Secretome of Hypoxia-Preconditioned Mesenchymal Stem Cells Ameliorates Hyperglycemia in Type 2 Diabetes Mellitus Rats
DOI:
https://doi.org/10.48048/tis.2024.7278Keywords:
T2DM, SH-MSCs, Hyperglycaemia, Insulin resistanceAbstract
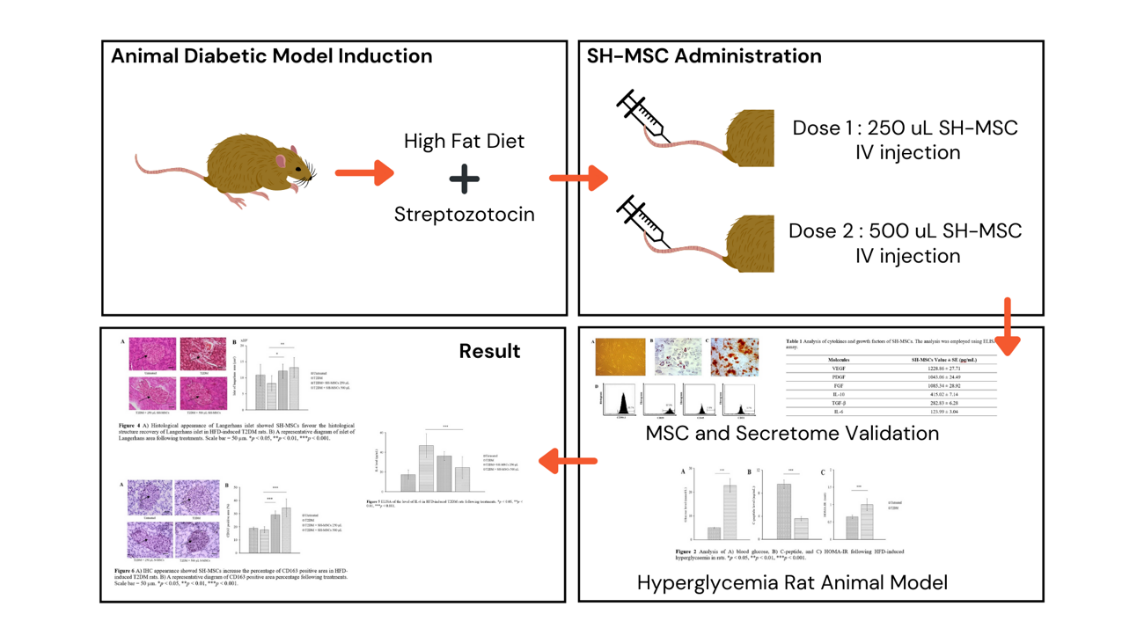
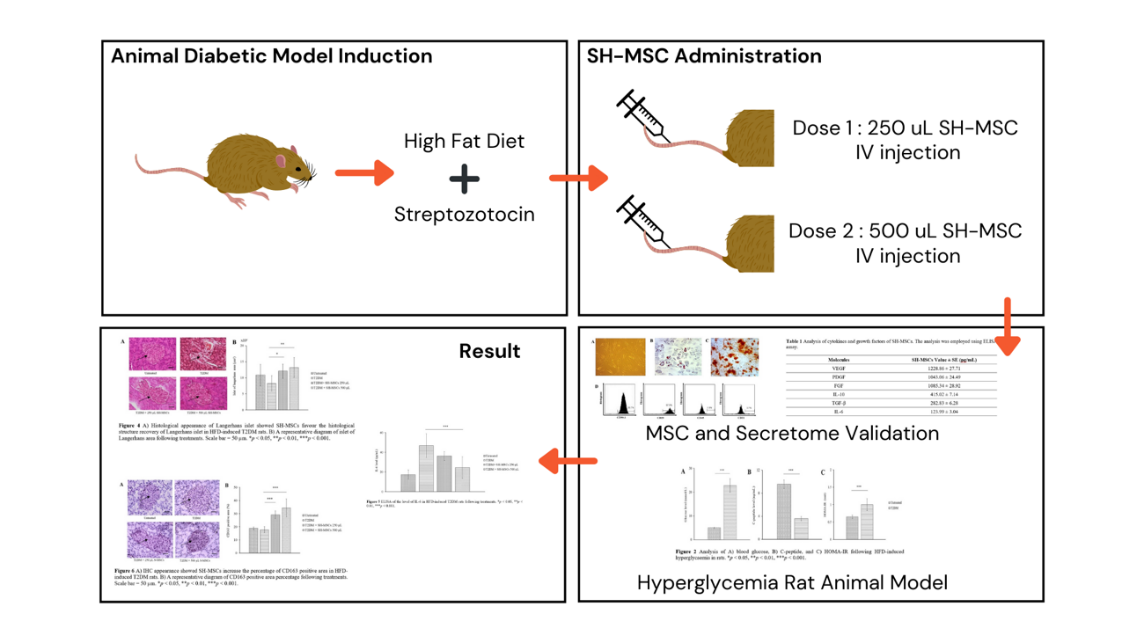
Introduction: Type 2 diabetes mellitus (T2DM) is a prevalent form of diabetes that affects 90 - 95 % of all diabetic patients. Insulin sensitizers and insulin exogenous supply could temporarily ameliorate hyperglycaemia; however, they are accompanied by side effects. As a result, new approaches are required to address insulin resistance and regenerate beta cells simultaneously. The secretome of hypoxic mesenchymal stem cells (SH-MSCs) contains various growth factors and anti-inflammatory cytokines that could potentially enhance insulin resistance and improve pancreatic function. Objectives: In this study, we performed SH-MSCs infusion to ameliorate HFD-induced hyperglycaemia in T2DM rats. Methods: We created a T2DM rat model using a combination of a high-fat diet (HFD) and streptozotocin (STZ) administration. Then, we administered SH-MSCs injection at doses of 250 and 500 µL and assessed the therapeutic effects of SH-MSCs. We also investigated the potential underlying mechanisms involved. Results: The administration of SH-MSCs improved hyperglycemia in rats with T2DM. Infusion of SH-MSCs at 500 µL dose decreased homeostatic model assessment for insulin resistance (HOMA-IR). Histological analysis revealed that injection of SH-MSCs alleviated morphological damage of pancreas. SH-MSCs administration also inhibit the level of IL-6 and promote the expression of CD163 type 2 macrophage. Conclusion: The results of our study indicate that SH-MSCs have the potential to improve hyperglycemia and exert a protective effect on T2DM rats.
HIGHLIGHTS
- Administration of SH-MSCs effectively improved hyperglycemia and decreased insulin resistance in TD2M rats through modulation of IL-6 levels and promotes the expression of CD163 type 2 macrophage
- Histological analysis demonstrated the protective effect of SH-MSCs on pancreatic morphology
- SH-MSCs hold promise for improving hyperglycemia, insulin resistance, and providing a protective effect in TD2M
GRAPHICAL ABSTRACT
Downloads
References
Y Zheng, SH Ley and FB Hu. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018; 14, 88-98.
MAB Khan, MJ Hashim, JK King, RD Govender, H Mustafa and JA Kaabi. Epidemiology of type 2 diabetes - global burden of disease and forecasted trends. J. Epidemiol. Glob. Health 2020; 10, 107-11.
U Galicia-Garcia, A Benito-Vicente, S Jebari, A Larrea-Sebal, H Siddiqi, KB Uribe, H Ostolaza and C Martín. Pathophysiology of type 2 diabetes mellitus. Int. J. Mol. Sci. 2020; 21, 6275.
A Kanwal, N Kanwar, S Bharati, P Srivastava, SP Singh and S Amar. Exploring new drug targets for type 2 diabetes: Success, challenges and opportunities. Biomedicines 2022; 10, 331.
ND Noronha, A Mizukami, C Caliári-Oliveira, JG Cominal, JL Rocha, DT Covas, K Swiech and KC Malmegrim. Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res. Ther. 2019; 10, 131.
A Putra, I Rosdiana, DM Darlan, I Alif, F Hayuningtyas, I Wijaya, R Aryanti, FR Makarim and AD Antari. Intravenous administration is the best route of mesenchymal stem cells migration in improving liver function enzyme of acute liver failure. Folia Medica 2020; 62, 52-8.
A Putra, I Alif, N Hamra, O Santosa, AR Kustiyah, AM Muhar and K Lukman. MSC-released TGF-β regulate α-SMA expression of myofibroblast during wound healing. J. Stem Cells Regen. Med. 2020; 16, 73-9.
S Bhansali, V Kumar, UN Saikia, B Medhi, V Jha, A Bhansali and Dutta. Effect of mesenchymal stem cells transplantation on glycaemic profile & their localization in streptozotocin induced diabetic Wistar rats. Indian J. Med. Res. 2015; 142, 63-71.
DM Darlan, D Munir, NK Jusuf, A Putra, R Ikhsan and I Alif. In vitro regulation of IL-6 and TGF-ß by mesenchymal stem cells in systemic lupus erythematosus patients. Medicinski Glasnik 2020; 17, 408-13.
NF Hamra, A Putra, A Tjipta, ND Amalina and Nasihun. Hypoxia Mesenchymal stem cells accelerate wound closure improvement by controlling α-smooth muscle actin expression in the full-thickness animal model. Open Access Maced. J. Med. Sci. 2021; 9, 35-41.
NS Hwang, C Zhang, YS Hwang and S Varghese. Mesenchymal stem cell differentiation and roles in regenerative medicine. Wiley Interdiscip. Rev. Syst. Biol. Med. 2009; 1, 97-106.
Y Si, Y Zhao, H Hao, J Liu, Y Guo, Y Mu, J Shen, Y Cheng, X Fu and W Han. Infusion of mesenchymal stem cells ameliorates hyperglycemia in type 2 diabetic rats: Identification of a novel role in improving insulin sensitivity. Diabetes 2012; 61, 1616-25.
ML Quaade, P Dhumale, SGC Steffensen, HC Beck, EB Harvald, CH Jensen, L Lund, DC Andersen and SP Sheikh. Adipose-derived stem cells from type 2 diabetic rats retain positive effects in a rat model of erectile dysfunction. Int. J. Mol. Sci. 2022; 23, 1692.
SK Yap, KL Tan, NYA Rahaman, NFS Hamid, J Ooi, YS Tor, QHD Looi, LKS Tan, CW How and JB Foo. Human umbilical cord mesenchymal stem cell-derived small extracellular vesicles ameliorated insulin resistance in type 2 diabetes mellitus rats. Pharmaceutics 2022; 14, 649.
A Utami, A Putra, JW Wibowo, ND Amalina and RC Satria. Hypoxic secretome mesenchymal stem cells inhibiting interleukin-6 expression prevent oxidative stress in type 1 diabetes mellitus. Medicinski glasnik 2023; 20, 148-55.
A Putra, A Widyatmoko, S Ibrahim, F Amansyah, F Amansyah, MA Berlian, R Retnaningsih, Z Pasongka, FE Sari and B Rachmad. Case series of the first three severe COVID-19 patients treated with the secretome of hypoxia-mesenchymal stem cells in Indonesia. F1000Research 2021; 10, 228.
KC Chao, KF Chao, YS Fu and SH Liu. Islet-like clusters derived from mesenchymal stem cells in Wharton’s Jelly of the human umbilical cord for transplantation to control type 1 diabetes. PLoS One 2008; 3, e1451.
QP Xie, H Huang, B Xu, X Dong, SL Gao, B Zhang and YL Wu. Human bone marrow mesenchymal stem cells differentiate into insulin-producing cells upon microenvironmental manipulation in vitro. Differentiation 2009; 77, 483-91.
H Sunarto, S Trisnadi, A Putra, NA Sa’dyah, A Tjipta and C Chodidjah. The role of hypoxic mesenchymal stem cells conditioned medium in increasing vascular endothelial growth factors (VEGF) levels and collagen synthesis to accelerate wound healing. Indonesian J. Cancer Chemopreve. 2020; 11, 134-43.
Z Fu, ER Gilbert and D Liu. Regulation of insulin synthesis and secretion and pancreatic Beta-cell dysfunction in diabetes. Curr. Diabetes Rev. 2013; 9, 25-53.
Y Onishi, T Hayashi, KK Sato, T Ogihara, N Kuzuya, M Anai, K Tsukuda, EJ Boyko, WY Fujimoto and M Kikuchi. Fasting tests of insulin secretion and sensitivity predict future prediabetes in Japanese with normal glucose tolerance. J. Diabetes Investig. 2010; 1, 191-5.
GSN Rao, G Prema, G Priya, SB Arumugam, V Kirthivasan, R Saibabu and KM Cherian. Comparison between serum insulin levels and its resistance with biochemical, clinical and anthropometric parameters in South Indian children and adolescents. Indian J. Clin. Biochem. 2011; 26, 22-7.
R Nakamura, A Sene, A Santeford, A Gdoura, S Kubota, N Zapata and RS Apte. IL10-driven STAT3 signalling in senescent macrophages promotes pathological eye angiogenesis. Nat. Commun. 2015; 6, 7847.
AC Cevey, FN Penas, CDA Soto, GA Mirkin and NB Goren. IL-10/STAT3/SOCS3 axis is involved in the anti-inflammatory effect of benznidazole. Front. Immunol. 2019; 10, 1267.
A Sica and A Mantovani. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012; 122, 787-95.
AF Cruz, R Rohban and F Esni. Macrophages in the pancreas: Villains by circumstances, not necessarily by actions. Immun. Inflamm. Dis. 2020; 8, 807-24.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






