Biflavonoids from Araucaria Genus as Selective PDE4 Inhibitors: Insights from In Silico and In Vitro Studies
DOI:
https://doi.org/10.48048/tis.2026.10899Keywords:
Anti-inflammatory, Biflavonoids, Enzyme inhibition, Molecular docking, Molecular dynamics, PDE4 inhibitors, Anti-inflammatory, Biflavonoids, Enzyme inhibition, Molecular docking, Molecular dynamics, PDE4 inhibitorsAbstract
Chronic inflammation is a major contributor to autoimmune diseases, necessitating the discovery of selective phosphodiesterase-4 (PDE4) inhibitors. Biflavonoids, with diverse biological activities, exhibit anti-inflammatory potential. This study employed molecular docking and molecular dynamics (MD) simulations to evaluate the interaction of 25 biflavonoid compounds with PDE4B and PDE4D. The most promising compound was validated using in vitro enzyme inhibition assays. Molecular docking identified 7,7''-di-O-methylamentoflavone as a potent PDE4B inhibitor with strong binding affinity and favourable MM/GBSA binding energy of –49.56 ± 4.12 kcal/mol, compared to its PDE4D binding energy of –39.77 ± 5.21 kcal/mol. Molecular dynamics simulations confirmed the stability of ligand–protein interactions. In vitro assays of six isolated biflavonoids from Araucaria hunsteinii and Araucaria cunninghamii confirmed that 7,7''-di-O-methylamentoflavone as a selective PDE4B inhibitor, with an IC50 value of 13.9 ± 2.38 μM. This study provides new insights into the potential of biflavonoids as selective PDE4B inhibitors. However, further research is required to validate their therapeutic potential, including in vivo evaluation and broader safety profiling.
HIGHLIGHTS
- Araucaria biflavonoids as anti-inflammatory agents: Investigated 25 biflavonoids from the Araucaria genus using in silico (docking, molecular dynamics) and in vitro (ELISA enzyme inhibition) methods.
- Promising PDE4B inhibitor identified: 7,7′′-di-O-methylamentoflavone showed high stability, strong binding affinity, and selective PDE4B inhibition (IC50 = 13.9 ± 2.38 μM).
- Potential for drug development: Findings support biflavonoids as potential anti- inflammatory drug leads, but further in vivo and pharmacokinetic studies are required.
GRAPHICAL ABSTRACT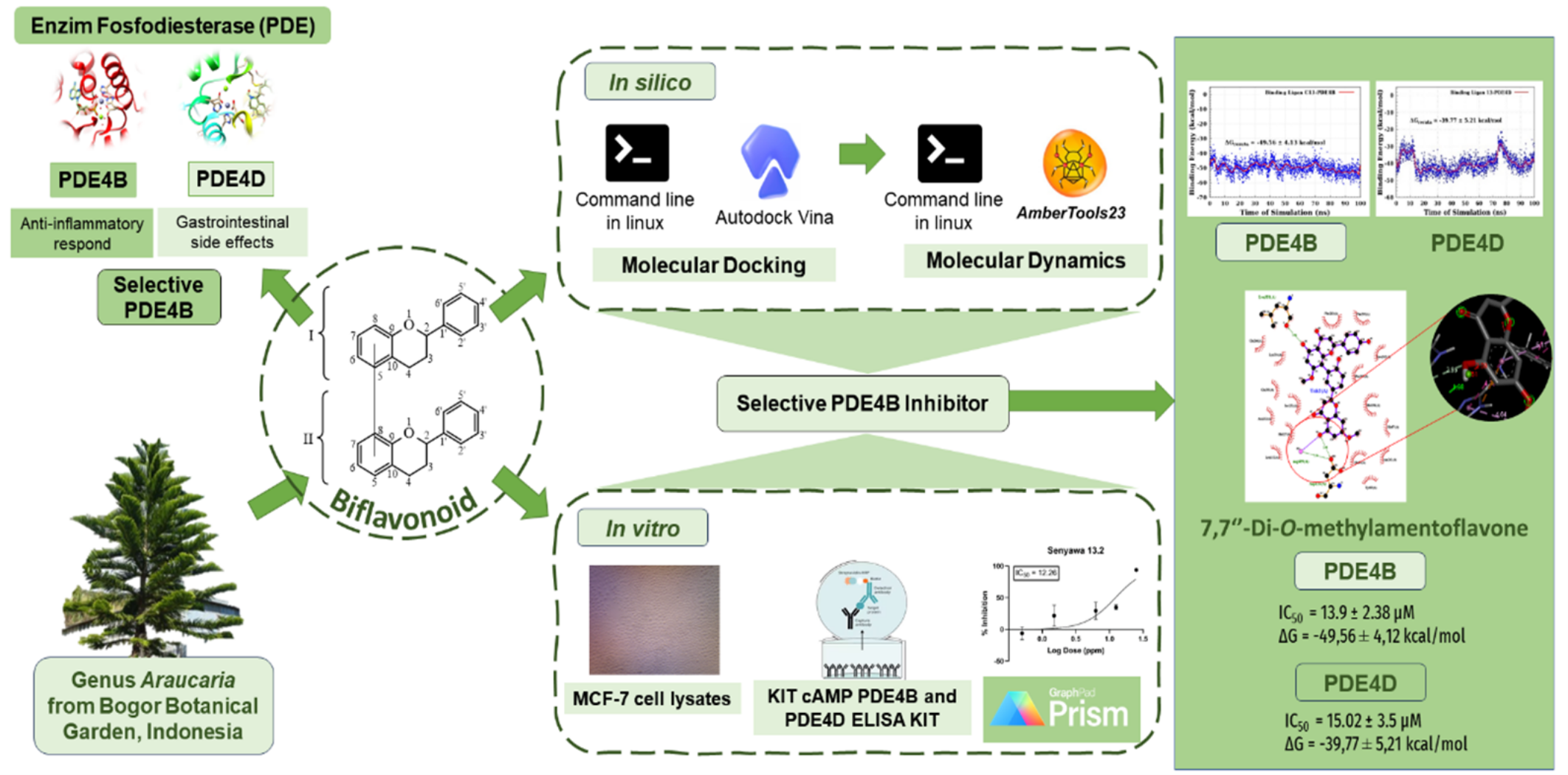
Downloads
References
SI Grivennikov, FR Greten and M Karin. Immunity, Inflammation and Cancer. Cell 2010; 140(6), 883-899.
K Komatsu, JY Lee, M Miyata, JH Lim, H Jono, T Koga, H Xu, C Yan, H Kai and JD Li. Inhibition of PDE4B suppresses inflammation by increasing expression of the deubiquitinase CYLD. Nature Communications 2013; 4, 1684.
T Peng, J Gong, Y Jin, Y Zhou, R Tong, X Wei, L Bai and J Shi. Inhibitors of phosphodiesterase as cancer therapeutics. European Journal of Medicinal Chemistry 2018; 150, 742-756.
L Crocetti, G Floresta, A Cilibrizzi and MP Giovannoni. An overview of PDE4 inhibitors in clinical trials: 2010 to early 2022. Molecules 2022; 27(15), 4964.
M Al-Nema, A Gaurav and VS Lee. Docking based screening and molecular dynamics simulations to identify potential selective PDE4B inhibitor. Heliyon 2020; 6(9), e04856.
J Jin, F Mazzacuva, L Crocetti, MP Giovannoni and A Cilibrizzi. PDE4 inhibitors: Profiling hits through the multitude of structural classes. International Journal of Molecular Sciences; 24(14), 11518.
H Li, J Zuo and W Tang. Phosphodiesterase-4 inhibitors for the treatment of inflammatory diseases. Frontiers in Pharmacology 2018; 9, 1048.
JE Phillips. Inhaled phosphodiesterase 4 (PDE4) inhibitors for inflammatory respiratory diseases. Frontiers in Pharmacology 2020; 11, 259.
M Chaabi, C Antheaume, B Weniger, H Justiniano, C Lugnier and A Lobstein. Biflavones of decussocarpus rospigliosii as phosphodiesterases inhibitors. Planta Medica 2007; 73(12), 1284-1286.
A Gaurav and V Gautam. Pharmacophore based virtual screening approach to identify selective PDE4B inhibitors. Iranian Journal of Pharmaceutical Research 2017; 16(3), 910-923.
RP Gangwal, MV Damre, NR Das, GV Dhoke, A Bhadauriya, RA Varikoti, SS Sharma and AT Sangamwar. Structure based virtual screening to identify selective phosphodiesterase 4B inhibitors. Journal of Molecular Graphics and Modelling 2015; 57, 89-98.
M Rahman, M Riaz and UR Desai. Synthesis of biologically relevant biflavanoids - a review. Chemistry and Biodiversity 2007; 4(11), 2495-2527.
B Gil, MJ Sanz, MC Terencio, R Gunasegaran, M Payá and MJ Alcaraz. Morelloflavone, a novel biflavonoid inhibitor of human secretory phospholipase A2 with anti-inflammatory activity. Biochemical Pharmacology 1997; 53(5), 733-740.
T Banerjee, G Valacchi, VA Ziboh and A Vliet. Inhibition of TNFα-induced cyclooxygenase-2 expression by amentoflavone through suppression of NF-κB activation in A549 cells. Molecular and Cellular Biochemistry 2002; 238(1-2), 105-110.
JK Son, MJ Son, E Lee, TC Moon, KH Son, CH Kim, HP Kim, SS Kang and HW Chang. Ginkgetin, a biflavone from Ginko biloba leaves, inhibits cyclooxygenases-2 and 5-lipoxygenase in mouse bone marrow-derived mast cells. Biological and Pharmaceutical Bulletin 2005; 28(12), 2181-2184.
X Su, ZH Zhu, L Zhang, Q Wang, MM Xu, C Lu, Y Zhu, J Zeng, JA Duan and M Zhao. Anti-inflammatory property and functional substances of Lonicerae japonicae Caulis. Journal of Ethnopharmacology 2021; 267, 113502.
HP Kim, H Park, KH Son, HW Chang and SS Kang. Biochemical pharmacology of biflavonoids: Implications for anti-inflammatory action. Archives of Pharmacal Research 2008; 31(3), 265-273.
YM Lin, MT Flavin, R Schure, FC Chen, R Sidwell, DL Barnard, JH Huffman and ER Kern. Antiviral activities of biflavonoids. Planta Medica 1999; 65(2), 120-125.
DD Agusta, H Dianhar, DUC Rahayu, IH Suparto and P Sugita. Anticancer and antivirus activities of two biflavonoids from Indonesian Araucaria hunsteinii K Schum Leaves. Journal of Human University (Natural Science) 2022; 49(3), 169-177.
MK Lee, SW Lim, H Yang, SH Sung, HS Lee, MJ Park and YC Kim. Osteoblast differentiation stimulating activity of biflavonoids from Cephalotaxus koreana. Bioorganic & Medicinal Chemistry Letters 2006; 16(11), 2850-2854.
O Kunert, RC Swamy, M Kaiser, A Presser, S Buzzi, AVNA Rao and W Schühly. Antiplasmodial and leishmanicidal activity of biflavonoids from Indian Selaginella bryopteris. Phytochemistry Letters 2008; 1(4), 171-174.
T Okoko. In vitro antioxidant and free radical scavenging activities of Garcinia kola seeds. Food and Chemical Toxicology 2009; 47(10), 2620-2623.
JH Hwang, H Choi, ER Woo and DG Lee. Antibacterial effect of amentoflavone and its synergistic effect with antibiotics. Journal of Microbiology and Biotechnology 2013, 23(7), 953-958.
P Sugita, DD Agusta, H Dianhar, IH Suparto, Kurniawanti, DUC Rahayu and L Irfana. The cytotoxicity and SAR analysis of biflavonoids isolated from Araucaria hunsteinii K. Schum. leaves against MCF-7 and HeLa cancer cells. Journal of Applied Pharmaceutical Science 2023; 13(10), 199-209.
CS Estevam, FM Oliveira, LM Conserva, LDFCO Lima, ECP Barros, ACP Barros, EMM Rocha and E Andrade. Constituintes químicos e avaliação preliminar in vivo da atividade antimalárica de Ouratea nitida Aubl (Ochnaceae). Revista Brasileira de Farmacognosia 2005; 15(3), 195-198.
Nafisah, P Sugita, B Arifin and ST Wahyudi. Biflavonoid anti-inflammatory activity of the araucariaceae family—a review. Tropical Journal of Phytochemistry and Pharmaceutical Sciences 2024; 3(9), 411-423.
N Ilyas, M Ilyas, W Rahman, M Okigawa and N Kawano. Biflavones from the leaves of Araucaria excelsa. Phytochemistry 1978; 17(5), 987-990.
N Parveen, HM Taufeeq and NU Khan. Biflavones from the leaves of Araucaria araucana. Journal of Natural Products 1987; 50(2), 332-333.
AM Freitas, MTR Almeida, CR Andrighetti-Fröhner, FTGS Cardozo, CRM Barardi, MR Farias and CMO Simões. Antiviral activity-guided fractionation from Araucaria angustifolia leaves extract. Journal of Ethnopharmacology 2009; 126(3), 512-517.
J Chen, ML Yang, J Zeng and K Gao. Antimicrobial activity of Araucaria cunninghamii sweet and the chemical constituents of its twigs and leaves. Phytochemistry Letters 2013; 6(1), 41-45.
C Frezza, DD Vita, L Fonti, O Giampaoli, CD Bosco, F Sciubba, A Venditti, C Scintu and F Attorre. Secondary metabolites of Araucaria cunninghamii Mudie from central Italy. Plant Biosystems - An International Journal Dealing with All Aspects of Plant Biology 2024; 158(4), 589-594.
AN Talaat, SS Ebada, RM Labib, A Esmat, FS Youssef and ANB Singab. Verification of the anti-inflammatory activity of the polyphenolic-rich fraction of Araucaria bidwillii Hook. using phytohaemagglutinin-stimulated human peripheral blood mononuclear cells and virtual screening. Journal of Ethnopharmacology 2018; 226, 44-47.
C Frezza, A Venditti, DD Vita, C Toniolo, M Franceschin, A Ventrone, L Tomassini , S Foddai, M Guiso, M Nicoletti, A Bianco, M Serafini. Phytochemistry, chemotaxonomy and biological activities of the Araucariaceae family—a review. Plants 2020; 9(7), 888.
Kurniawanti, DD Agusta, P Sugita, IH Suparto, H Dianhar and DUC Rahayu. Bioactive compounds of flavone dimers from Indonesian Araucaria columnaris leaves. Rasayan Journal of Chemistry 2023; 16(3), 1872-1882.
P Sugita, SDP Handayani, DD Agusta, L Ambarsari, H Dianhar and DUC Rahayu. Combined in-silico and in-vitro approaches to evaluate the inhibitory potential of biflavonoids from Araucaria plants against α-glucosidase as target protein. Rasayan Journal of Chemistry 2023; 16(1), 361-375
SS El-Hawary, MA Rabeh, MAE Raey, EMA El-Kadder, M Sobeh, UR Abdelmohsen, A Albohy, AM Andrianov, IP Bosko, MM Al-Sanea and DG El-Kolobby. Metabolomic profiling of three Araucaria species and their possible potential role against COVID-19. Journal of Biomolecular Structure and Dynamics 2021; 40(14), 6426-6438.
L Irfana, DD Agusta, B Arifin, ST Wahyudi, SS Achmadi and P Sugita. Biflavonoid from Indonesian Araucaria cunninghamii Mudie leaves activity against breast cancer and 20s proteasome. Trends in Sciences 2025; 22(3), 9198.
K Ramayanti, H Riza and I Fajriaty. Molecular docking of drymaritin, tiptonine A and triptonine B compounds against HIV enzymes. Jurnal Mahasiswa Farmasi Fakultas Kedokteran UNTAN 2019; 1, 1-6.
VTT Le, HV Hung, NX Ha, CH Le, PTH Minh and DT Lam. Natural phosphodiesterase-4 inhibitors with potential anti-inflammatory activities from Millettia dielsiana. Molecules 2023; 28(21), 7253.
F Awaluddin, I Batubara and ST Wahyudi. Molecular dynamics simulation of bioactive compounds against six protein targets of SARS-CoV-2 as COVID-19 antivirus candidates. Jurnal Kimia Valensi 2021; 7(2), 178-187.
T Hou, J Wang, Y Li and W Wang. Assessing the performance of the MM/PBSA and MM/GBSA methods: The accuracy of binding free energy calculations based on molecular dynamics simulations. Journal of Chemical Information and Modeling 2011; 51(1), 69-82.
M Sasikala, R Sundaraganapathy and S Mohan. MTT assay on anticancer properties of phytoconstituents from Ipomoea aquatica Forssk. using MCF–7 cell lines for breast cancer in women. Research Journal of Pharmacy and Technology 2020; 13(3), 1356-1360.
J Cheng, Y Li and J Kong. Ginkgetin inhibits proliferation of HeLa cells via activation of p38/NF-κB pathway. Cellular and Molecular Biology 2019; 65(4), 79-82.
M Luthfia, A Eryandini, D Geraldi, C Narita, CM Jannah and L Ambarsari. Potency of bioactive compounds in Indramayu mango peel waste to inhibit ACE2. Current Biochemistry 2021; 8(2), 51-62.
MZS Al-Khayyat and AGA Al-Dabbagh. In silico prediction and docking of tertiary structure of LuxI, an inducer synthase of vibrio fischeri. Reports of Biochemistry and Molecular Biology 2016; 4(2), 66-75.
K Anwar, E Suhartono and N Komari. Three dimension structure modeling of the superoxide dismutase (SOD) of rice (Oryza sativa) using fold recognition method using Phyre2 web server. Jurnal Ilmiah Berkala Sains dan Terapan Kimia 2022; 16(2), 86-97.
Nafisah, Sarmila, H Habibah, I Saputri, I Setiawati and N Komari. Effect of Kelakai (Stenochlaena palustris) extract on organophosphate pesticide exposure: Cytotoxic studies in silico and in ovo. Jurnal Ilmiah Berkala Sains dan Terapan Kimia 2023; 17(2), 1-14.
R Vaidyanathan, SM Sreedevi, K Ravichandran, SM Vinod, YH Krishnan, LH Babu, PS Parthiban, L Basker, T Perumal, V Rajaraman, G Arumugam, K Rajendran and V Mahalingam. Molecular docking approach on the binding stability of derivatives of phenolic acids (DPAs) with human serum albumin (HSA): Hydrogen-bonding versus hydrophobic interactions or combined influences?. Journal of Colloid and Interface Science Open 2023; 12, 100096.
S Genheden and U Ryde. The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opinion on Drug Discovery 2015; 10(5), 449-461.
GJ Kim, EJ Yang, YS Kim, J Moon, YK Son, JW Nam, I Choi, H Choi and KS Song. Diterpene and biflavone derivatives from Thuja koraiensis and their cytotoxicities against A549 cells. Phytochemistry 2023; 211, 113711.
LS Sreeshma and BR Nair. A simple protocol for the isolation of amentoflavone from two species of Biophytum DC. (Oxalidaceae) and evaluation of its antiproliferative potential. Industrial Crops and Products 2021; 160, 113099.
CAD Lima, LK Maquedano, LS Jaalouk, DCD Santos and GB Longato. Biflavonoids: Preliminary reports on their role in prostate and breast cancer therapy. Pharmaceuticals 2024; 17(7), 874.
AG Mercader and AB Pomilio. Naturally-occurring dimers of flavonoids as anticarcinogens. Anti-Cancer Agents in Medicinal Chemistry 2013; 13(8),1217-1235.
M Drees, R Zimmermann and G Eisenbrand. 3’,5’-Cyclic nucleotide phosphodiesterase in tumor cells as potential target for tumor growth inhibition. Cancer Research1993; 53(13), 3058-3061.
SJ MacKenzie and MD Houslay. Action of rolipram on specific PDE4 cAMP phosphodiesterase isoforms and on the phosphorylation of cAMP-response-element-binding protein (CREB) and p38 MAP kinase in U937 monocytic cells. Biochemical Journal 2000; 347(2), 571-578.
R Yuniarti, S Nadia, A Alamanda, M Zubir, RA Syahputra and M Nizam. Characterization, phytochemical screenings and antioxidant activity test of Kratom leaf ethanol extract (Mitragyna speciosa Korth) using DPPH method. Journal of Physics: Conference Series 2020; 1462, 012026.
MM Alanazi, E Alaa, NA Alsaif, AJ Obaidullah, HM Alkahtani, AA Al-Mehizia, SM Alsubaie, MS Taghour and IH Eissa. Discovery of new 3-methylquinoxalines as potential anti-cancer agents and apoptosis inducers targeting VEGFR-2: design, synthesis and in silico studies. Journal of Enzyme Inhibition and Medicinal Chemistry 2021; 36(1), 1732-1750.
Y Yu, Y Zhao, Y Wang and X Huang. Design and synthesis of novel PDE4 inhibitors as potential candidates for antidepressant agents. Journal of Chemical Research 2023. https://doi.org/10.1177/17475198231202967
YN Liu, YY Huang, JM Bao, YH Cai, YQ Guo, SN Liu, HB Luo and S Yin. Natural phosphodiesterase-4 (PDE4) inhibitors from Crotalaria ferruginea. Fitoterapia 2014; 94, 177-182.
CY Teo, KM Loh, JF Chai, HX Wang, RP Tan, SW Ho and LP Lim. Discovery of a new class of inhibitors for the protein arginine deiminase type 4 (PAD4) by structure-based virtual screening. BMC Bioinformatics 2012; 13(S17), S4.
X Zhang L Jin, Y Wu, B Huang, K Chen, W Huang and J Li. Anti-inflammatory properties of biflavonoids derived from Selaginella moellendorffii Hieron: Targeting NLRP3 inflammasome-dependent pyroptosis. Journal of Ethnopharmacology 2025; 340, 119172.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






