The Effect of Various Extraction Methods and Solvents on the Phytochemical Contents and Antioxidant Capacities of Safflower Florets (Carthamus Tinctorius L.) from South Sulawesi
DOI:
https://doi.org/10.48048/tis.2024.7576Keywords:
Safflower, Carthamus tinctorius, Kasumba turate, flavonoids, Phenolics, Carotenoids, Radical scavenging, Phosphomolybdenum assay, FRAP, CUPRACAbstract
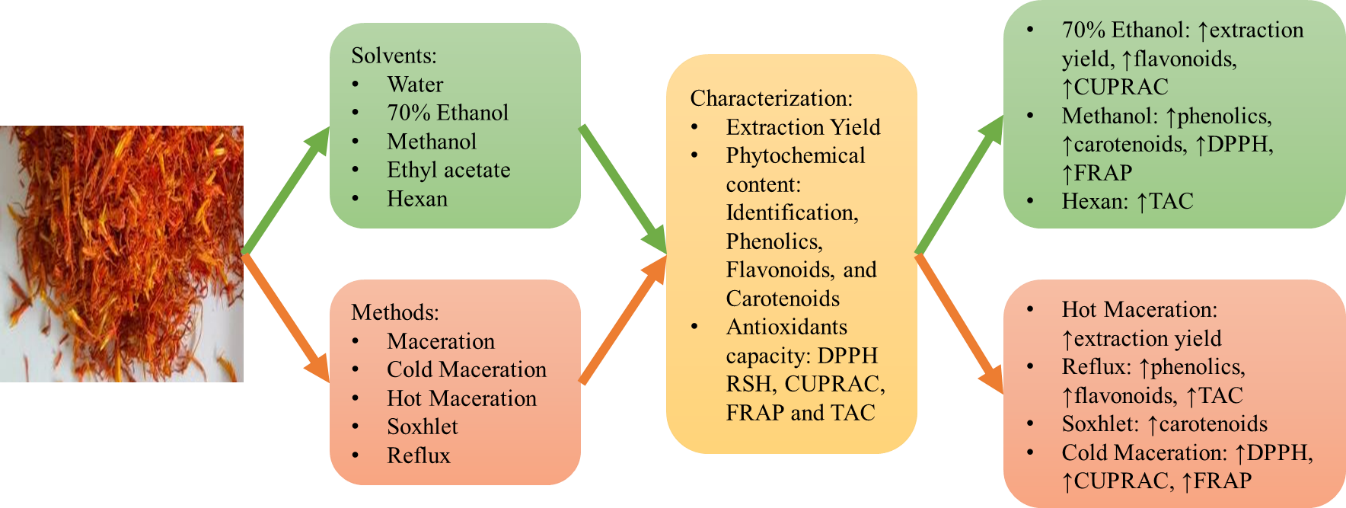
Safflower is a traditional medicine commonly applied for the treatment of varicella in South Sulawesi. The aim of this study is to study the effect of extraction methods and solvents on phytochemical content and antioxidant capacity in the extraction of safflower. The study began with sample extraction with various solvents, i.e., water, ethanol 70, methanol, ethyl acetate and hexane and various methods, i.e., reflux, Soxhlet, hot maceration, cold maceration and room temperature maceration. Each extract was standardized for its phytochemical content, i.e., phenolics, flavonoids and carotenoids and the antioxidant capacities were also determined by the radical scavenging 2,2-diphenyl-1-picrylhydrazyl, total antioxidant capacity, cupric ion reducing antioxidant capacity and Ferric reducing antioxidant power. The results indicated that the optimum total phenolic content was obtained when the florets were refluxed with methanol, flavonoids were refluxed with 70 % ethanol and carotenoids were Soxhleted with methanol. The research data also showed that 70 % ethanol and methanol have maximal antioxidant capacity. The results conclude that 70 % ethanol and methanol as solvents have the potential to extract the highest levels of phytochemical content and antioxidant substances.
HIGHLIGHTS
- The phytochemical content and antioxidant capacity of safflower floret extract are influenced by the selection of solvent and extraction method.
- Polar solvents (70 % ethanol or methanol) can efficiently extract higher levels of phytochemical components and antioxidant substances from safflower florets.
- The hot extraction approach exhibits a higher potential for extracting phytochemical content compared to the cold extraction method, but its antioxidant capacity exhibits the opposite.
- The application of cold maceration, combined with methanol or 70 % ethanol as solvents, has been identified as a promising method for the extraction of phytochemical compounds and antioxidant substances.
GRAPHICAL ABSTRACT
Downloads
References
MI Idrus. 2021, Penyakit menular di jeneponto tahun 1949 - 1960 tinjauan sejarah kesehatan. Ph. D. Dissertation. Universitas Hasanuddin, Makassar, Indonesia.
N Tahar, F Satrianegara, R Rukmana, N Hamzah, S Rukmana, F Alwi, A Roni and M Mukhriani. Brine Shrimp Lethality, aktivitas antioksidan dan kadar total fitokimia dari ekstrak etanol kasumba turate (Carthamus tinctorius). Jurnal Farmasi Indonesia 2023; 15, 72-8.
X Zhou, L Tang, Y Xu, G Zhou and Z Wang. Towards a better understanding of medicinal uses of Carthamus tinctorius L. in traditional Chinese medicine: A phytochemical and pharmacological review. J. Ethnopharmacol. 2014; 151, 27-43.
J Asgarpanah and N Kazemivas. Phytochemistry, pharmacology and medicinal properties of Carthamus tinctorius L. Chin. J. Integr. Med. 2013; 19, 153-9.
QW Zhang, LG Lin and WC Ye. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018; 13, 20.
M Boulfia, F Lamchouri and H Toufik. Chemical analysis, phenolic content, and antioxidant activities of aqueous and organic Moroccan Juglans regia L. bark extracts. Curr. Bioact. Compd. 2020; 16, 1328-39.
Y Ma, A Meng, P Liu, Y Chen, A Yuan, Y Dai, K Ye, Y Yang, Y Wang and Z Li. Reflux extraction optimization and antioxidant activity of phenolic compounds from Pleioblastus amarus (Keng) shell. Molecules 2022; 27, 362.
M Hirondart, N Rombaut, A Fabiano-Tixier, A Bily and F Chemat. Comparison between pressurized liquid extraction and conventional Soxhlet extraction for rosemary antioxidants, yield, composition, and environmental footprint. Foods 2020; 9, 584.
E Sabdoningrum, S Hidanah and S Chusniati. Characterization and phytochemical screening of Meniran (Phyllanthus niruri linn) extract’s nanoparticles used ball mill method. Pharmacogn. J. 2021; 13, 1568-72.
N Haruna, Z Hamzah, S Syakri, I Ismail and N Hamzah. Effect of methanol extract and partition of java bark (Lannea coromandelica Houtt. Merr.) on HeLa and MCF-7. cell growth. Ad-Dawaa’ J. Pharm. Sci. 2018; 1, 71-7.
SH Alotaibi. Biophysical properties and finger print of Boswellia Sp. Burseraceae. Saudi J. Biol. Sci. 2019; 26, 1450-7.
Kementerian Kesehatan Republik Indonesia. Farmakope herbal Indonesia. 2 eds. Kementerian Kesehatan Republik Indonesia, Jakarta, Indonesia, 2017, p. 561.
AF Ahmed, FAK Attia, Z Liu, C Li, J Wei and W Kang. Antioxidant activity and total phenolic content of essential oils and extracts of sweet basil (Ocimum basilicum L.) plants. Food Sci. Hum. Wellness 2019; 8, 299-305.
W Nurcholis, DNS Putri, Husnawati, SI Aisyah and BP Priosoeryanto. Total flavonoid content and antioxidant activity of ethanol and ethyl acetate extracts from accessions of Amomum compactum fruits. Ann. Agr. Sci. 2021; 66, 58-32.
HK Lichtenthaler. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Meth. Enzymol. 1987; 148, 350-82.
HK Lichtenthaler and C Buschmann. Chlorophylls and carotenoids: Measurement and characterization by UV-VIS spectroscopy. In: RE Wrolstad, TE Acree, H An, EA Decker, MH Penner, DS Reid, SJ Schwartz, CF Shoemaker and P Sporns (Eds.). Current protocols in food analytical chemistry. John Wiley & Sons, New York, 2001.
Z Khiya, Y Oualcadi, A Gamar, F Berrekhis, T Zair and FE Hilali. Correlation of total polyphenolic content with antioxidant activity of hydromethanolic extract and their fractions of the salvia officinalis leaves from different regions of Morocco. J. Chem. 2021; 2021, 8585313.
T Afzal, Y Bibi, M Ishaque, S Masood, A Qayyum, S Nisa, ZH Shah, H Alsamadany and G Chung. Pharmacological properties and preliminary phytochemical analysis of Pseudocaryopteris foetida (D.Don) P.D. Cantino leaves. Saudi J. Biol. Sci. 2022; 29, 1185-90.
S Sethi, A Joshi, B Arora, A Bhowmik, R Sharma and P Kumar. Significance of FRAP, DPPH, and CUPRAC assays for antioxidant activity determination in apple fruit extracts. Eur. Food Res. Tech. 2020; 246, 591-8.
C Irawan, I Putri, M Sukiman, A Utami, Ismail, R Putri, A Lisandi and A Pratama. Antioxidant activity of DPPH, CUPRAC, and FRAP methods, as well as activity of alpha-glucosidase inhibiting enzymes from Tinospora crispa (L.) stem ultrasonic extract. Pharmacogn. J. 2022; 14, 511-20.
MA Islam, RJ Brown, I O’Hara, M Kent and K Heimann. Effect of temperature and moisture on high pressure lipid/oil extraction from microalgae. Energ. Convers. Manag. 2014; 88, 307-16.
M Premi and HK Sharma. Oil extraction optimization and kinetics from moringa oleifera (PKM 1) seeds. Int. J. Agr. Food Sci. Tech. 2013; 4, 371-8.
DB Nde and AC Foncha. Optimization methods for the extraction of vegetable oils: A review. Processes 2020; 8, 209.
M Arias, I Penichet, F Ysambertt, R Bauza, M Zougagh and Á Ríos. Fast supercritical fluid extraction of low- and high-density polyethylene additives: Comparison with conventional reflux and automatic Soxhlet extraction. J. Supercrit. Fluids 2009; 50, 22-8.
J Jegal, EJ Jeong and MH Yang. A review of the different methods applied in ginsenoside extraction from Panax ginseng and Panax quinquefolius roots. Nat. Prod. Commun. 2019; 14, 1-10.
N Salem, K Msaada, S Elkahoui, G Mangano, S Azaeiz, IB Slimen, S Kefi, G Pintore, F Limam and B Marzouk. Evaluation of antibacterial, antifungal, and antioxidant activities of safflower natural dyes during flowering. Biomed. Res. Int. 2014; 2014, 762397.
LL Zhang, K Tian, ZH Tang, XJ Chen, ZX Bian, YT Wang and JJ Lu. Phytochemistry and pharmacology of Carthamus tinctorius L. Am. J. Chin. Med. 2016; 44, 197-226.
I Adamska and P Biernacka. Bioactive substances in safflower flowers and their applicability in medicine and health-promoting foods. Int. J. Food Sci. 2021; 2021, 6657639.
R Hamsidi, W Wahyuni, A Fristiohady, MH Malaka, S Sahidin, W Ekasari, A Widyawaruyanti and AF Hafid. Steroid compounds isolation from carthamus tinctorius linn as antimalarial. Res. J. Pharm. Tech. 2020; 14, 5297-304.
E Hong and GH Kim. Comparison of extraction conditions for phenolic, flavonoid content and determination of rosmarinic acid from Perilla frutescens var. acuta. Int. J. Food Sci. Tech. 2010; 45, 1353-9.
J He, L Wu, L Yang, B Zhao and C Li. Extraction of phenolics and flavonoids from four Hosta species using reflux and ultrasound-assisted methods with antioxidant and α-glucosidase inhibitory activities. Biomed. Res. Int. 2020, 2020, 6124153.
S Kongkiatpaiboon and W Gritsanapan. Optimized extraction for high yield of insecticidal didehydrostemofoline alkaloid in Stemona collinsiae root extracts. Ind. Crop. Prod. 2013; 112, 371-4.
N Mezzomo, B Maestri, RLD Santos, M Maraschin and SR Ferreira. Pink shrimp (P. brasiliensis and P. paulensis) residue: Influence of extraction method on carotenoid concentration. Talanta 2011; 85, 1383-91.
MDA Saldaña, L Sun, SE Guigard and F Temelli. Comparison of the solubility of β-carotene in supercritical CO2 based on a binary and a multicomponent complex system. J. Supercrit. Fluids 2006; 37, 342-9.
F Bashipour and SM Ghoreishi. Experimental optimization of supercritical extraction of β-carotene from Aloe barbadensis miller via genetic algorithm. J. Supercrit. Fluids. 2012; 72, 312-9.
MN Alam, NJ Bristi and M Rafiquzzaman. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 2013; 21, 143-52.
C Andreini, I Bertini, G Cavallaro, GL Holliday and JM Thornton. Metal ions in biological catalysis: From enzyme databases to general principles. J. Biol. Inorg. Chem. 2008; 13, 1205-18.
NB Sadeer, D Montesano, S Albrizio, G Zengin and MF Mahomoodally. The versatility of antioxidant assays in food science and safety-chemistry, applications, strengths, and limitations. Antioxidants 2020; 9, 709.
A Thouri, H Chahdoura, AE Arem, AO Hichri, RB Hassin and L Achour. Effect of solvents extraction on phytochemical components and biological activities of Tunisian date seeds (var. Korkobbi and Arechti). BMC Compl. Altern. Med. 2017; 17, 248.
J Junlatat and B Sripanidkulchai. Hair growth-promoting effect of Carthamus tinctorius floret extract. Phytother. Res. 2014; 28, 1030-6.
F Ebadia, M Mohsenia and AM Alizadeh. Evaluation of antioxidant activity of Safflower florets (Carthamus tinctorius L.) as food coloring agents. J. Chem. Pharmaceut. Res. 2014; 6, 539-44.
N Salem, K Msaada, G Hamdaoui, F Limam and B Marzouk. Variation in phenolic composition and antioxidant activity during flower development of safflower (Carthamus tinctorius L.). J. Agr. Food Chem. 2011; 59, 4455-63.
N Salem, K Msaada, W Dhifi, J Sriti, H Mejri, F Limam and B Marzouk. Effect of drought on safflower natural dyes and their biological activities. EXCLI J. 2014; 13, 1-18.
N Salem, K Msaada, W Dhifi, F Limam and B Marzouk. Effect of salinity on plant growth and biological activities of Carthamus tinctorius L. extracts at two flowering stages. Acta Physiol. Plant 2014; 36, 433-45.
M Karimkhani, R Shaddel, M Khodaparast, M Vazirian and S Piri-Gheshlaghi. Antioxidant and antibacterial activity of safflower (Carthamus tinctorius L.) extract from four different cultivars. Qual. Assur. Saf. Crop. Foods 2016; 8, 565-74.
S Erbaş and M Mutlucan. Investigation of flower yield and quality in different color safflower genotypes. Agronomy 2023; 13, 956.
H Soumia, R Farah, G Imane, K Rokaya and OEHK Aminata. Assesment of safflower flowers (Carthamus tinctorius L.) as potential food antioxidants. Revue des BioRessources 2023; 13, 2-10.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






