Comparative Analysis of the Effects of the Diterpene Alkaloid Napelline and its Derivative on the MitoK+ATP Channel and mPTP in Rat Liver and Heart Mitochondria
DOI:
https://doi.org/10.48048/tis.2025.10483Keywords:
Napelline, 1-O-benzоylnаpelline, MitоK ATP -chаnnel, mPTP, Mitоchondria, Liver, HeartAbstract
Mitochondria play crucial role in cellular energy metabolism and ion homeostasis, and their dysfunction is associated with various pathological conditions. The mitochondrial ATP-sensitive potassium channel (mitoK+ATP channel) and the mitochondrial permeability transition pore (mPTP) are considered key regulators of mitochondrial function and potential targets for cytoprotective therapies. This study examined the effects of the diterpene alkaloid napelline and its derivative, 1-O-benzoylnapelline hydrochloride, on mitoK⁺ATP channel activity and mPTP opening in liver and heart mitochondria isolated from male Wistar rats. Mitochondrial swelling assays were used to assess mitoK⁺ATP channel activation in the presence of ATP (as an inhibitor), and calcium-induced swelling was used to evaluate mPTP opening. Napelline (50 μM) activated the activity of the liver mitoK+ATP channel by 191.0 ± 2.3% compared to the control, and 1-O-benzoylnapelline (10 μM) - by 208%. In heart mitochondria, these values were 127.6 ± 3.1% and 156.5 ± 2.5%, respectively. Both compounds inhibited mPTP opening, with 1-O-benzoylnapelline showing greater efficacy: At 10 µM, it suppressed swelling by 92.3 ± 2.5% in liver and 78.6 ± 2.4% in heart mitochondria. Liver mitochondria exhibited a more pronounced response, indicating tissue-specific sensitivity. The increased activity of 1-O-benzoylnapelline may be attributed to the acylation of the C-1 hydroxyl group, which likely enhances its interaction with mitochondrial membranes. These results indicate that 1-O-benzoylnapelline is a more effective modulator of mitochondrial ion channels than napelline and may serve as a promising lead compound for the development of novel cytoprotective agents, warranting further investigation.
HIGHLIGHTS
The higher influence of 1-O-benzoylnapelline on the activity of the mitoK+ATP-channel of the liver and heart of rats compared to the napelline alkaloid can be explained by the fact that the benzoyl group is attached to the C-1 position. According to the results of the study, 1-O-benzoylnapelline at low concentrations reliably activated the mitoK+ATP-channel of the liver and heart of rats, and also exhibited membrane-active properties, more strongly inhibiting the Ca2+-dependent conductive pore compared to the alkaloid napelline.
GRAPHICAL ABSTRACT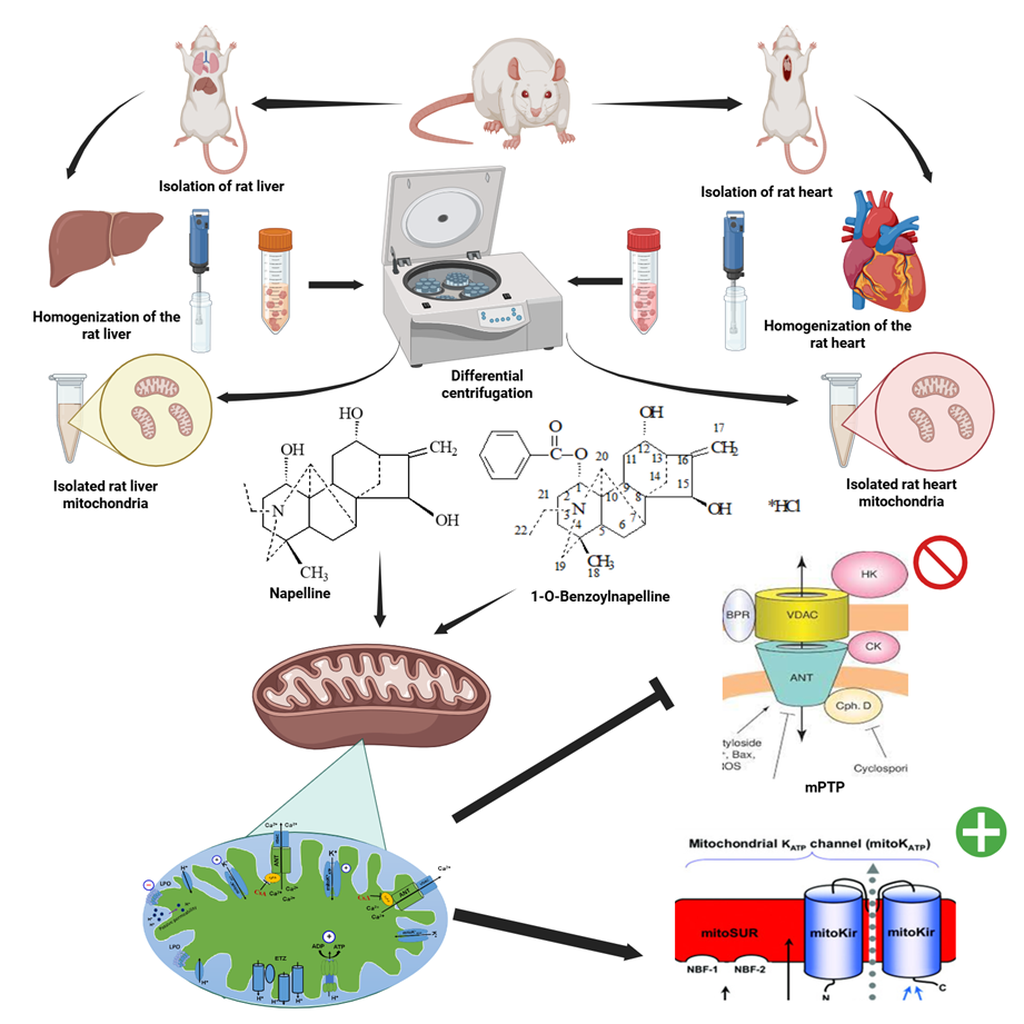
Downloads
References
A Szewczyk and L Wojtczak. Mitochondria as a pharmacological target. Pharmacological Reviews 2002; 54(1), 101-127.
M Akao, A Ohler, B O'Rourke and E Marbán. Mitochondrial ATP-sensitive potassium channels inhibit apoptosis induced by oxidative stress in cardiac cells. Circulation Research 2001; 88(12), 1267-1275.
ADT Costa, CL Quinlan, A Andrukhiv, IC West, M Jaburek and KD Garlid. The direct physiological effects of mitoKATP opening on heart mitochondria. American Journal of Physiology-Heart and Circulatory Physiology 2006; 290(1), H406-H415.
DJ Hausenloy, R Schulz, H Girao, BR Kwak, D De Stefani, R Rizzuto, P Bernardi and F Di Lisa. Mitochondrial ion channels as targets for cardioprotection. Journal of Cellular and Molecular Medicine 2020; 24(13), 7102-7114.
RB Strutynskyi, RA Rovenets and OOMoibenko. Mechanisms of cardiprotective activity of domestic activator of КАТP channels flokali. Tavricheskiy Medico-Biological Vestnik 2012; 15(3), 226-229.
H Ardehali and B O’Rourke. Mitochondrial K(ATP) channels in cell survival and death. Journal of Molecular and Cellular Cardiology 2005; 39(1), 7-16.
GD Mironova, MI Shigaeva, EN Gritsenko, SV Murzaeva, EL Germanova, OS Gorbacheva and LLD Ukyanova. Activity of mitochondrial ATP-dependent potassium channel in animals with different resistance to hypoxia before and after the course of hypoxic training. Bulletin of Experimental Biology and Medicine 2011; 151(1), 25-29.
AO Garlid, M Jaburek, JP Jacobs and KD Garlid. Mitochondrial reactive oxygen species: Which ROS signals cardioprotection? American Journal of Physiology-Heart and Circulatory Physiology 2013; 305, 960-968.
KD Garlid, P Paucek, V Yarov-Yarovoy, HN Murray, RB Darbenzio, AJ D’Alonzo, NJ Lodge, MASmith and GJ Grover. Cardioprotective effect of diazoxide and its interaction with mitochondrial ATP‐sensitive K+ channels: Possible mechanism of cardioprotection. Circulation Research 1997; 81(6), 1072‐1082.
OS Gorbacheva, NI Venediktova and GD Mironova. Study of the kinetics and regulation of the potassium cycle. Pathogenesis 2011; 9(3), 26-27.
Y Nakai, H Horimoto, S Mieno and S Sasaki. Mitochondrial ATP-sensitive potassium channel plays a dominant role in ischemic preconditioning of rabbit heart. European Surgical Research 2001; 33(2), 57-63.
GJ Grover and KD Garlid. ATP-sensitive potassium channels: A review of their cardioprotective pharmacology. Journal of Molecular and Cellular Cardiology 2000; 32(4), 677-695.
P Bernardi, A Rasola, M Forte and G Lippe. The mitochondrial permeability transition pore: Channel formation by F-ATP synthase, integration in signal transduction, and role in pathophysiology. Physiological Reviews 2015; 95(4), 1111-1155.
J Sileikyte and M Forte. The mitochondrial permeability transition in mitochondrial disorders. Oxidative Medicine and Cellular Longevity 2019; 2019(1), 3403075.
JJ Lemasters, TP Theruvath, Z Zhong and AL Nieminen. Mitochondrial calcium and the permeability transition in cell death. Biochimica et Biophysica Acta (BBA)-Bioenergetics 2009; 1787(11), 1395-1401.
A Rasola and P Bernardi. Mitochondrial permeability transition in Ca2+-dependent apoptosis and necrosis. Cell Calcium 2011; 50(3), 222-233.
P Bernardi, C Gerle, AP Halestrap, EA Jonas, J Karch, N Mnatsakanyan, E Pavlov, SS Sheu and AA Soukas. Identity, structure, and function of the mitochondrial permeability transition pore: Controversies, consensus, recent advances, and future directions. Cell Death & Differentiation 2023; 30, 1869-1885.
M Crompton, H Ellinger and A Costi. Inhibition by cyclosporin A of a Ca2+-dependent pore in heart mitochondria activated by inorganic phosphate and oxidative stress. Biochemical Journal 1988; 255(1), 357-360.
C Piot, P Croisille, P Staat, H Thibault, G Rioufol, N Mewton, R Elbelghiti, TT Cung, E Bonnefoy, D Angoulvant, C Macia, F Raczka, C Sportouch, G Gahide, G Finet, X André-Fouët, D Revel, G Kirkorian, JP Monassier, G Derumeaux and M Ovize. Effect of cyclosporine on reperfusion injury in acute myocardial infarction. New England Journal of Medicine 2008; 359(5), 473-481.
DK Muratova, NA Ergashev and MI Asrarov. Effect of talatisamine and its derivate 14-O-benzoyltalatisamine on functional state of rat liver and heart mitochondria. Biomedical & Pharmacology Journal 2023; 16(4), 2333-2343.
P Bernardi and FD Lisa. The mitochondrial permeability transition pore: Molecular nature and role as a target in cardioprotection. Journal of Molecular and Cellular Cardiology 2015; 78, 100-106.
A Nicolli, E Basso, V Petronilli, RM Wenger and P Bernardi. Interactions of cyclophilin with the mitochondrial inner membrane and regulation of the permeability transition pore, and cyclosporin A-sensitive channel. Journal of Biological Chemistry 1996; 271(4), 2185-2192.
AP Halestrap. What is the mitochondrial permeability transition pore? Journal of Molecular and Cellular Cardiology 2009; 46(6), 821-831.
UG Gayibov, EJ Komilov, RN Rakhimov, NA Ergashev, NG Abdullajanova, MI Asrarov and TF Aripov. Influence of new polyphenol compound from Euphorbia plant on mitochondrial function. The Journal of Microbiology, Biotechnology and Food Sciences 2019; 8(4), 1021-1025.
MD Kh, NA Ergashev, AV Shkinev, MI Asrarov and UK Kurbanov. The effects of songorine on the activity of the ATP-dependent K+-channel and the state of the megapore of rat liver mitochondria Exp. Clinical Pharmacology 2021; 84, 12-15.
DK Muratova, NA Ergashev, JJ Sobirov, UKH Kurbanov and MI Asrarov. Effects of diterpene alkaloids on lipid peroxidation in mitochondria. Nova Biotechnologica et Chimica 2021; 20(2), e850.
MK Pozilov, U Gayibov, MI Asrarov, NG Abdulladjanova, HS Ruziboev and TF Aripov. Physiological alterations of mitochondria under diabetes condition and its correction by polyphenol gossitan. Journal of Microbiology, Biotechnology and Food Sciences 2022; 12(2), e2224.
Z Ernazarov, MK Pozilov, MI Asrarov and SN Zhurakulov. Effects of dihydroquercetin, 1-aryltetrahydroisoquinoline, and conjugate on the functional condition mitochondrial membrane of the rat liver. Nova Biotechnologica et Chimica 2023; 22(1), e1414.
MMN Sultankhodzhaev, LV Beshitaishvili, MS Yunusov and SY Yunusov. Alkaloids of Aconitum karakolicum. Structure of acetylnapelline. Chemistry of Natural Compounds 1978; 4, 407-409.
FN Dzhakhangirov, FM Tursunkhodzhaeva, MN Sultankhodzhaev and BT Salimov. Spasmolytic activity of diterpenoid alkaloids and their derivatives. Chemistry of Natural Compounds 2013; 49, 702-706.
MN Sultankhodzhaev, B Tashkhodzhaev, KK Turgunov, UK Kurbanov and NI Mukarramov. Structure and conformational analysis of napelline-type diterpenoid alkaloids. Chemistry of Natural Compounds 2017; 53(1), 99-104.
WC Schneider and GH Hogeboom. Cytochemical studies of mammalion tissues: The isolation of cell components by differential centrifugation. Cancer Research 1951; 11, 1-22.
GL Peterson. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Analytical Biochemistry 1977; 83, 346-356.
OB Vadzyuk and SA Kosterin. Diazoxide-induced swelling of rat myometrial mitochondria as evidence of activation of the ATP-sensitive K⁺-channel. Ukrainian Biochemical Journal 2008; 80(5), 45-51.
L He and JJ Lemasters. Heat shock suppresses the permeability transition in rat liver mitochondria. Journal of Biological Chemistry 2003; 278, 16755-16760.
A Paggio, V Checchetto, A Campo, R Menabò, G Di Marco, F Di Lisa, I Szabo, R Rizzuto and DD Stefani. Identification of an ATP‐sensitive potassium channel in mitochondria. Nature 2019; 72(7771), 609‐613.
D Garlid, P Dos Santos, ZJ Xie, ADT Costa and P aucek. Mitochondrial potassium transport: The role of the mitochondrial ATP-sensitive K+ channel in cardiac function and cardioprotection. Biochimica et Biophysica Acta-Bioenergetics 2003; 1606(1-3), 1-21.
FN Dzhakhangirov, MN Sultankhodzhaev, B Tashkhodzhaev and BT Salimov. Diterpenoid alkaloids as a new class of antiarrhythmic agents. Structure-activity relationship. Chemistry of Natural Compounds 1997; 33, 190-202.
L Zhang, M Siyiti, J Zhang, M Yao and F Zhao. Anti-inflammatory and anti-rheumatic activities in vitro of alkaloids separated from Aconitum soongoricum Stapf. Experimental and Therapeutic Medicine 2021; 21(5), 493.
A Wrzosek, B Augustynek, M Żochowska and A Szewczyk. Mitochondrial potassium channels as druggable targets. Biomolecules 2020; 10(8), 1200.
EL Holmuhamedov, A Jahangir, A Oberlin, A Komarov, M Colombini and A Terzic. Potassium channel openers are uncoupling protonophores: implication in cardioprotection. FEBS Letters 2004; 568(1-3), 167-170.
H Zhu, J Dong, W Zhu, H Ding and Z Zhou. ATP-dependent potassium channels involved in cardiac protection induced by intermittent hypoxia against ischemia-reperfusion injury. Life Sciences 2003; 73(10), 1275-1287.
MK Paul, K Rajinder and AK Mukhopadhyay. Characterization of rat liver mitochondrial permeability transition pore by using mitochondrial swelling assay. African Journal of Pharmacy and Pharmacology 2008; 2(2), 14-21.
A Panov, S Dikalov, N Shalbuyeva, R Hemendinger, JT Greenamyre and J Rosenfeld. Species- and tissue-specific relationships between mitochondrial permeability transition and generation of ROS in brain and liver mitochondria of rats and mice. American Journal of Physiology-Cell Physiology 2007; 292(2), 708-718.
R Endlicher, P Krivakova, H Lotkova, M Milerova, Z Drahota and Z Červinkova. Tissue specific sensitivity of mitochondrial permeability transition pore to Ca2+ ions. Acta medica (Hradec Králové) 2009; 52(2), 69-72.
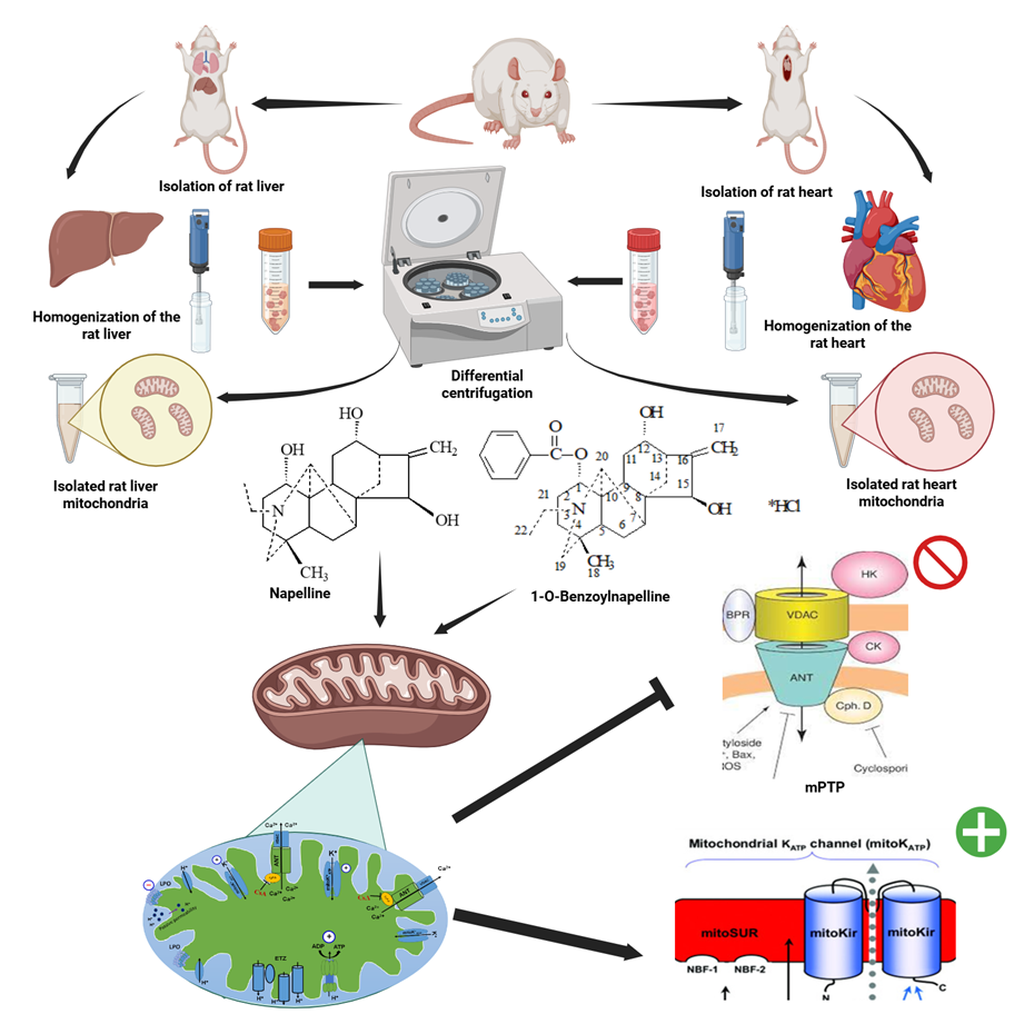
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






