Synthesis of AgMn₃O₄/C and AgSn/C Cathodes and Pd/C Anode Catalyst for Sorbitol Membraneless Alkaline Fuel Cells
DOI:
https://doi.org/10.48048/tis.2025.10462Keywords:
Alkaline fuel cell, Catalyst, Sorbitol, Anode, Cathode, Fuel cell, Oxidation reaction, Reduction reaction, Alkaline membraneless fuel cell, Catalyst, Sorbitol, Anode, Cathode, Fuel cell, Oxidation reaction, Reduction reactionAbstract
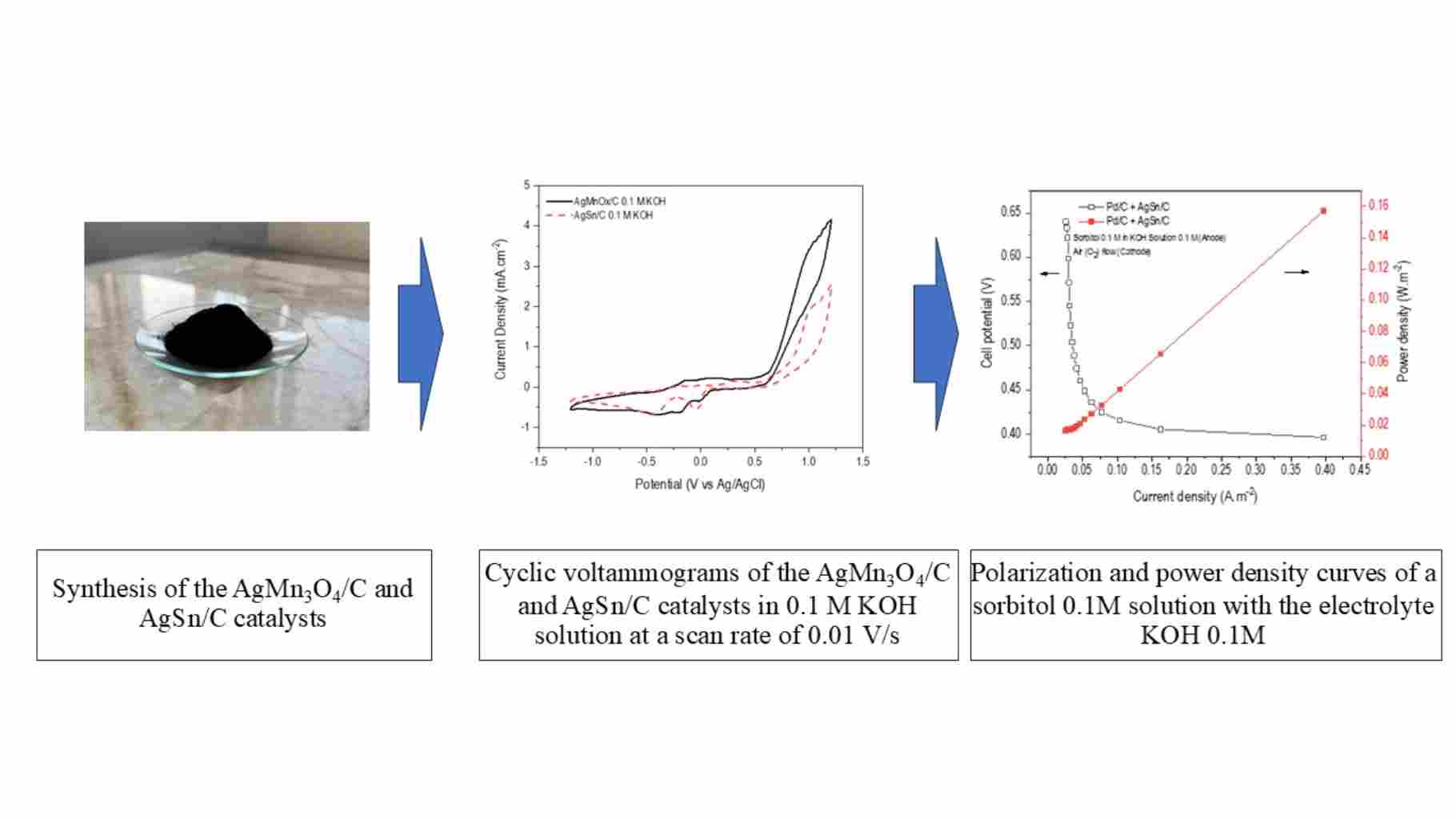
This study explored the synthesis of catalysts for sorbitol membraneless alkaline fuel cells (SMAFC) that target both cathode and anode fuels. The synthesized catalysts included AgMn3O4/C, AgSn/C, and Pd/C for the cathode and anode. This research examined the electrochemical properties of alkaline fuel oxidation by employing sorbitol as the fuel. Various sorbitol concentration ratios, ranging from 0.1 to 0.5 M per 0.1 M alkaline KOH solution, were analyzed using cyclic voltammetry with a scan rate of 0.01 V/s. At a concentration of 0.1 M sorbitol, the Pd/C catalyst was identified as an anode catalyst. The oxidation sites exhibited the greatest potential difference within the range of –0.5 to 0.5 V, resulting in a maximum current density of 0.75 mA/cm–2. The AgMn3O4/C and AgSn/C catalysts were tested as cathode side catalysts in the reduction reaction using sorbitol solution concentrations ranging from 0.1 to 0.5 M per 0.1 M alkaline KOH solution. The reduction reaction of the catalyst decreased with increasing sorbitol concentration. It appears that hydrocarbon adsorption did not result in any oxidation reactions. As sorbitol fuel crosses from the anode to the cathode side, the cathode catalyst must be resistant to oxidation reactions. An alkaline membraneless sorbitol fuel cell was tested, achieving peak performance with specific catalyst pairings. The cell, fueled by a 0.1 M sorbitol solution and 0.1 M potassium hydroxide at the anode (2.5 mL/min flow) and air at the cathode (3 mL/min flow), generated a maximum current density of 0.40 A/m² and a maximum power density of 0.155 W/m². These results were observed using a Pd/C catalyst for the anode and an AgSn/C catalyst for the cathode. The average calculated value of the number of electrons involved in the catalytic reaction (n) was 3.20.
HIGHLIGHTS
- The AgMn₃O₄/C and AgSn/C catalysts at 0.01 V/s dilution influenced the reduction reaction. Effective performance requires cathode oxidation resistance.
- Hydrodynamic voltammetry and RDE confirmed the good kinetics and favorable conditions of the AgSn/C catalyst, with an average electron transfer number of 3.20.
- The experiments revealed that the Pd/C anode and AgSn/C cathode yielded the highest current and power densities.
GRAPHICAL ABSTRACT
Downloads
References
A Harjanne and JM Korhonenc. Abandoning the concept of renewable energy. Energy Policy 2019; 127, 330-340.
T Seetawan. Alternative energy: Future energy. Journal of Industrial Technology and Innovation 2022; 1(1), 1.
W Palasai and P Phrommes. A case studying of light source used in combination of small-sized regenerative fuel cell system. Princess of Naradhiwas University Journal 2017; 9(1), 86-99.
O Muneeb, E Do, D Boyd, J Perez and JL Haan. PdCu/C anode catalysts for the alkaline ascorbate fuel cell. Applied Energy 2019; 235, 473-479.
S Lekapat and S Teekasap. Fuel cell. EAU Heritage Journal Science and Technology 2013; 7(1), 1-10.
E Gülzow. Alkaline fuel cells. Fuel Cells 2004; 4(4), 251-255.
F Bidault and PH Middleton. Alkaline fuel cells: Theory and application. In: TM Letcher (Ed.). Comprehensive Renewable Energy 2012; 4, 179-202.
LJ Torres-Pacheco, L Álvarez-Contreras, V Lair, M Cassir, J Ledesma-García, M Guerra-Balcázar and N Arjona. Electrocatalytic evaluation of sorbitol oxidation as a promising fuel in energy conversion using Au/C, Pd/C and Au–Pd/C synthesized through ionic liquids. Fuel 2019; 250, 103-116.
I Kruusenberg, L Matisen, Q Shah, AM Kannan and K Tammeveski. Non-platinum cathode catalysts for alkaline membrane fuel cells. International Journal of Hydrogen Energy 2012; 37(5), 4406-4412.
J Weiss, H Zhang and P Zelenay. Recent progress in the durability of Fe-N-C oxygen reduction electrocatalysts for polymer electrolyte fuel cells. Journal of Electroanalytical Chemistry 2020; 875(15), 114696.
W Xu, R Zeng, M Rebarchik, A Posada-Borbon, H Li, CJ Pollock, M Mavrikakis and HD Abruna. Atomically dispersed Zn/Co-N-C as ORR electrocatalysts for alkaline fuel cells. Journal of the American Chemical Society 2024; 146(4), 2593-2603.
JH Ho, Y Li, Y Dai, T Kim, J Wang, J Ren, HS Yun and X Liu. Ionothermal synthesis of N-doped carbon supported CoMn2O4 nanoparticles as ORR catalyst in direct glucose alkaline fuel cell. International Journal of Hydrogen Energy 2021; 46(39), 20503-20515.
J Hu, H Lu, M Li, G Xiao, M Li, X Xiang, Z Lu and Y Qiao. Cobalt valence modulating in CoOx incorporated carbon nanofiber for enhanced glucose electrooxidation. Materials Reports: Energy 2022; 2(2), 100091.
Y Dai, J Ding, J Li, Y Li, Y Zong, P Zhang, Z Wang and X Liu. N, S and transition-metal co-doped graphene nanocomposites as high-performance catalyst for glucose oxidation in a direct glucose alkaline fuel cell. Nanomaterials 2021; 11(1), 202.
T Suzuki, M Fujino, and Y Yamada. Formation of a palladium catalyst layer using an anionic Surfactant and subsequent fabrication of a glucose fuel cell. Journal of Chemical Education 2023; 100(12), 4780-4785.
J Huang, P Simons, Y Sunada, J Rupp and S Yagi. Pt-catalyzed d-glucose oxidation reactions for glucose fuel cells. Journal of The Electrochemical Society 2021; 168, 064511.
X Xu, X Dong, D Li, M Qi and H Huang. ZIF-8-derived three-dimensional nitrogen-doped porous carbon as a Pt catalyst support for electrocatalytic oxidation of glucose in a glucose fuel cell. ACS Applied Energy Materials 2023; 6(5), 2886-2896.
D Basu and S Basu. A study on direct glucose and fructose alkaline fuel cell. Electrochimica Acta 2010; 55(20), 5775-5779.
T Liu. Glucose fuel cells and membranes: A brief overview and literature analysis. Sustainability 2022; 14(14), 8376.
Z Daşdelen, A Özcan and A Özcan. Preparation of anode catalysts for sorbitol electrooxidation based on the nanocomposites of fumed silica, reduced graphene oxide and gold nanoparticles. International Journal of Hydrogen Energy 2021; 46(55), 28121-28133.
MB Askari, P Salarizadeh and AD Bartolomeo. NiCo2O4-rGO/Pt as a robust nanocatalyst for sorbitol electrooxidation. International Journal of Energy Research 2022; 46(5), 6745-6754.
LJ Torres-Pacheco, AD Leon-Rodriguez, L Álvarez-Contreras, M Guerra-Balcazar and N Arjona. Sorbitol electro-oxidation reaction on sub < 10 nm PtAu bimetallic nanoparticles. Electrochimica Acta 2020; 353, 136593.
LJ Torres-Pacheco, AD Leon-Rodriguez, JA Bañuelos, L Álvarez-Contreras, M Guerra-Balcázar and N Arjona. Electrocatalytic oxidation of sorbitol on PdxAuy/C bimetallic nanocatalysts. Fuel 2022; 314, 122788.
C Liu, X Yang, H Yuan, Z Zhou and D Xiao. Preparation of silver nanoparticle and its application to the determination of ct-DNA. Sensors 2007; 7(5), 708-718.
DVKD Sara, A Rouhollahi, SM Pourmortazavi and M Shamsipur. High current density chronopotentiometric electrosynthesis and SEM characterization of hexanethiol-monolayer-protected silver planar nanotriangles (Ag@C6SH). Journal of Nanomaterials 2016; 1, 1-12.
R Tholkappiyan, AN Naveen, K Vishista and F Hamed. Investigation on the electrochemical performance of hausmannite Mn3O4 nanoparticles by ultrasonic irradiation assisted co-precipitation method for supercapacitor electrodes. Journal of Taibah University for Science 2018; 12(5), 669-677.
BGS Raj, AM Asiri, JJ Wu and S Anandan. Synthesis of Mn3O4 nanoparticles via chemical precipitation approach for supercapacitor application. Journal of Alloys and Compounds 2015; 636(7), 234-240.
SZ Kure-Chu and H Yashiro. Corrosion resistance of multilayered Sn/Ag3Sn films electroplated on Cu alloys for highly reliable automotive connectors. Journal of The Electrochemical Society 2014; 161(10), C441-C449.
W Chen, Q Zhou, F Wan and T Gao. Gas sensing properties and mechanism of nano-SnO2-based sensor for hydrogen and carbon monoxide. Journal of Nanomaterials 2012; 2012(20), 1-9.
LY Liang, ZM Liu, HT Cao and XQ Pan. Microstructural, optical, and electrical properties of SnO thin films prepared on quartz via a two-step method. ACS Applied Materials & Interfaces 2010; 2(4), 1060-1065.
F Akbar, M Tariq, HU Khan, J Khan, MK Uddin, SS Ahmed and A Rahim. Development of Ag-Ni NPs loaded on MWCNTs for highly sensitive, selective and reproducible nonenzymatic electrochemical detection of glucose. Journal of Materials Science: Materials in Electronics 2021; 32(12), 16166-16181.
M Khan, M Khan, M Kuniyil, SF Adil, A Al-Warthan, HZ Alkhathlan, W Tremel, MN Tahir and MRH Siddiqui. Biogenic synthesis of palladium nanoparticles using Pulicaria glutinosa extract and their catalytic activity towards the Suzuki coupling reaction. Dalton Transactions 2014; 43(24), 9026-9031.
J Castillo-Rodríguez, C Tzompantzi-Flores, Y Piña-Pérez, F Tzompantzi, P Salinas-Hernández, F Morales-Anzures, CE Santolalla-Vargas and R Gómez. High photoactivity of Bi2O2(CO3)/Zn5(CO3)2(OH)6 prepared by a facile one-pot synthesis for the efficient degradation of phenol under UV light. Journal of Photochemistry and Photobiology A: Chemistry 2020; 402(CO3), 112816.
S Yu,Q Han,Y Yang, X Ma, L Gao, S Yan, Y Wu and L Lu. Electrochemical performance of the Pd/C catalyst synthesized by polyol process. International Journal of Electrochemical Science 2019; 14(8), 7871-7883.
K Iwase, T Kamimura and I Honma. Ag-Sn intermetallic compounds synthesized via mechanical alloying as electrocatalysts for CO2 reduction reaction. Electrochemistry 2024; 92(7), 077001.
IG Kim, F Ghani, KY Lee, S Park, S Kwak, HS Kim, IW Nah and JC Lim. Electrochemical performance of Mn3O4 nanorods by N-doped reduced graphene oxide using ultrasonic spray pyrolysis for lithium storage. International Journal of Energy Research 2020; 44(6), 1-14.
MA Kostowskyj, DW Kirk and SJ Thorpe. Ag and Ag–Mn nanowire catalysts for alkaline fuel cells. International Journal of Energy Research 2010; 35(11), 5666-5672.
C Chakkrapong, S Kantrakorn and B Sitanan. Efficiency of cathode catalyst between Ag/C and AgMnxOy/C for glucose membraneless alkaline fuel cell. Journal of Applied Research on Science and Technology 2022, 21(2), 126-136.
R Pittkowski, P Krtil and J Rossmeis. Rationality in the new oxygen evolution catalyst development. Current Opinion in Electrochemistry 2018; 12, 218-224.
G Koscher and K Kordesh. Can refillable alkaline methanol-air systems replace metal-air cells? Journal of Power Sources 2004; 136(2), 215-219.
J Qiao, L Xu, L Ding, P Shi, R Baker and J Zhang. Effect of KOH concentration on the oxygen reduction kinetics catalyzed by heat-treated Co-Pyridine/C electrocatalysts. International Journal of Electrochemical Science 2013; 8(1), 1189-1208.
M Chatenet, MB Molina-Concha, N El-Kissi, G Parrour and JP Diard. Direct rotating ring-disk measurement of the sodium borohydride diffusion coefficient in sodium hydroxide solutions. Electrochimica Acta 2009; 54(18), 4426-4435.
M Hunsom. PEM fuel cell and electrochemical analysis. Chulalongkorn University Press, Bankok, 2012.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






