Immune Response to the Dual Antigen Vaccine of Actinobacillus pleuropneumoniae in a Mouse Model
DOI:
https://doi.org/10.48048/tis.2023.6798Keywords:
Actinobacillus pleuropneumoniae, Apx toxin, TonB2 system, Subunit vaccineAbstract
Actinobacillus pleuropneuminiae is the major cause of invasive respiratory disease in swine. To develop an efficient subunit vaccine against pathogens, we designed a dual antigen in a single vaccine that increased the potential for an immune response. We present a dual antigen vaccine (pore-forming domain and nutritional immunity) that induces humoral and cell-mediated T-cell responses, antibodies and TH1 and TH2 cells. Mice were immunized with the dual antigen and individual antigen and then subjected to immune response analysis. A significant antibody response was observed for the dual antigen group. For the cellular immune response, CD4+ and CD8+ T-cell expansion, proinflammatory cytokine (IL-1b, IL-6) and TH2-type cytokine (IL-4, IL-10) gene expression was observed for the dual antigen. Finally, in a challenge test with A. pleuropneumoniae serotype1, the dual antigen, individual antigen and PBS vaccine conferred 100, 40 and 0 % protection. In conclusion, the dual antigen presents critical antigens and increases the efficacy of the vaccine.
HIGHLIGHTS
- The dual antigen vaccine of Actinobacillus pleuropneumonia (ApxII pore - forming domain and TonB2-the nutritional immunity antigen) were formulated as vaccines
- Mice were vaccinated, immune response analysis and challenge test were performed
- significant antibody response was observed and improved by approximately 2.5-fold for the dual antigen groups
- CD4 + and CD8 + T cell expansion was observed for the dual antigen group
- Protection rate of 100 %, 40 %, and 0 % was observed for the dual antigen, single antigen and negative control groups, respectively
GRAPHICAL ABSTRACT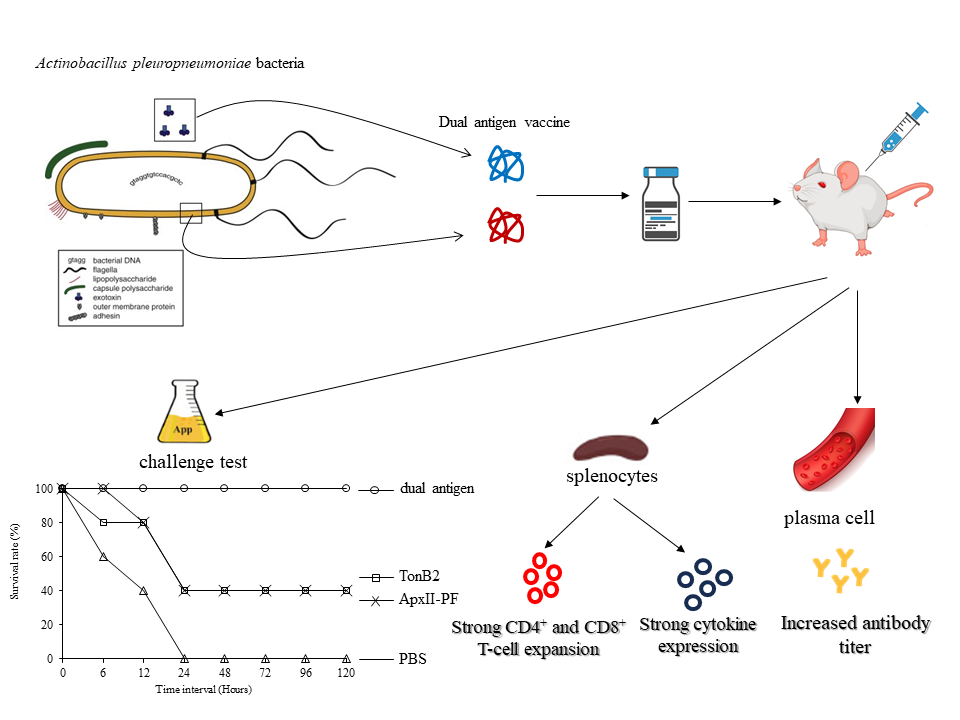
Downloads
References
B Plasencia-Muñoz, FJ Avelar-González, MDL Garza, M Jacques, A Moreno-Flores and AL Guerrero-Barrera. Actinobacillus pleuropneumoniae interaction with swine endothelial cells. Front. Vet. Sci. 2020; 7, 569370.
W Sipos, V Cvjetković, B Dobrokes and S Sipos. Evaluation of the efficacy of a vaccination program against actinobacillus pleuropneumoniae based on lung‐scoring at slaughter. Animals 2021; 11, 2778.
P Hölzen, T Warnck, S Hoy, K Schlegel, I Hennig-Pauka and H Gaumann. Comparison of protectivity and safety of two vaccines against actinobacillus pleuropneumoniae in a field study. Agriculture 2021; 11, 1143.
A Loera-Muro and C Angulo. New trends in innovative vaccine development against Actinobacillus pleuropneumoniae. Vet. Microbiol. 2018; 217, 66-75.
L Zhang, W Luo, R Xiong, H Li, Z Yao, W Zhuo, G Zou, Q Huang and R Zhou. A combinatorial vaccine containing inactivated bacterin and subunits provides protection against Actinobacillus pleuropneumoniae infection in mice and pigs. Front. Vet. Sci. 2022; 9, 902497.
V Viviani, A Biolchi and M Pizza. Synergistic activity of antibodies in the multicomponent 4CMenB vaccine. Expet. Rev. Vaccines 2022; 21, 645-58.
S Peterhans, MJA Stevens, N Cernela, X Sidler, R Stephan and S Scherrer. Draft genome sequences of two clinical Actinobacillus pleuropneumoniae serotype 19 strains from pigs in Switzerland. Microbiol. Resour. Announc. 2021; 10, e00588-21.
CC Murdoch and EP Skaar. Nutritional immunity: the battle for nutrient metals at the host-pathogen interface. Nat. Rev. Microbiol. 2022; 20, 657-70.
N Solis and SJ Cordwell. Current methodologies for proteomics of bacterial surface-exposed and cell envelope proteins. Proteomics 2011; 11, 3169-89.
J Wang, K Xiong, Q Pan, W He and Y Cong. Application of TonB-dependent transporters in vaccine development of gram-negative bacteria. Front. Cell. Infect. Microbiol. 2021; 10, 589115.
M Jung, H Won, MK Shin, M Whan Oh, S Shim I Yoon and H Sang Yoo. Development of Actinobacillus pleuropneumoniae ApxI, ApxII, and ApxIII-specific ELISA methods for evaluation of vaccine efficiency. J. Vet. Sci. 2019; 20, e2.
J Xiao, J Liu, C Bao, R Zhu, J Gu, C Sun, X Feng, C Du, W Han, Y Li and L Lei. Recombinant tandem epitope vaccination provides cross protection against Actinobacillus pleuropneumoniae challenge in mice. AMB Express 2020; 10, 123.
JN Seah, J Frey and J Kwang. The N-terminal domain of RTX toxin ApxI of Actinobacillus pleuropneumoniae elicits protective immunity in mice. Infect Immun. 2002; 70, 6464-7.
Y Cao, L Gao, L Zhang, L Zhou, J Yang, L Deng, J Zhao, C Qi and J Liu. Genome-wide screening of lipoproteins in Actinobacillus pleuropneumoniae identifies three antigens that confer protection against virulent challenge. Sci Rep. 2020; 10, 2343.
M Shao, Y Wang, C Wang, Y Guo, Y Peng J Liu, G Li, H Liu and S Liu. Evaluation of multicomponent recombinant vaccines against Actinobacillus pleuropneumoniae in mice. Acta Vet. Scand. 2010; 52, 52.
KA Pilewski, KJ Kramer and IS Georgiev. Simultaneous immunization with multiple diverse immunogens alters development of antigen-specific antibody-mediated immunity. Vaccines 2021; 9, 964.
HT Dao, WS Shin, VT Do, QL Truong, JY Choi and TW Hahn. A multivalent vaccine containing Actinobacillus pleuropneumoniae and Mycoplasma hyopneumoniae antigens elicits strong immune responses and promising protection in pigs. J. Pure Appl. Microbiol. 2021; 15, 164-74.
HC Wu, PH Yeh, KJ Hsueh, WJ Yang and Cy Chu. Recombinant ApxIV protein enhances protective efficacy against Actinobacillus pleuropneumoniae in mice and pigs. J. Appl. Microbiol. 2018; 124, 1366-76.
M Giuliani, E Bartolini, B Galli, L Santini1, PL Surdo, F Buricchi, M Bruttini, B Benucci, N Pacchiani, L Alleri, D Donnarumma, W Pansegrau, I Peschiera, I Ferlenghi, R Cozzi, N Norais, MM Giuliani, D Maione, M Pizza, R Rappuoli, O. Finco and V Masignani. Human protective response induced by meningococcus B vaccine is mediated by the synergy of multiple bactericidal epitopes. Sci. Rep. 2018; 8, 3700.
A Saul and MP Fay. Human immunity and the design of multi-component, single target vaccines. PLoS One 2007; 2, e850.
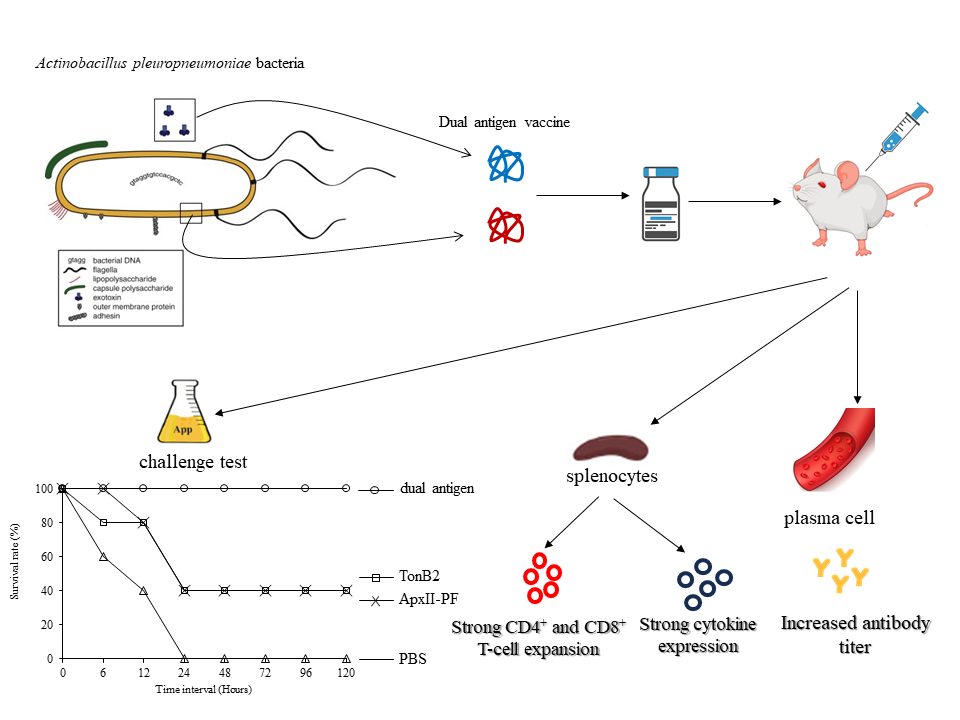
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






