Chemical Synthesis of Modularly Modified Analogs of E2/NS1 Peptides on a Novel Chemically and Mechanically Stable Terpolymer of 1,6-Hexanediol Diacrylate, N-Vinylpyrrolidone on Styrene Support
DOI:
https://doi.org/10.48048/tis.2023.6840Keywords:
Polymer, Resin, Polystyrene, N-vinylpyrrolidone, PeptideAbstract
Analysis of the chemokine analogs synthesised on a terpolymer-polystyrene, N-vinylpyrrolidone, 1,6-hexanediol diacrylate crosslinked resin (PS-NVP-HDDA) showed it can be used a good solid support for the chemical synthesis of polypeptides. The polymer with a good swelling character and chemical resistance was synthesised by the radical aqueous suspension polymerisation technique. HPLC analysis of the crude peptides synthesised on PS-NVP-HDDA resin showed that it is a better substitute for most of the currently available polymer support for peptide synthesis.
HIGHLIGHTS
- This article includes the synthesis of modularly modified analogs of E2/NS1 peptides
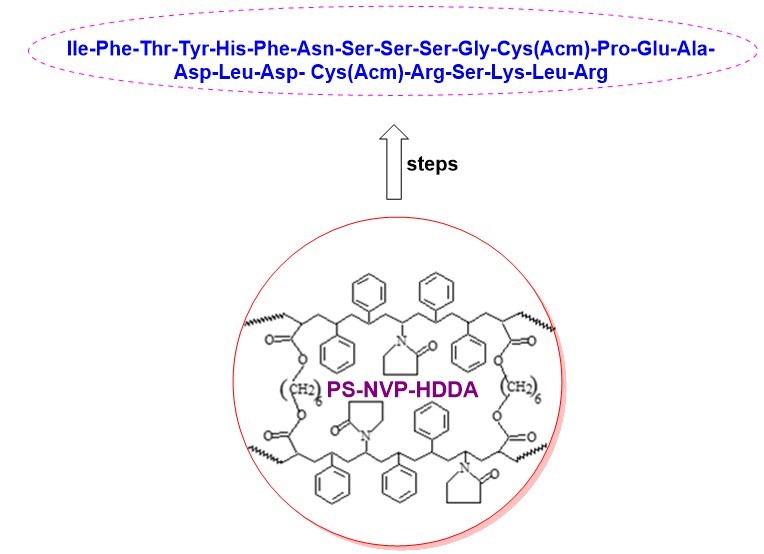
- The synthesized peptide is IIe-Phe-Thr-Tyr-His-Phe-Asn-Ser-Ser-Ser-Gly-Cys(Acm)-Pro-Glu-Ala-Asp-Leu-Asp- Cys(Acm)-Arg-Ser-Lys-Leu-Arg
- The peptide is synthesized on a chemically and mechanically stable terpolymer of 1,6-hexanediol diacrylate, N-vinylpyrrolidone on styrene support
- The polymer exhibited a good swelling character and chemical resistance
- The peptide was synthesized with great purity
GRAPHICAL ABSTRACT 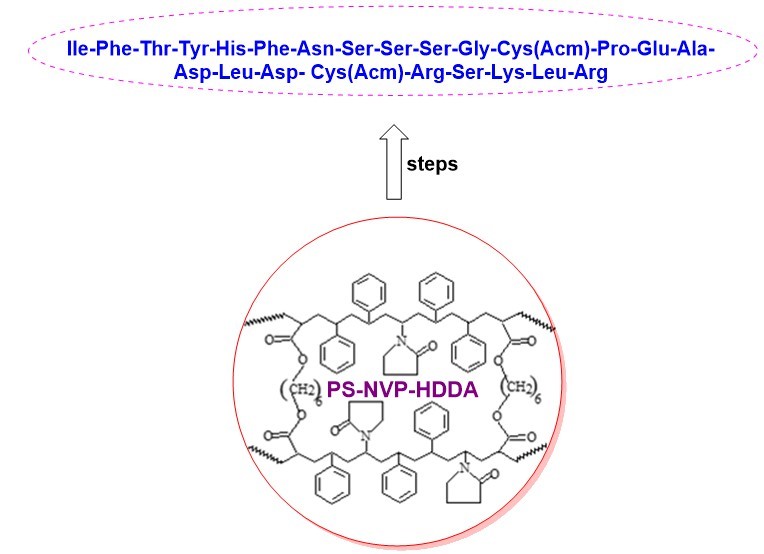
Downloads
References
RB Merrifield. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 1963; 85, 2149.
G Raos and B Zappone. Polymer adhesion: Seeking new solutions for an old problem. Macromolecules 2021; 54, 10617.
NS Schauser, DJ Grzetic, T Tabassum, GA Kliegle, ML Le, EM Susca, S Antoine, TJ Keller, KT Delaney, S Han, R Seshadri, GH Fredrickson and RA Segalman. The role of backbone polarity on aggregation and conduction of ions in polymer electrolytes. J. Am. Chem. Soc. 2020; 142, 7055.
S Ramkisson, YE Jad, A Sharma, BGDL Torre and Fernando. OctaGel Resin - A New PEG-PS-based Solid Support for Solid-Phase Peptide Synthesis. Lett. Org. Chem. 2019; 16, 935-40.
PG Sasikumar, KS Kumar, C Arunan and VNR Pillai. Synthesis and optimization of tri(propylene glycol) glycerolate diacrylate cross-linked polystyrene resin in polypeptide synthesis: Role of the macromolecular support in solid phase peptide synthesis. J. Chem. Soc., Perkin Trans. 2002; 1, 2886.
D Verma and VNR Pillai. Synthesis and characterization of cyclic PNC-28 Peptide, residues 17 - 26 (ETFSDLWKLL), a binding domain of p53. Chem. Data Collect. 2020; 27, 100367.
CJ Neufeldt, M Cortese, EG Acosta and R Bartenschlager. Rewiring cellular networks by members of the Flaviviridae family. Nat. Rev. Microbiol. 2018; 16, 125.
DE Metawlly, AN Amer, HM Mostafa, GED Elsawaf and OAE Kader. Low cost detection of hepatitis C virus RNA in HCV infected patients by SYBR Green I real-time PCR. Alexandria J. Med. 2018; 54, 481.
M Berliner and K Belecki. Synthesis of alpha-halo ethers from symmetric acetals and in situ methoxymethylation of an alcohol. Org. Synth. 2007; 84, 102.
A Carbone, R Pedicini, I Gatto, A Saccà, A Patti, G Bella and M Cordaro. Development of polymeric membranes based on quaternized polysulfones for AMFC applications. Polymers 2020; 12, 283.
J Adamek, R Mazurkiewicz, A Węgrzyk and K Erfurt. 1-Imidoalkylphosphonium salts with modulated Cα-P+ bond strength: Synthesis and application as new active α-imidoalkylating agents. Beilstein J. Org. Chem. 2017; 13, 1446.
JK Kasim, I Kavianinia, PWR Harris and MA Brimble. Three decades of amyloid beta synthesis: Challenges and advances. Front Chem. 2019; 7, 472.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






