160.
T Matsui, C Yoshimoto, K Osajima, T Oki and Y Osajima. In vitro survey of α-glucosidase inhibitory food components. Bioscience, Biotechnology, and Biochemistry 1996; 60(12), 2019-2022.
R Chirinos, H Rogez, D Campos, R Pedreschi and Y Larondelle. Optimization of extraction conditions of antioxidant phenolic compounds from mashua (Tropaeolum tuberosum Ruiz & Pavón) tubers. Separation and Purification Technology 2013; 55(2), 217-225.
A Pandey and SL Rizvi. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Medicine and Cellular Longevity 2009; 2(5), 270-278.
G Spigno, L Tramelli and DMD Faveri. Effects of extraction time, temperature and solvent on concentration and antioxidant activity of grape marc phenolics. Journal of Food Engineering 2007; 81(1), 200-208.
M Naczk and F Shahidi. Extraction and analysis of phenolics in food. Journal of Chromatography A 2004; 1054(1-2), 95-111.
G Spigno and DMD Faveri. Microwave-assisted extraction of tea phenols: A phenomenological study. Journal of Food Engineering 2009; 93(2), 210-217.
PC Veggi, J Martinez and MA Meireles. Fundamentals of microwave extraction. In: F Chemat and G Cravotto (Eds.). Microwave-assisted extraction for bioactive compounds. Springer, New York, 2013, p. 15-52.
MP Sonar and VK. Rathod. Microwave assisted extraction (MAE) used as a tool for rapid extraction of Marmelosin from Aegle marmelos and evaluations of total phenolic and flavonoids content, and antioxidant and anti-inflammatory activity. Chemical Data Collections 2020; 30, 100545.
D Tungmunnithum, A Thongboonyou, A Pholboon and A Yangsabai. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018; 5(3), 93.
MG Miguel. Antioxidant activity of medicinal and aromatic plants. A review. Flavour and Fragrance Journal 2011; 25(5), 291-312.
A Ghasemzadeh and N Ghasemzadeh. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. Journal of Medicinal Plants Research 2011; 5(31), 6697-6703.
T Mol, E Grotewold and R Koes. How genes paint flowers and seeds. Trends in Plant Science 1998; 3(6), 212-217.
C Proestos and M Komaitis. Application of microwave-assisted extraction to the fast extraction of plant phenolic compounds. LWT-Food Science and Technology 2008; 41(4), 652-659.
QD Do, AE Angkawijaya, PL Tran-Nguyen, LH Huynh, FE Soetaredjo, S Ismadji and YH Ju. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. Journal of Food and Drug Analysis 2014; 22(3), 296-302.
J Dai and RJ Mumper. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010; 15(10), 7313-7352.
A Wojdylo, J Oszmianski and R Czemerys. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chemistry 2007; 105(3), 940-949.
PG Pietta. Flavonoids as antioxidants. Journal of Natural Products 2000; 63(7), 1035-1042.
TK Lim. Edible medicinal and non-medicinal plants: Volume 3, fruits. Springer, Dordrecht, Netherlands, 2012.
M Zakaria, K Simpson, PR Brown and A Krstulovic. Use of reversed-phase high-performance liquid chromatographic analysis for the determination of provitamin A carotenes in tomatoes. Journal of Chromatography A 1979; 176(1), 109-117.
S Surveswaran, YZ Cai, H Corke and M Sun. Systematic evaluation of natural phenolic antioxidants from 133 Indian medicinal plants. Food Chemistry 2007; 102(3), 938-953.
T Belwal, A Pandey, ID Bhatt, RS Rawal and V Pande. Optimized microwave assisted extraction (MAE) of alkaloids and polyphenols from Berberis roots using multiple-component analysis. Scientific Reports 2020; 10, 917.
OR Alara, NH Abdurahman, CI Ukaegbu and NH Azhari. Microwave-assisted extraction of antioxidant compounds using natural deep eutectic solvents: A sustainable approach for valorization of hazelnut pomace. Journal of Environmental Chemical Engineering 2022; 10(2), 107267.
P Kaur, A Chaudhary, B Singh and Gopichand. An efficient microwave assisted extraction of phenolic compounds and antioxidant potential of Ginkgo biloba. Natural Product Communications 2012; 7(2), 203-206.
I González-Palma, HB Escalona-Buendía, E Ponce-Alquicira, M Téllez-Téllez, VK Gupta, G Díaz-Godínez and J Soriano-Santos. Evaluation of the antioxidant activity of aqueous and methanol extracts of Pleurotus ostreatus in different growth stages. Frontiers in Microbiology 2016; 7, 1099.
NS Rajurkar and SM Hande. Estimation of phytochemical content and antioxidant activity of some selected traditional Indian medicinal plants. Indian Journal of Pharmaceutical Sciences 2011; 73(2), 146-151.
G Clarke, KN Ting, C Wiart and J Fry. High correlation of 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging, ferric reducing activity potential and total phenolics content indicates redundancy in use of all three assays to screen for antioxidant activity of extracts of plants from the Malaysian rainforest. Antioxidants 2013; 2(1), 1-10.
W Wang, Y Gao, W Wang, J Zhang, J Yin, T Le, J Xue, UH Engelhardt and H Jiang. Kojic acid showed consistent inhibitory activity on tyrosinase from mushroom and in cultured B16F10 cells compared with Arbutins. Antioxidants 2022; 11(3), 502.
E Neeley, G Fritch, A Fuller, J Wolfe, J Wright and W Flurkey. Variations in IC50 values with purity of mushroom tyrosinase. International Journal of Molecular Sciences 2008; 10(9), 3811-3823.
B Deri, M Kanteev, M Goldfeder, D Lecina, V Guallar, N Adir and A Fishman. The unravelling of the complex pattern of tyrosinase inhibition. Scientific Reports 2016; 6, 34993.
AD Petrillo, AM González-Paramás, B Era, R Medda, F Pintus, C Santos-Buelga and A Fais. Tyrosinase inhibition and antioxidant properties of Asphodelus microcarpus extracts. BMC Complementary Medicine and Therapies 2016; 16(1), 472.
Y Wang, G Zhang, J Yan and D Gong. Inhibitory effect of morin on tyrosinase: Insights from spectroscopic and molecular docking studies. Food Chemistry 2014; 163, 226-233.
NAN Gowda, C Gurikar, MB Anusha and S Gupta. Ultrasound-assisted and microwave-assisted extraction, GC-MS characterization and antimicrobial potential of freeze-dried L. camara flower. Journal of Pure and Applied Microbiology 2022; 16(1), 678-689.
A Nisca, R Ștefănescu, C Moldovan, A Mocan, AD Mare, CN Ciurea, A Man, DL Muntean and C Tanase. Optimization of microwave assisted extraction conditions to improve phenolic content and in vitro antioxidant and anti-microbial activity in Quercus cerris bark extracts. Plants 2022; 11(3), 240.
N Ibrahim and A Kebede. In vitro antibacterial activities of methanol and aqueous leave extracts of selected medicinal plants against human pathogenic bacteria. Saudi Journal of Biological Sciences 2016; 16(2), 629-639.
W Chaisawangwong, T Juckmeta and D Juckmeta. Antibacterial activity of Vernonia cinerea (L.) less. leaf and flower crude extracts against clinical isolates. Thai Journal of Science and Technology 2021; 10(2), 150-159.
FD Gonelimali, J Lin, W Miao, J Xuan, F Charles, M Chen and SR Hatab. Antimicrobial properties and mechanism of action of some plant extracts against food pathogens and spoilage microorganisms. Frontiers in Microbiology 2018; 9, 1639.
R Mogana, A Adhikari, MN Tzar, R Ramliza and C Wiart. Antibacterial activities of the extracts, fractions and isolated compounds from Canarium patentinervium Miq. against bacterial clinical isolates. BMC Complementary Medicine and Therapies 2020; 20(1), 55.
A Al-Mariri and M Safi. In vitro antibacterial activity of several plant extracts and oils against some gram-negative bacteria. Iranian Journal of Medical Sciences 2014; 39(1), 36-43.
HA Hemeg, IM Moussa, S Ibrahim, TM Dawoud, JH Alhaji, AS Mubarak, SA Kabli, RA Alsubki, AM Tawfik and SA Marouf. Antimicrobial effect of different herbal plant extracts against different microbial population. Saudi Journal of Biological Sciences 2020; 27(12), 3221-3227.
AA Mostafa, AA Al-Askar, KS Almaary, TM Dawoud, EN Sholkamy and MM Bakri. Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi Journal of Biological Sciences 2017; 25(2), 361-366.
A Klancnik, S Piskernik, B Jersek and SS Mozina. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. Journal of Microbiological Methods 2010; 81(2), 121-126.
J Oracz, S Kowalski, D Żyżelewicz, G Kowalska, D Gumul, K Kulbat-Warycha, J Rosicka-Kaczmarek, A Brzozowska, A Grzegorczyk and A Areczuk. The influence of microwave-assisted extraction on the phenolic compound profile and biological activities of extracts from selected Scutellaria species. Molecules 2023; 28(9), 3877.
Y Yuan, J Zhang, J Fan, J Clark, P Shen, Y Li and C Zhang. Microwave assisted extraction of phenolic compounds from four economic brown macroalgae species and evaluation of their antioxidant activities and inhibitory effects on α-amylase, α-glucosidase, pancreatic lipase and tyrosinase. Food Research International 2018; 113, 288-297.
EE Maaiden, S Bouzroud, B Nasser, K Moustaid, K Moustaid, AE Mouttaqi, M Ibourki, H Boukcim and A Hirich. A Comparative study between conventional and advanced extraction techniques: Pharmaceutical and cosmetic properties of plant extracts. Molecules 2022; 27(7), 2074.
L Mousavi, RM Salleh and V Murugaiyah. Antidiabetic and in vitro enzyme inhibition studies of methanol extract of Ocimum tenuiflorum Linn leaves and its fractions. Tropical Life Sciences Research 2020; 31(1), 141-158.
A Bhatia, B Singh, R Arora and S Arora. In vitro evaluation of the α-glucosidase inhibitory potential of methanolic extracts of traditionally used antidiabetic plants. BMC Complementary and Alternative Medicine 2019; 19, 74.
S Poovitha and M Parani. In vitro and in vivo α-amylase and α-glucosidase inhibiting activities of the protein extracts from two varieties of bitter gourd (Momordica charantia L.). BMC Complementary and Alternative Medicine 2016; 16, 185.
TB Nguelefack, CK Fofie, EP Nguelefack-Mbuyo and AK Wuyt. Multimodal α-glucosidase and α-amylase inhibition and antioxidant effect of the aqueous and methanol extracts from the trunk bark of Ceiba pentandra. BioMed Research International 2020; 2020, 3063674.
UF Magaji, O Sacan and R Yanardag. Alpha amylase, alpha glucosidase and glycation inhibitory activity of Moringa oleifera extracts. South African Journal of Botany 2020; 128, 225-230.
MS Shah, MSH Talukder, AMK Uddin, MN Hasan, SAJ Sayem, G Mostafa-Hedeab, MM Rahman, R Sharma, AA Swelum, AAR Mohamed and TB Emran. Comparative assessment of three medicinal plants against diabetes and oxidative stress using experimental and computational approaches. Evidence-Based Complementary and Alternative Medicine 2023; 2023, 6022212.
S Thengyai, P Thiantongin, C Sontimuang, C Ovatlarnporn and P Puttarak. α-Glucosidase and α-amylase inhibitory activities of medicinal plants in Thai antidiabetic recipes and bioactive compounds from Vitex glabrata R. Br. stem bark. Journal of Herbal Medicine 2020; 19(7), 100302.
CH Jhong, J Riyaphan, SH Lin, YC Chia and CF Weng. Screening alpha-glucosidase and alpha-amylase inhibitors from natural compounds by molecular docking in silico. BioFactors 2015; 41(4), 242-251.
R Attaallah, D Elfadil and A Amine. Screening study of enzymatic inhibition of medicinal plants for the treatment of diabetes using a glucometer biosensor approach and optical method. Journal of Herbal Medicine 2021; 28, 100441.
S Kumar, S Narwal, V Kumar and O Prakash. α-glucosidase inhibitors from plants: A natural approach to treat diabetes. Pharmacognosy Reviews 2011; 5(9), 19-29.
K Khadayat, BP Marasini, H Gautam, S Ghaju and N Parajuli. Evaluation of the alpha-amylase inhibitory activity of Nepalese medicinal plants used in the treatment of diabetes mellitus. Clinical Phytoscience 2020; 6, 34.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






