Immunomodulatory and Histoprotective Effects of Luteolin and Related Flavonoids in Autoimmune Thyroiditis: Evidence from A Rat Model
DOI:
https://doi.org/10.48048/tis.2026.10981Keywords:
Autoimmune thyroiditis, Hashimoto’s thyroiditis, Luteolin, Quercetin, Dihydroquercetin, Anti-TPO, Thyroid hormones, Immunomodulation, Oxidative stress, Thyroid histologyAbstract
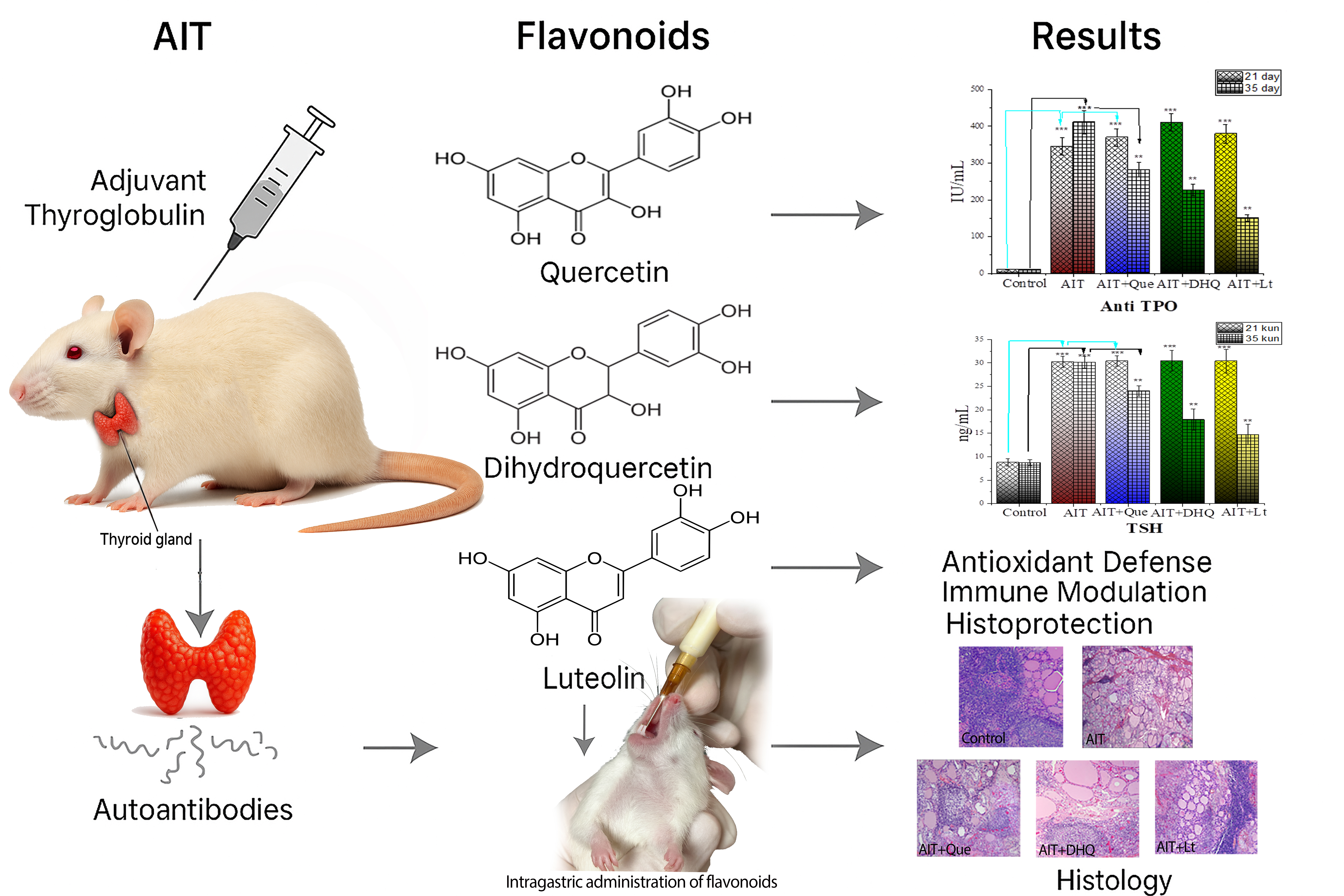
Autoimmune thyroiditis (AIT), particularly Hashimoto’s thyroiditis, is a common endocrine disorder characterized by immune-mediated follicular destruction, lymphocytic infiltration, and hormonal imbalance. Current management relies mainly on hormone replacement therapy, which does not address underlying immune dysregulation. This study evaluated the immunomodulatory, antioxidant, and histoprotective effects of 3 plant-derived flavonoids - quercetin, dihydroquercetin, and luteolin - in an experimental rat model of AIT. Thirty male Wistar rats were divided into 5 groups: Healthy control, untreated AIT, and AIT treated with quercetin (25 mg/kg/day), dihydroquercetin (20 mg/kg/day), or luteolin (10 mg/kg/day) for 14 days. AIT was induced by bovine thyroglobulin with Freund’s adjuvant. Serum anti-thyroid peroxidase (anti-TPO) antibodies, thyroid-stimulating hormone (TSH), metabolic parameters, and liver/pancreatic enzyme activity were measured. Histological analysis assessed thyroid architecture and lymphocytic infiltration. All flavonoids significantly reduced anti-TPO titers and normalized TSH, glucose, lipid, and uric acid levels. Luteolin showed the strongest effect, decreasing anti-TPO by 63%, restoring liver enzyme activity, improving lipid/glucose metabolism, and reducing lymphocytic infiltration. Histologically, luteolin preserved thyroid follicular integrity and prevented tertiary lymphoid structure formation. Luteolin demonstrated superior immunomodulatory and metabolic benefits compared to quercetin and dihydroquercetin, highlighting its translational potential as an adjunctive therapy for autoimmune thyroiditis.
HIGHLIGHTS
- Luteolin, quercetin, and dihydroquercetin exerted strong immunomodulatory, antioxidant, and histoprotective effects in an experimental autoimmune thyroiditis (AIT) rat model.
- Luteolin showed the greatest efficacy, reducing anti-TPO antibodies by 63%, normalizing TSH, and improving metabolic parameters.
- Flavonoid treatment corrected AIT-induced hyperglycemia, dyslipidemia, and hyperuricemia, restoring systemic biochemical balance.
- Histological and biochemical evaluations confirmed luteolin’s ability to preserve thyroid architecture, restore endocrine balance, and prevent tertiary lymphoid structure formation.
GRAPHICAL ABSTRACT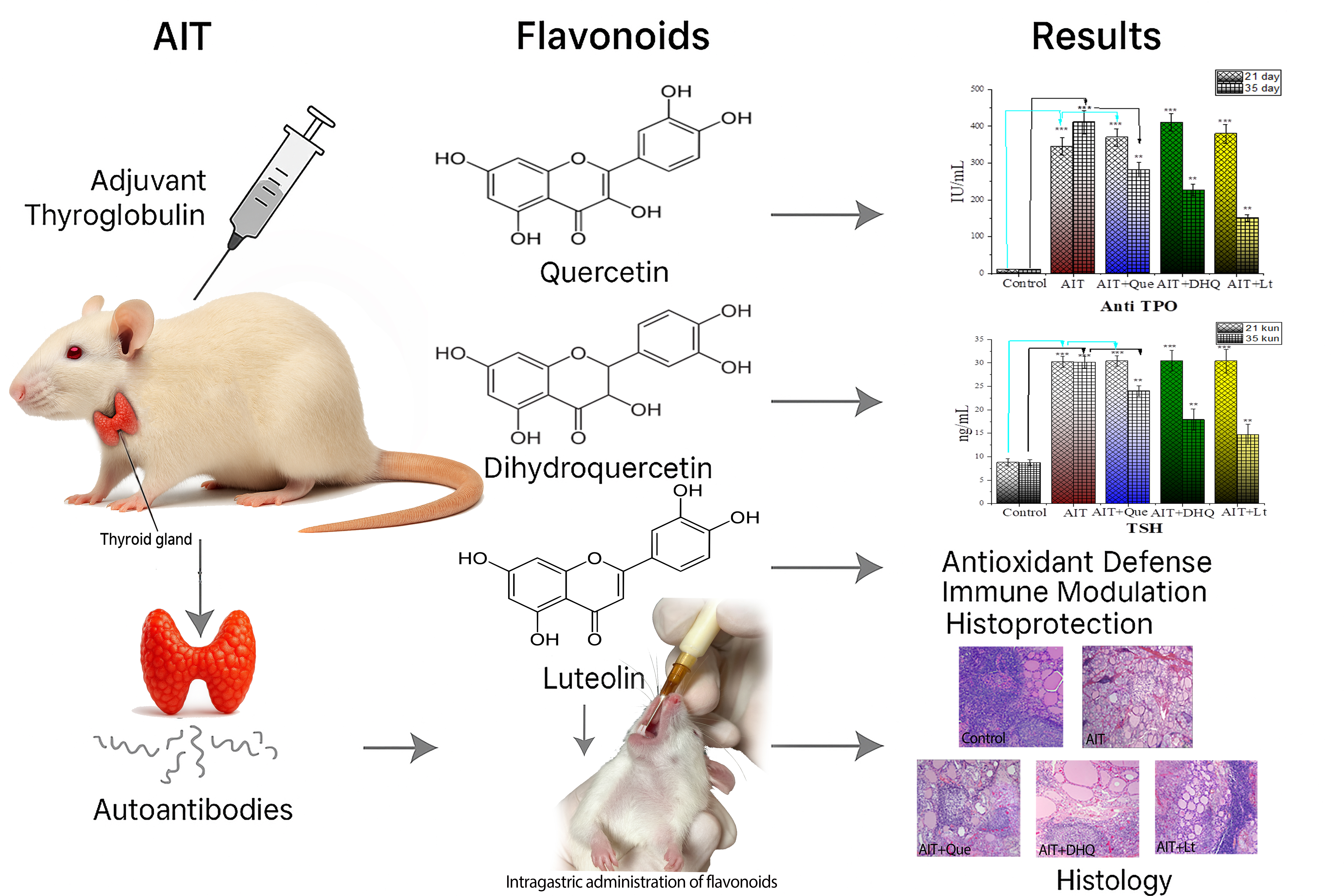
Downloads
References
CM Dayan and GH Daniels. Chronic autoimmune thyroiditis. The New England Journal of Medicine 1996; 335(2), 99-107.
Y Tomer and TF Davies. Searching for the autoimmune thyroid disease susceptibility genes: From gene mapping to gene function. Endocrine Reviews 2003; 24(5), 694-717.
JM Anaya J Castiblanco, A Rojas-Villarraga, R Pineda-Tamayo, RA Levy, J Gómez-Puerta, C Dias, RD Mantilla, JE Gallo, R Cervera, Y Shoenfeld and M Arcos-Burgos. The multiple autoimmune syndromes. A clue for the autoimmune tautology. Clinical Reviews in Allergy & Immunology 2012; 43(3), 256-264.
MT McDermott. Hypothyroidism. Annals of Internal Medicine 2020; 173(1), ITC1-ITC16.
TC Davison and AH Letton. Hashimoto’s disease. The Journal of Clinical Endocrinology & Metabolism 1949; 9(10), 980-6.
P Caturegli, AD Remigis, K Chuang, M Dembele, A Iwama and S Iwama. Hashimoto’s thyroiditis: celebrating the centennial through the lens of the Johns Hopkins hospital surgical pathology records. Thyroid 2013; 23(2), 142-150.
A Antonelli, SM Ferrari, A Corrado, AD Domenicantonio and P Fallahi. Autoimmune thyroid disorders. Autoimmunity Reviews 2015; 14(2), 174-180.
RM Ruggeri, TM Vicchio, M Cristani, R Certo, D Caccamo, A Alibrandi, S Giovinazzo, A Saija, A Campennì and F Trimarchi. Oxidative stress and advanced glycation end products in Hashimoto’s thyroiditis. Thyroid 2016; 26(4), 504-511.
I Ates, FM Yilmaz, M Altay, N Yilmaz, D Berker and S Güler. The relationship between oxidative stress and autoimmunity in Hashimoto’s thyroiditis, United Kingdom. European Journal of Endocrinology 2015; 173(6), 791-799.
G Batóg, A Dołoto, E Bąk, I Piątkowska-Chmiel, P Krawiec, E Pac-Kożuchowska, M Herbet. The interplay of oxidative stress and immune dysfunction in Hashimoto's thyroiditis and polycystic ovary syndrome: a comprehensive review. Front Immunol. 2023 14 1211231.
VP Reddy. Oxidative Stress in health and disease. Biomedicines 2023; 11(11), 2925.
J Kochman, K Jakubczyk, P Bargiel and K Janda-Milczarek. The influence of oxidative stress on thyroid diseases. Antioxidants 2021; 10(9), 1442.
C Thanas, PG Ziros, DV Chartoumpekis, CO Renaud and GP Sykiotis. The keap1/Nrf2 signaling pathway in the thyroid-2020 update. Antioxidants 2020; 9(11), 1082.
N Suganthy, KP Devi, SF Nabavi, N Braidy and SM Nabavi. Bioactive effects of quercetin in the central nervous system: Focusing on the mechanisms of actions. Biomedicine & Pharmacotherapy 2016; 84, 892-908.
Deepika and PK Maurya. Health benefits of quercetin in age-related diseases. Molecules 2022; 27(8), 2498.
F Babaei, M Mirzababaei and M Nassiri-Asl. Quercetin in food: Possible mechanisms of its effect on memory. Journal of Food Science 2018; 83(9), 2280-2287.
G El-Saber Batiha, AM Beshbishy, M Ikram, ZS Mulla, ME Abd El-Hack, AE Taha, AM Algammal and YHA Elewa. The pharmacological activity, biochemical properties, and pharmacokinetics of the major natural polyphenolic flavonoid: Quercetin. Foods 2020; 9(3), 374.
S Ozgen, OK Kilinc and Z Selamoğlu. Antioxidant activity of quercetin: A mechanistic review. Turkish Journal of Agriculture - Food Science and Technology 2016; 4(12), 1134-1138.
JI Dower, JM Geleijnse, L Gijsbers, C Schalkwijk, D Kromhout and PC Hollman. Supplementation of the pure flavonoids epicatechin and quercetin affects some biomarkers of endothelial dysfunction and inflammation in (Pre)hypertensive adults: A randomized double-blind, placebo-controlled, crossover trial. The Journal of Nutrition 2015; 145(7), 1459-1463.
G Li, X Shen, Y Wei, X Si, X Deng and J Wang. Quercetin reduces streptococcus suis virulence by inhibiting suilysin activity and inflammation. International Immunopharmacology 2019; 69, 71-78.
D Xu, MJ Hu, YQ Wang and YL Cui. Antioxidant activities of quercetin and its complexes for medicinal application. Molecules 2019; 24(6), 1123.
L Xiao, G Luo, Y Tang and P Yao. Quercetin and iron metabolism: What we know and what we need to know. Food and Chemical Toxicology 2018; 114, 190-203.
A Rauf, M Imron, IA Khan, M Ur-Rehman, SA Gilani and Z Mehmood. The anticancer potential of quercetin: A comprehensive review. Phytotherapy Research 2018; 32(11), 2109-2130.
SV Orlova and VV Tatarinov. Bioavailability and safety of dihydroquercetin. Pharmaceutical Chemistry Journal 2022; 55(11), 1133-1137.
Z Liu, D Qiu, T Yang, J Su, C Liu, X Su, A Li, P Sun, J Li, L Yan, C Ding and S Zhang. Research progress of dihydroquercetin in the treatment of skin diseases. Molecules 2023; 28(19), 6989.
L Lei, Y Chai, H Lin, C Chen, M Zhao, W Xiong, J Zhuang and X Fan. Dihydroquercetin activates AMPK/Nrf2/HO-1 signaling in macrophages and attenuates inflammation in LPS-induced endotoxemic mice. Frontiers in Pharmacology 2020; 11, 662.
EM Naoual, AE Allam, S Aboulaghras, I Jaouadi, S Bakrim, NE Omari, MA Shariati, A Miftakhutdinov, P Wilairatana, MS Mubarak and A Bouyahya. Inflammatory auto-immune diseases of the intestine and their management by natural bioactive compounds. Biomedicine & Pharmacotherapy 2022; 151, 113158.
MT Macvanin, Z Gluvic, S Zafirovic, X Gao, M Essack and ER Isenovic. The protective role of nutritional antioxidants against oxidative stress in thyroid disorders. Frontiers in Endocrinology 2023; 13, 1092837.
Y Chen and P Deuster. Comparison of quercetin and dihydroquercetin: Antioxidant-independent actions on erythrocyte and platelet membrane. Chemico-Biological Interactions 2009; 182(1), 7-12.
JE Kim, SS Kim, CG Hyun and NH Lee. Antioxidative chemical constituents from the stems of Cleyera japonica Thunberg. International Journal of Pharmacology 2012; 8(5), 410-415.
UG Gayibov, SN Gayibova, MK Pozilov, FSH Tuxtaeva, UR Yusupova, GMK Djabbarova, ZA Mamatova, NA Ergashev and TF Aripov. Influence of quercetin and dihydroquercetin on some functional parameters of rat liver mitochondria. Journal of Microbiology, Biotechnology and Food Sciences 2021; 11(1), e2924.
Y Xu, Z Li, Y Wang, C Li, M Zhang, H Chen, W Chen, Q Zhong, J Pei, W Chen, GRMM Haenen and M Moalin. Unraveling the antioxidant activity of 2R,3R-dihydroquercetin. International Journal of Molecular Sciences 2023, 24(18), 14220.
AJ Day, MS DuPont, S Ridley, M Rhodes, MJ Rhodes, MR Morgan and G Williamson. Deglycosylation of flavonoid and isoflavonoid glycosides by human small intestine and liver β-glucosidase activity. FEBS Letters 1998; 436(1), 71-75.
K Ioku, Y Pongpiriyadacha, Y Konishi, Y Takei, N Nakatani and J Terao. β-Glucosidase activity in the rat small intestine toward quercetin monoglucosides. Bioscience, Biotechnology, and Biochemistry 1998; 62(7), 1428-1431.
C Vanessa, M Christine, B Catherine, M Claudine, D Christian and R Christian. Comparison of the intestinal absorption of quercetin, phloretin and their glucosides in rats. The Journal of Nutrition 2001; 131(8), 2109-2114.
H Wei, T Zhao, X Liu, Q Ding, J Yang, X Bi, Z Cheng, C Ding and W Liu. Mechanism of action of dihydroquercetin in the prevention and therapy of experimental liver injury. Molecules 2024; 29(15), 3537.
W Qi, W Qi, D Xiong and M Long. Quercetin: Its antioxidant mechanism, antibacterial properties and potential application in prevention and control of toxipathy. Molecules 2022; 27(19), 6545.
R Menghini, U Campia, M Tesauro, A Marino, V Rovella, G Rodia, F Schinzari, B Tolusso, N di Daniele, M Federici, A Zoli, G Ferraccioli and C Cardillo. Toll-like receptor 4 mediates endothelial cell activation through NF-κB but is not associated with endothelial dysfunction in patients with rheumatoid arthritis. Plos One 2014; 9(6), e99053.
M López-Lázaro. Distribution and biological activities of the flavonoid luteolin. Mini-Reviews in Medicinal Chemistry 2009; 9(1), 31-59.
N Aziz, MY Kim and JY Cho. Anti-inflammatory effects of luteolin: A review of in vitro, in vivo, and in silico studies. Journal of Ethnopharmacology 2018; 225, 342-358.
ME Ludgate, G Masetti and P Soares. The relationship between the gut microbiota and thyroid disorders. Nature Reviews Endocrinology 2024; 20(9), 511-525.
B Gong, C Wang, F Meng, H Wang, B Song, Y Yang and Z Shan. Association between gut microbiota and autoimmune thyroid disease: A systematic review and meta-analysis. Frontiers in Endocrinology 2021; 12, 774362.
WL Sun, JW Yang, HY Dou, GQ Li, XY Li, L Shen and HF Ji. Anti-inflammatory effect of luteolin is related to the changes in the gut microbiota and contributes to preventing the progression from simple steatosis to nonalcoholic steatohepatitis. Bioorganic Chemistry 2021; 112, 104966.
SF Nabavi, N Braidy, O Gortzi, E Sobarzo-Sanchez, M Daglia, K Skalicka-Woźniak and SM Nabavi. Luteolin as an anti-inflammatory and neuroprotective agent: A brief review. Brain Research Bulletin 2015; 119, 1-11.
R Wang, X Li, Y Xu, Y Li, W Zhang, R Guo and J Song. Progress, pharmacokinetics and future perspectives of luteolin modulating signaling pathways to exert anticancer effects: A review. Medicine 2024; 103(34), e39398.
Z Jia, P Nallasamy, D Liu, H Shah, JZ Li, R Chitrakar, H Si, J McCormick, H Zhu, W Zhen and Y Li. Luteolin protects against vascular inflammation in mice and TNF-alpha-induced monocyte adhesion to endothelial cells via suppressing IΚBα/NF-κB signaling pathway. Journal of Nutritional Biochemistry 2015; 26(3), 293-302.
JF Zambrano-Zaragoza, EJ Romo-Martínez, JM Durán-Avelar, N García-Magallanes and N Vibanco-Pérez. Th17 cells in autoimmune and infectious diseases. International Journal of Inflammation 2014; 2014, 651503.
J Jingwen, Z Fengting, Z Hui, S Tao, F Fangfang, Ch Xuewu and Z Yanhong. Luteolin suppresses the growth of colon cancer cells by inhibiting the IL-6/STAT3 signaling pathway. Journal of Gastrointestinal Oncology 2022; 13(4) 1722-1732.
S Song, Z Su, H Xu, M Niu, X Chen, H Min, B Zhang, G Sun, S Xie, H Wang and Q Gao. Luteolin selectively kills STAT3 highly activated gastric cancer cells through enhancing the binding of STAT3 to SHP-1. Cell Death & Disease 2017; 8(2), e2612.
Z Zhou, L Zhang, Y Liu, C Huang, W Xia, H Zhou, Z Zhou and X Zhou. Luteolin protects chondrocytes from H2O2-induced oxidative injury and attenuates osteoarthritis progression by activating AMPK-Nrf2 signaling. Oxidative Medicine and Cellular Longevity 2022; 2022, 5635797.
PDSM Fernando, DO Ko, MJ Piao, KA Kang, HMUL Herath and JW Hyun. Protective effect of luteolin against oxidative stress‑mediated cell injury via enhancing antioxidant systems. Molecular Medicine Reports 2024; 30(1), 121.
A Rauf, P Wilairatana, PB Joshi, Z Ahmad, A Olatunde, N Hafeez, HA Hemeg and MS Mubarak. Revisiting luteolin: An updated review on its anticancer potential. Heliyon 2024; 10(5), e26701.
Z Pan, T Zhu, Y Liu and N Zhang. Role of the CXCL13/CXCR5 Axis in autoimmune diseases. Frontiers in Immunology 2022; 13, 850998.
P Taweesap, P Potue, J Khamseekaew, M Iampanichakul, B Jan-O, P Pakdeechote and P Maneesai. Luteolin relieves metabolic dysfunction-associated fatty liver disease caused by a high-fat diet in rats through modulating the AdipoR1/AMPK/PPARγ signaling pathway. International Journal of Molecular Sciences 2025; 26(8), 3804.
N Xia, G Chen, M Liu, X Ye, Y Pan, J Ge, Y Mao, H Wang, J Wang and S Xie. Anti-inflammatory effects of luteolin on experimental autoimmune thyroiditis in mice. Experimental and Therapeutic Medicine 2016; 12(6), 4049-4054.
S Jang, KW Kelley and RW Johnson. Luteolin reduces IL-6 production in microglia by inhibiting JNK phosphorylation and activation of AP-1. Proceedings of the National Academy of Sciences of the United States of America 2008; 105(21), 7534-7539.
LS Kuchkarova, KY Kayumov, NA Ergashev and GT Kudeshovа. Effect of quercetin on the intestinal carbohydrases activity in the offspring of the lead intoxicated mother. Journal of Natural Remedies 2024; 24(2), 391-396.
YM Kong. Experimental autoimmune thyroiditis in the mouse. Current Protocols in Immunology 2007; 15, 15.7.1-15.7.21.
H Li, F Qi, D Li, Y Fan, J Liu, C Li, J Chen, X Wu, X Zhang and F Xu. Acupuncture attenuates experimental autoimmune thyroiditis by modulating intestinal microbiota and palmitic acid metabolism. Frontiers in Immunology 2025; 16, 1541728.
CA Spencer. Assay of thyroid hormone and related substances. In: KR Feingold, SF Ahmed, B Anawalt, MR Blackman, A Boyce, G Chrousos, E Corpas, WWD Herder, K Dhatariya, K Dungan, J Hofland, S Kalra, G Kaltsas, N Kapoor, C Koch, P Kopp, M Korbonits, CS Kovacs, W Kuohung, B Laferrère, …, DP Wilson (Eds.). Endotext. MDText, Massachusetts, 2000.
G Shaker and M Zubair. Peroxidase-coupled glucose method. StatPearls. Treasure Island, Florida, 2025.
CE Shideler, KK Stewart, J Crump, MR Wills, J Savory and BW Renoe. Automated multiple flow-injection analysis in clinical chemistry: determination of total protein with Biuret reagent. Clinical Chemistry 1980; 26(10), 1454-1458.
PA Christensen. Stability of plasma albumin depends on measurement method. Clinical Chemistry and Laboratory Medicine 2016; 54(11), e327-e329.
E Henkel and M Stoltz. A newly drafted colour test for the determination of triglycerides convenient for manual and mechanized analysis (glycerolphosphate-oxidase-PAP method). Journal of Analytical Chemistry 1982; 311(4), 451-452.
SMT Islam, B Osa-Andrews, PM Jones, AR Muthukumar, I Hashim and J Cao. Methods of low-density lipoprotein-cholesterol measurement: Analytical and clinical applications. The Electronic Journal of IFCC 2022; 33(4), 282-294.
WG Barr. Uric acid. In: HK Walker, WD Hall and JW Hurst (Eds.). Clinical methods: The history, physical, and laboratory examinations. Butterworths, Boston, Massachusetts, 1990.
AY Foo and R Bais. Amylase measurement with 2-chloro-4-nitrophenyl maltotrioside as substrate. Clinica Chimica Acta 1998; 272(2),137-147.
DH Vroon and Z Israili. Aminotransferases. In: HK Walker, WD Hall and JW Hurst (Eds.). Clinical methods: The history, physical, and laboratory examinations. Butterworths, Boston, Massachusetts, 1990.
FC Sitzmann and H Orth. N-acetyl-cystein-(NAC)-aktivierte Kreatinkinase (CK) und Isoenzym CK-MB im Serum von Kindern (N-acetyl-cystein-(NAC)-activated creatinkinase (CK) and isoenzyme CK-MB in the serum of children). Monatsschrift Kinderheilkunde 1983; 131(8), 509-512.
WP Venetz, C Mangan and IW Siddiqi. Kinetic determination of alkaline phosphatase activity based on hydrolytic cleavage of the P F bond in monofluorophosphate and fluoride ion-selective electrode. Analytical Biochemistry 1990; 191(1), 127-132.
XX Gan, LK Zhong, F Shen, JH Feng, YY Li, SJ Li, WS Cai and B Xu. Network pharmacology to explore the molecular mechanisms of prunella vulgaris for treating Hashimoto’s thyroiditis. Frontiers in Pharmacology 2021 12, 700896.
Y Chen, B Jiang, C Qu, C Jiang, C Zhang, Y Wang, F Chen, X Sun, L Su and Y Luo. Bioactive components in prunella vulgaris for treating Hashimoto’s disease via regulation of innate immune response in human thyrocytes. Heliyon 2024; 10(16), e36103.
MS Hussain, G Gupta, A Goyal, R Thapa, WH Almalki, I Kazmi, SI Alzarea, S Fuloria, DU Meenakshi, V Jakhmola, M Pandey, SK Singh and K Dua. From nature to therapy: Luteolin’s potential as an immune system modulator in inflammatory disorders. Journal of Biochemical and Molecular Toxicology 2023; 37(11), e23482.
CV Rizos, MS Elisaf and EN Liberopoulos. Effects of thyroid dysfunction on lipid profile. The Open Cardiovascular Medicine Journal 2011; 5, 76-84.
DM Carragher, J Rangel-Moreno and TD Randall. Ectopic lymphoid tissues and local immunity. Seminars in Immunology 2008; 20(1), 26-42.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






