Synthesis and Characterization of Chitosan-Alginate Hydrogel Adsorbent for Paracetamol Removal from Wastewater
DOI:
https://doi.org/10.48048/tis.2025.10767Keywords:
Alginate, Beads, Adsorption, Paracetamol Waste, Chitosan, Alginate, Hydrogel, Adsorption, Paracetamol wasteAbstract
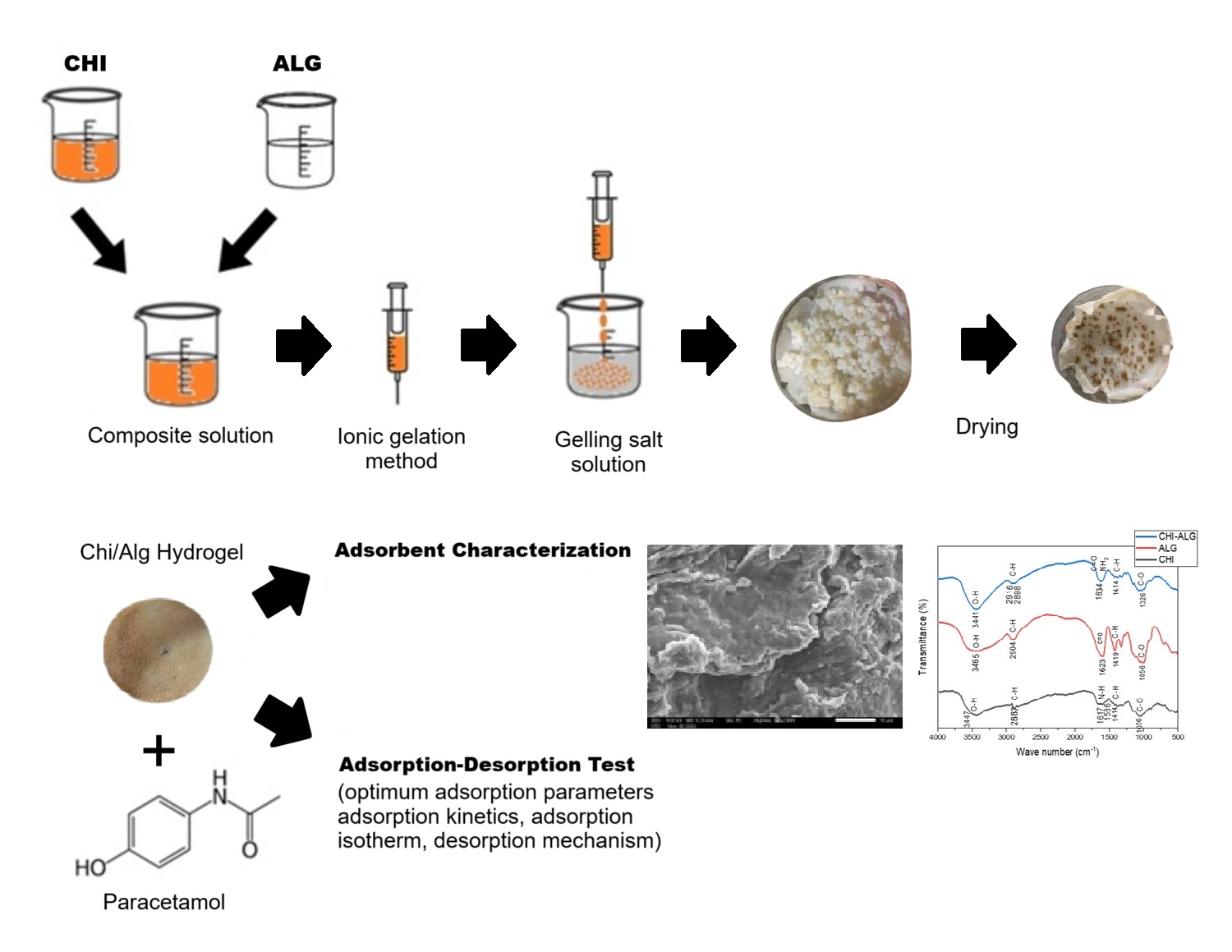
The extensive use of paracetamol has led to the increasing of paracetamol contamination in aquatic ecosystems, posing risks to both human health and the environment. Adsorption is an efficient and cost-effective strategy for addressing paracetamol contamination in the environment. The main target of this study is to develop an efficient and low-cost adsorbent from natural biopolymers derivate for paracetamol removal in wastewater. A Chitosan-Alginate (Chi/Alg) hydrogel was prepared using ionic gelation method by mixing the Alg and Chi solutions in a 4:1 volumetric ratio. The synthesized hydrogel was characterized using Fourier Transform Infrared (FTIR). x-ray diffraction (XRD), Scanning Electron Microscopy (SEM), and Differential Thermal-Thermogravimetric (DTA-TGA) analyzer. Batch adsorption tests were conducted to study the effects of pH, contact time, and adsorbate concentration. UV-Vis spectrophotometer was used to measure the concentration of paracetamol in solution. Adsorption isotherm study was also performed to investigate the kinetic and thermodynamic behavior of the adsorption process. The FTIR data showed the vibration of hydroxyl (O-H), carboxyl (C=O) and amine (N-H2) groups. The XRD data showed that Chi/Alg hydrogel has a semicrystalline structure. Analysis SEM showed that modification of Chi/Alg creates a larger pore size than the initial compound. Finally, DTA-TGA data showed that the beads have higher thermal stability.
The batch test showed that the optimum adsorption conditions were achieved at pH 6, contact time of 45 min, and adsorbate concentration of 80 mg/L. The kinetic studies revealed that the adsorption process followed pseudo-second order reaction kinetics model and the Freundlich isotherm model. It also indicated a robust adsorption of the membrane, with the reaction rate constant of 0.1368 g/mg.min and adsorption capacity of 67.58 mg/g. The adsorption mechanism occurs via both physical and chemical interactions, with physical interactions predominating. This demonstrates the efficacy of Chi/Alg hydrogel as an adsorbent for the removal of paracetamol from water.
HIGHLIGHTS
- Adding alginate to the chitosan can improve the stability of the Chitosan adsorbent
- The manufacture of hydrogel beads Chitosan-Alginate is made by ionic gelation method
- The adsorbent showed different removal behavior depending on the paracetamol concentration and the length of contact time between the adsorbent and paracetamol.
- Adsorbent adsorb paracetamol in waste water through chemical and physical mechanisms.
- The adsorbent maintained over 82.9% efficiency for adsorption of paracetamol in wastewater
GRAPHICAL ABSTRACT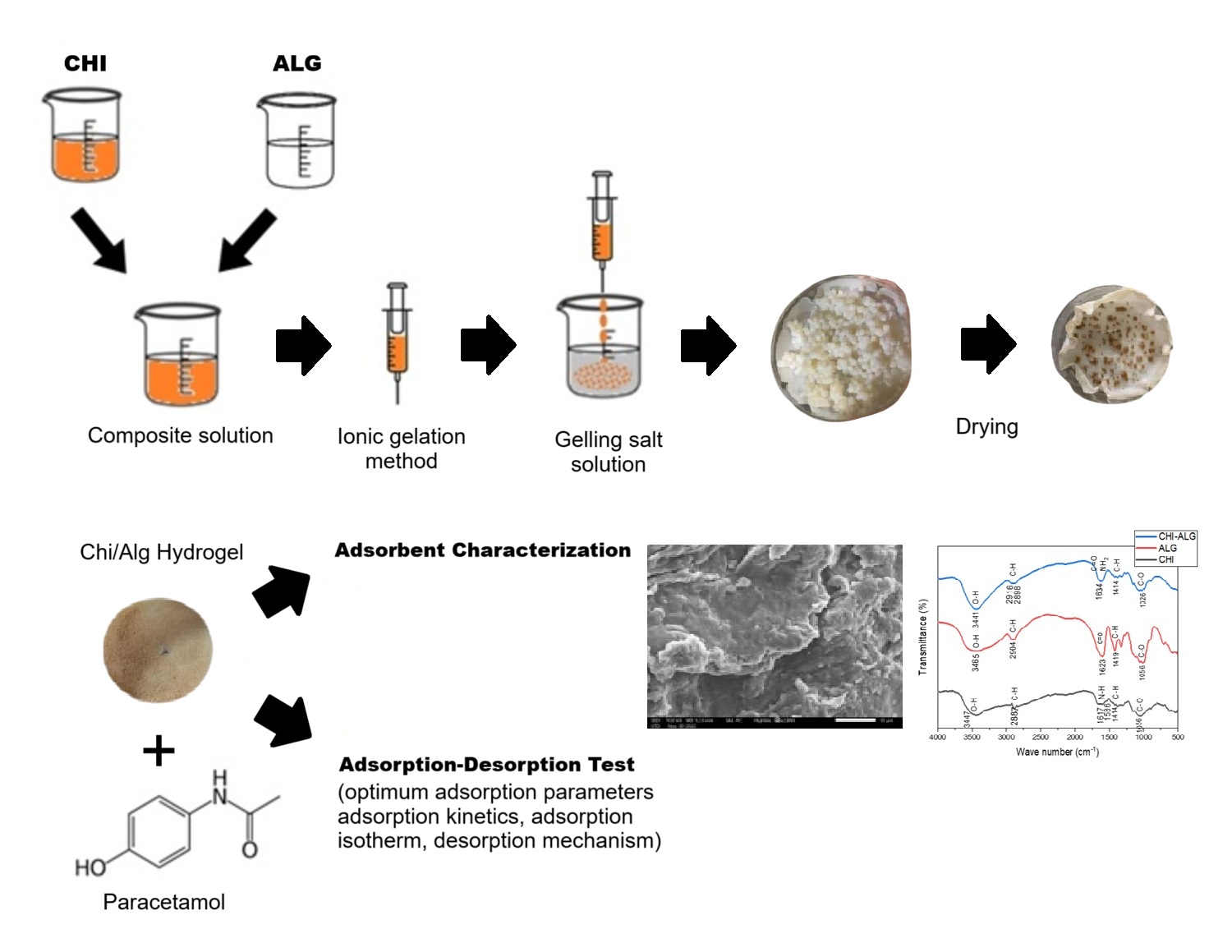
Downloads
References
M Vera, DM Juela, C Cruzat and E Vanegas. Modeling and computational fluid dynamic simulation of acetaminophen adsorption using sugarcane bagasse. Journal of Environmental Chemical Engineering 2021; 9(2), 105056.
M Kumar, S Jaiswal, KK Sodhi, P Shree, DK Singh, PK Agrawal and P Shukla. Antibiotics Bioremediation: Perspectives on its ecotoxicity and resistance. Environment International 2019; 124, 448-461.
M Patel, R Kumar, K Kishor, T Mlsna, CU Pittman, P Jr and D Mohan. Pharmaceuticals of emerging concern in aquatic systems: Chemistry, occurrence, effects, and removal methods. Chemical Reviews 2019; 119, 3453-4470.
EZ MD, D Calamari, MN MSC and R Fanelli. Presence of therapeutic drugs in the environment. The Lancet 2000; 355(9217), 1789-1790.
WC Li. Occurrence, sources, and fate of pharmaceuticals in aquatic environment and soil. Environmental Pollution 2014; 187, 193-201.
HNP Vo, GK Le, TMH Nguyen, XT Bui, KH Nguyen, ER Rene, TDH Vo, NDT Cao and R Mohan. Acetaminophen micropollutant: Historical and current occurrences, toxicity, removal strategies and transformation pathways in different environments. Chemosphere 2019; 236, 124391.
A Al-Kaf, KM Naji, QM Abdullah and WH Edrees. Occurrence of paracetamol in aquatic environments and transformation by microorganisms: A review. Chronicles of Pharmaceutical Science 2017; 1(6), 341-355.
W Koagouw, NA Stewart and C Ciocan. Long-Term exposure of marine mussels to paracetamol: Is time a healer or A Killer? Environmental Science and Pollution Research 2021; 28(35), 48823-48836.
W Koagouw, Z Arifin, GWJ Olivier and C Ciocan. High concentrations of paracetamol in effluent dominated waters of Jakarta Bay, Indonesia. Marine Pollution Bulletin 2021; 169, 112558.
KG Akpomie and J Conradie. Efficient adsorptive removal of paracetamol and thiazolyl blue from polluted water onto biosynthesized copper oxide nanoparticles. Scientific Reports 2023; 13, 859.
S Ahmadzadeh and M Dolatabadi. Removal of acetaminophen from hospital wastewater using electro-fenton process. Environmental Earth Sciences 2018; 77(2), 53.
H Zeng, F Wang, K Xu, J Zhang and D Li. Optimization and regeneration of Chi/Alg hybrid adsorbent embedding iron-manganese sludge for arsenic removal. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2020; 607, 125500.
M Sonmez, A Ficai, D Ficai, R Trusca and E Andronescu. Alg/Cellulose composite beads for environmental applications. Materiale Plastice 2016; 78(2), 165-176.
S Gokila, T Gomathi, PN Sudha and S Anil. Removal of the heavy metal ion chromium (VI) using chi and alg nanocomposites. International Journal of Biological Macromolecules 2017; 104, 1459-1468.
HB Quesada, TPD Araujo, DT Vareschini, MASDD Barros, RG Gomes and R Bergamasco. Chitosan, alginate and other macromolecules as activated carbon immobilizing agents: A review on composite adsorbents for the removal of water contaminants. International Journal of Biological Macromolecules 2020; 164, 2535-2549.
TN Batugedara and CS Rajapakse. Chitosan beads as a natural adsorbent for the removal of Cd(II) from aqueous solutions. International Journal of Science, Environment 2017; 6(1), 606-619.
BA Riyandari, S Suherman and D Siswanta. The physico-mechanical properties and release kinetics of eugenol in Chi/Alg polyelectrolyte complex films as active food packaging. Indonesian Journal of Chemistry 2018; 18(1), 82-91.
X Chen, M Fan, H Tan, B Ren, G Yuan, Y Jia, J Li, D Xiong, Xiaodong Xing, X Niu and X Hu. Magnetic and self-healing Chi/Alg hydrogel encapsulated gelatin microspheres via covalent cross-linking for drug delivery. Materials Science and Engineering: C 2019; 101, 619-629.
C Qiao, X Ma, X Wang and L Liu. Structure and properties of chi films: Effect of the type of solvent acid. LWT 2021; 135, 109984.
A Djelad, A Mokhtar, A Khelifa, A Bengueddach and M Sassi. Alginate-whey an effective and green adsorbent for crystal violet removal: Kinetic, thermodynamic and mechanism studies. International Journal of Biological Macromolecules 2019; 139, 944-954.
P Karthikeyan, HAT Banu and S Meenakshi. Synthesis and characterization of metal loaded Chi/Alg biopolymeric hybrid beads for the efficient removal of phosphate and nitrate ions from aqueous solution. International Journal of Biological Macromolecules 2019; 130, 407-418.
AS Shoubagya, A Moorthi and M Prabaharan. Reparation and characterization of Chi/Pectin/ZnO porous films for wound healing. International Journal of Biological Macromolecules 2020; 157, 135-145.
V Nurmayanti and E Hastuti. Karakterisasi sifat fisis membran Polimer Matrik Komposit (PMC) dari karbon aktif tempurung kelapa untuk adsorbsi logam berat pada minyak goreng bekas. Jurnal Neutrino: Jurnal Fisika dan Aplikasinya 2014; 6(2), 119-128.
GA Dakroury, EAA El-Shazly and HS Hassan. Preparation and characterization of ZnO/Chi nanocomposite for Cs(I) and Sr(II) sorption from aqueous solutions. Journal of Radioanalytical and Nuclear Chemistry 2021; 330, 159-174.
A El-Houssaine, Z Hanani, N Eladlani, M Rhazi and M Taourirte. Chitosan microspheres/sodium alginate hybrid beads: An efficient green adsorbent for heavy metals removal from aqueous solutions. Sustainable Environment Research 2019; 29(1), 5.
R Kumar, D Varandani, BR Mehta, VN Singh, Z Wen, X Feng and K Mullen. Fast response and recovery of hydrogen sensing in Pd–Pt Nanoparticle–Graphene composite layers. Nanotechnology 2011; 22(27), 275719.
YA Prasetyo, T Rusdiana and M Abdassah. Preparation and characterization of glucosamine nanoparticle by ionic gelation method using chitosan and alginate. Indonesian Journal of Pharmaceutics 2019; 1(1), 1-10.
TPD Arajo, HB Quesada, DFD Santos, BCDS Fonseca, JZ Barbieri, R Bergamasco and MASDD Barros. Acetaminophen removal by calcium alginate/activated hydrochar composite beads: Batch and fixed-bed studies. International Journal of Biological Macromolecules 2022; 203, 553-562.
N Rahman and M Nasir. Effective removal of acetaminophen from aqueous solution using Ca (II)-doped chitosan/β-cyclodextrin composite. Journal of Molecular Liquids 2020; 301, 112454.
S Daikh, D Ouis, A Benyoucef and B Mouffok. Equilibrium, kinetic and thermodynamic studies for evaluation of adsorption capacity of a new potential hybrid adsorbent based on polyaniline and chi for acetaminophen. Chemical Physics Letters 2022; 789, 139565.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






